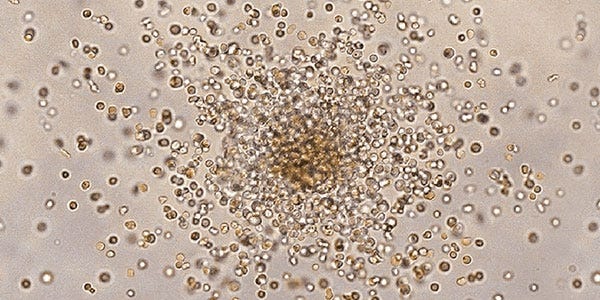
Hematopoietic
The ability of hematopoietic stem and progenitor cells (HSPCs) to self-renew and differentiate is fundamental for the formation and maintenance of life-long hematopoiesis; deregulation of these processes may lead to severe clinical consequences.
Select your research area below to start exploring related and complementary products for each stage.