TeSR™ Feeder-Free Pluripotent Stem Cell (PSC) Culture Media
TeSR™ Pluripotent Stem Cell Culture Media
Feeder-Free Media for Human ES and iPS Cell Reprogramming, Maintenance, and Differentiation
The TeSR™ family of feeder-free media are produced using rigorously pre-screened materials to ensure the highest levels of batch-to-batch consistency and experimental reproducibility, allowing you to minimize variation in your research. Each medium is based on published formulations1-3 from the laboratory of James Thomson, and allows researchers to maintain high quality human pluripotent stem cell (hPSC) culture systems. These products provide a continuous TeSR™ media-based workflow, from generation of induced pluripotent stem (iPS) cells, to maintenance, differentiation and cryopreservation of embryonic stem (ES) and iPS cells.
Why Use mTeSR™ and the TeSR™ Media Family?
- Feeder-free hPSC culture minimizes variability by limiting the presence of undefined components and immunogenic material.
- mTeSR™ is the most widely published medium for hPSC culture with > 1500 peer-reviewed publications.
- The same base formulation in each medium allows for establishment of a continuous TeSR™ media-based workflow.
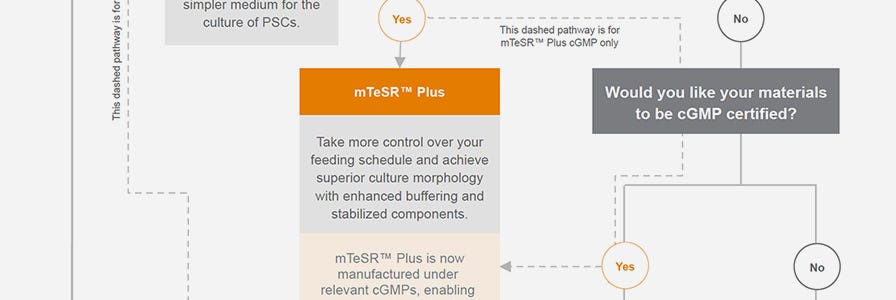
Choose Your TeSR™ Maintenance Medium
Use the Interactive Product Finder or this infographic to choose the TeSR™ hPSC culture medium that is best suited for your needs.
cGMP hPSC Maintenance Medium
Think forward to the clinic in your hPSC research. Our stabilized feeder-free hPSC maintenance medium, mTeSR™ Plus, is now manufactured and tested following relevant cGMPs under a certified quality management system. Find more information about regulatory compliance at STEMCELL >
How is mTeSR™ Plus different from other maintenance media?
mTeSR™ Plus was designed based on the formulation of mTeSR™1. This version contains stabilized components including FGF2 and unlike other media offers enhanced buffering to reduce medium acidification so that cell quality is preserved during skipped media changes.
Advantages:
- Enhanced buffering and stabilized FGF2 support cell quality while allowing for alternate feeding schedules
- Supports superior culture morphology and cell growth characteristics
- Enables heightened single-cell survival when used with CloneR™
- Fully compatible with established genome editing and differentiation protocols
- Manufactured under relevant cGMPs, enabling a seamless transition from fundamental research to drug and cell therapy development
Learn more about mTeSR™ Plus and try it in your own lab.
Consistent differentiation of ES and iPS cell lines can be challenging. We recommend our STEMdiff™ suite of products for optimal and reproducible differentiation.
What Are the Functions of Cytokines in TeSR™ Media and Their Impact on hPSC Culture?
- promotes cell survival and proliferation, while also inhibiting differentiation to specific lineages (e.g. cardiomyocytes). It is present in all of our TeSR™ media (except for TeSR™-E5).
- is an important cytokine for hPSC self-renewal and expansion and can be found in the TeSR™ reprogramming (ReproTeSR™, TeSR™-E7™) and maintenance (, , ) media.
- inhibits reprogramming and is important for maintenance of hPSC pluripotency. TGFβ is found in all three TeSR™ maintenance media (, , ).
Brand History
In 2006, Dr. Tenneille Ludwig and colleagues of Dr. James Thomson’s lab at the University of Wisconsin reported the derivation of a new ES cell line in fully defined, feeder-free culture conditions.1,2 This first defined medium significantly improved human ES cell culture, and was commercially released as mTeSR™1, becoming the most widely published feeder-free medium, used in over 1100 peer-reviewed publications. Later, a xeno-free medium based on the same formulation was released as TeSR™2. In 2012, a new, low-protein maintenance medium called TeSR™-E8™ was released. Based on the E8 formulation3 published by Dr. Guokai Chen of Dr. James Thomson’s lab, TeSR™-E8™ contains only the most essential media components required, thereby providing a simpler medium for maintenance of hPSCs.
