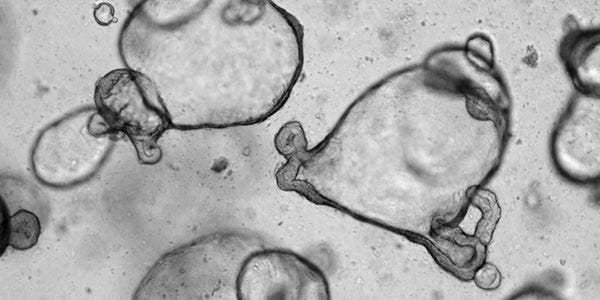
Intestinal
Since their development in 2009, intestinal organoids have revolutionized intestinal epithelial cell culture by providing an organotypic model system that can be readily maintained and manipulated in vitro. Through re-creation of the intestinal structure and cellular complement, intestinal organoids serve as valuable model systems for a range of uses from basic research to diagnostic and translational applications.
Start by selecting your research area to explore related or complementary products for each workflow stage.