More recently, mTeSR™ Plus (2019) and TeSR™-AOF (Animal Origin-Free; 2021) have been released as part of STEMCELL Technologies' TeSR™ product line. mTeSR™-Plus, a feeder-free maintenance medium, allows for weekend-free schedules and differs from mTeSR™1 in its enhanced pH buffering. TeSR™-AOF is guaranteed to be free of human and animal materials to the secondary level of manufacturing, providing users with peace-of-mind around viral safety. mTeSR™ Plus, TeSR™-AOF, and mTeSR™1 are all manufactured under relevant cGMPs.
In addition to TeSR™ maintenance media, STEMCELL Technologies has developed TeSR™-based media to support other facets of the pluripotent stem cell research workflow, including media optimized for reprogramming fibroblasts (TeSR™-E7™), reprogramming blood cells and fibroblasts (ReproTeSR™), differentiation (TeSR™-E6 and TeSR™-E5), and cryopreservation (mFreSR™ and FreSR™-S).
Scientific Resources
Quality Control for Pluripotent Stem Cells
Get to know the key quality attributes of hPSC cultures, including techniques for maintaining and assessing genomic integrity, pluripotency, and morphology.
Panel: Challenges in Ensuring hPSC Quality
Hear global experts discuss key issues impacting the use of human pluripotent stem cells in this series of webinars provided in partnership with Nature Research.
Explore more helpful resources for your hPSC research in our pluripotent resource centers and cell culture methods library
Key Applications
Toxicity Testing with Human iPS Cells
Kleinstreuer NC, Smith AM, West PR, Conard KR, Fontaine BR, Weir-Hauptman AM, Palmer JA, Knudsen TB, Dix DJ, Donley ELR and Cezar GG (2011), Identifying developmental toxicity pathways for a subset of ToxCast chemicals using human embryonic stem cells and metabolomics, Toxicology and Applied Pharmacology., November, 2011. Vol. 257(1), pp. 111-121.
Liang P, Lan F, Lee AS, Gong T, Sanchez-Freire V, Wang Y, Diecke S, Sallam K, Knowles JW, Wang PJ, Nguyen PK, Bers DM, Robbins RC and Wu JC (2013), Drug screening using a library of human induced pluripotent stem cell-derived cardiomyocytes reveals disease-specific patterns of cardiotoxicity. Circulation., April, 2013. Vol. 127(16), pp. 1677-1691.
Liu J, Sun N, Bruce MA, Wu JC and Butte MJ (2012), Atomic Force Mechanobiology of Pluripotent Stem Cell-Derived Cardiomyocytes, PLoS ONE., May, 2012. Vol. 7(5), pp. e37559.
Mehta A, Chung YY, Ng A, Iskandar F, Atan S, Wei H, Dusting G, Sun W, Wong P and Shim W (2011), Pharmacological response of human cardiomyocytes derived from virus-free induced pluripotent stem cells, Cardiovascular Research., September, 2011. Vol. 91(4), pp. 577-586.
Differentiating to Hematopoietic Cells
Carpenter L, Malladi R, Yang C-T, French A, Pilkington KJ, Forsey RW, Sloane-Stanley J, Silk KM, Davies TJ, Fairchild PJ, Enver T and Watt SM (2011), Human induced pluripotent stem cells are capable of B-cell lymphopoiesis, Blood., April, 2011. Vol. 117(15), pp. 4008-4011.
Dravid G, Zhu Y, Scholes J, Evseenko D and Crooks GM (2011), Dysregulated gene expression during hematopoietic differentiation from human embryonic stem cells, Mol Ther. Vol. 19, pp. 768-81.
Niwa A, Heike T, Umeda K, Oshima K, Kato I, Sakai H, Suemori H, Nakahata T and Saito MK (2011), A novel serum-free monolayer culture for orderly hematopoietic differentiation of human pluripotent cells via mesodermal progenitors, PLoS One. Vol. 6, pp. e22261.
Salvagiotto G, Burton S, Daigh CA, Rajesh D, Slukvin II and Seay NJ (2011), A Defined, Feeder-Free, Serum-Free System to Generate In Vitro Hematopoietic Progenitors and Differentiated Blood Cells from hESCs and hiPSCs, PLoS ONE., March, 2011. Vol. 6(3), pp. e17829.
Differentiating to Definitive Endoderm
Jaramillo M and Banerjee I (2012), Endothelial Cell Co-culture Mediates Maturation of Human Embryonic Stem Cell to Pancreatic Insulin Producing Cells in a Directed Differentiation Approach, Journal of Visualized Experiments., March, 2012. (61)
Miki T, Ring A and Gerlach J (2011), Hepatic differentiation of human embryonic stem cells is promoted by three-dimensional dynamic perfusion culture conditions, Tissue Eng Part C Methods. Vol. 17, pp. 557-68.
Mou H, Zhao R, Sherwood R, Ahfeldt T, Lapey A, Wain J, Sicilian L, Izvolsky K, Lau FH, Musunuru K, Cowan C and Rajagopal J (2012), Generation of Multipotent Lung and Airway Progenitors from Mouse ESCs and Patient-Specific Cystic Fibrosis iPSCs, Cell Stem Cell., April, 2012. Vol. 10(4), pp. 385-397.
Spence JR, Mayhew CN, Rankin SA, Kuhar MF, Vallance JE, Tolle K, Hoskins EE, Kalinichenko VV, Wells SI, Zorn AM, Shroyer NF and Wells JM (2011), Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro, Nature., February, 2011. Vol. 470(7332), pp. 105-109.
Scale-Up and Bioreactor Culture
Oh SKW, Chen AK, Mok Y, Chen X, Lim U-M, Chin A, Choo ABH and Reuveny S (2009), Long-term microcarrier suspension cultures of human embryonic stem cells, Stem Cell Research., May, 2009. Vol. 2(3), pp. 219-230.
Olmer R, Haase A, Merkert S, Cui W, Palecek J, Ran C, Kirschning A, Scheper T, Glage S, Miller K, Curnow EC, Hayes ES and Martin U (2010), Long term expansion of undifferentiated human iPS and ES cells in suspension culture using a defined medium, Stem Cell Res. Vol. 5, pp. 51-64.
Singh H, Mok P, Balakrishnan T, Rahmat SN and Zweigerdt R (2010), Up-scaling single cell-inoculated suspension culture of human embryonic stem cells, Stem Cell Res. Vol. 4, pp. 165-79.
Zweigerdt R, Olmer R, Singh H, Haverich A and Martin U (2011), Scalable expansion of human pluripotent stem cells in suspension culture, Nature Protocols. Vol. 6(5), pp. 689-700.
Differentiating to Cardiomyocytes
Hazeltine LB, Simmons CS, Salick MR, Lian X, Badur MG, Han W, Delgado SM, Wakatsuki T, Crone WC, Pruitt BL and Palecek SP (2012), Effects of Substrate Mechanics on Contractility of Cardiomyocytes Generated from Human Pluripotent Stem Cells, International Journal of Cell Biology. Vol. 2012, pp. 1-13.
Lian X, Hsiao C, Wilson G, Zhu K, Hazeltine LB, Azarin SM, Raval KK, Zhang J, Kamp TJ and Palecek SP (2012), Robust cardiomyocyte differentiation from human pluripotent stem cells via temporal modulation of canonical Wnt signaling, Proceedings of the National Academy of Sciences., July, 2012. Vol. 109(27), pp. 10759-10760.
Mehta A, Chung YY, Ng A, Iskandar F, Atan S, Wei H, Dusting G, Sun W, Wong P and Shim W (2011), Pharmacological response of human cardiomyocytes derived from virus-free induced pluripotent stem cells, Cardiovascular Research., September, 2011. Vol. 91(4), pp. 577-586.
Zhang H, Zou B, Yu H, Moretti A, Wang X, Yan W, Babcock JJ, Bellin M, McManus OB, Tomaselli G, Nan F, Laugwitz K-L and Li M (2012), Modulation of hERG potassium channel gating normalizes action potential duration prolonged by dysfunctional KCNQ1 potassium channel, Proceedings of the National Academy of Sciences., July, 2012. Vol. 109(29), pp. 11866-11871.
References
- Ludwig TE et al. (2006) Feeder-independent culture of human embryonic stem cells. Nat Methods 3(8): 637–46.
- Ludwig TE et al. (2006) Derivation of human embryonic stem cells in defined conditions. Nat Biotechnol 24(2): 185–7.
- Chen G et al. (2011) Chemically defined conditions for human iPSC derivation and culture. Nat Methods 8(5): 424–9.