STEMdiff™ T Cell Kit
For expansion and differentiation of hPSCs to T cells
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration
-
 Reversible Strainers
Reversible StrainersReversible strainer for filtration of single cells and isolation of cellular aggregates, including embryoid bodies and spheroids
-
 AggreWell™400
AggreWell™400Microwell culture plates for easy and reproducible production of embryoid bodies and spheroids
-
 Y-27632 (Dihydrochloride)
Y-27632 (Dihydrochloride)RHO/ROCK pathway inhibitor; Inhibits ROCK1 and ROCK2
-
Labeling Antibodies
Compatible antibodies for purity assessment of isolated cells
Overview
The STEMdiff™ T Cell Kit protocol first uses animal component-free STEMdiff™ Hematopoietic - EB reagents for the generation of embryoid bodies (EBs) from hPSCs which are then further differentiated into CD34+ cells. The kit includes all the EB reagents necessary for this step:
• STEMdiff™ Hematopoietic - EB Basal Medium
• STEMdiff™ Hematopoietic - EB Supplement A
• STEMdiff™ Hematopoietic - EB Supplement B
The CD34+ cells are then further directed to differentiate to T cells using StemSpan™ reagents:
• StemSpan™ SFEM II
• StemSpan™ Lymphoid Progenitor Expansion Supplement (10X)
• StemSpan™ Lymphoid Differentiation Coating Material (100X)
• StemSpan™ T Cell Progenitor Maturation Supplement (10X)
For your convenience, all STEMdiff™ Hematopoietic - EB and StemSpan™ reagents included with the kit are also available individually.
Data Figures
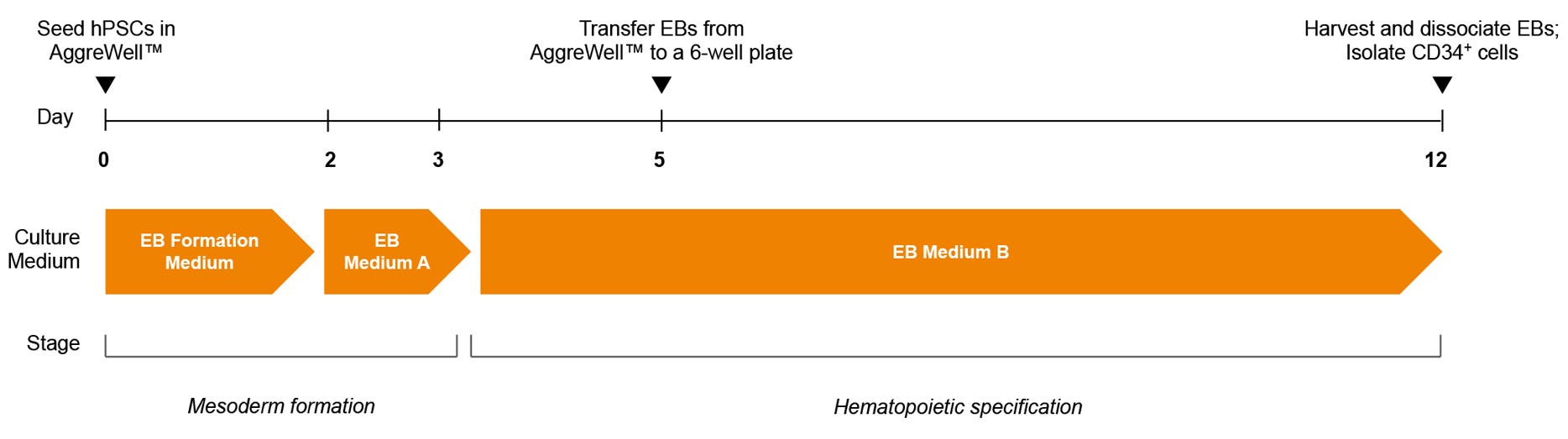
Figure 1. STEMdiff™ Hematopoietic - EB Progenitor Differentiation Protocol
hPSCs are harvested and dissociated into a single-cell suspension prior to seeding into AggreWell™ plates in EB Formation Medium (EB Medium A + 10 μM Y-27632) to form 600-cell aggregates. After 3 days of mesoderm formation, the medium is changed to EB Medium B to induce hematopoietic lineage differentiation. On day 5, EBs are transferred onto non-tissue culture-treated plates. After a total of 12 days, EBs are harvested and dissociated, and CD34+ cells are then enriched by EasySep™ positive selection.
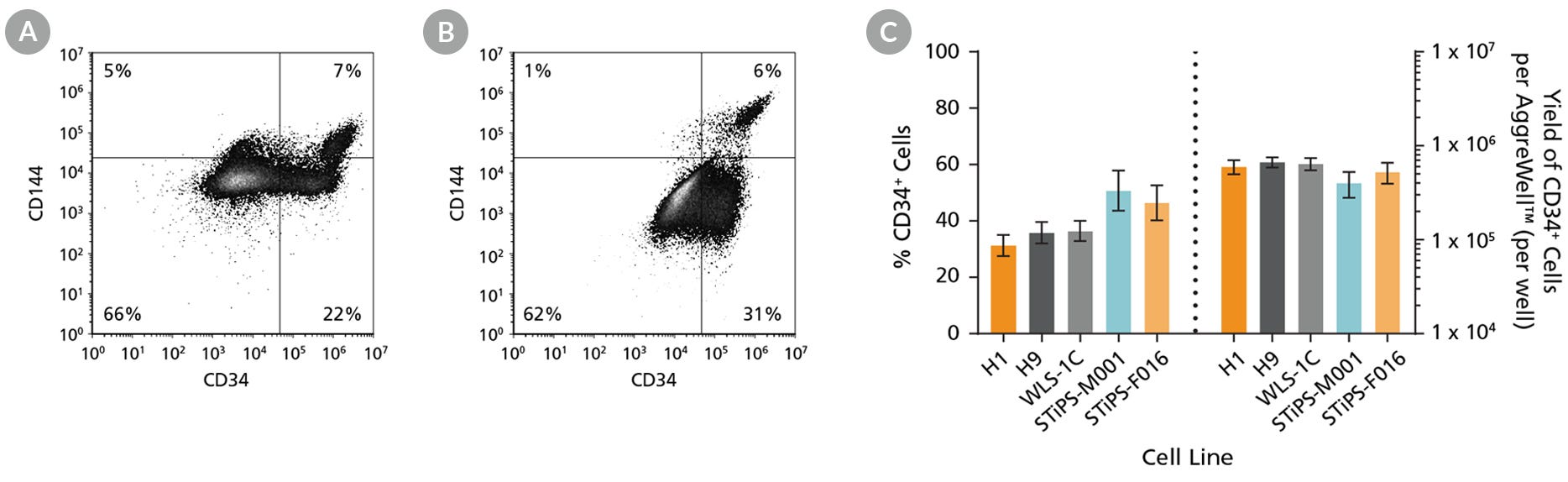
Figure 2. hPSCs Differentiate to CD34+ Hematopoietic Progenitor Cells After 12 Days of Culture
Human ES and iPS cells were induced to differentiate to CD34+ cells using the 12-day protocol shown in Figure 1. At the end of the culture period, cells were harvested, dissociated into a single-cell suspension, and analyzed by flow cytometry for CD34 and CD144 expression. Dead cells were excluded by light-scatter profile and DRAQ7™ staining. Representative flow cytometry plot for (A) ES (H1) cell-derived and (B) iPS (STiPS-F016) cell-derived cells analyzed on day 12 are shown. (C) The average frequency of viable CD34+ cells on day 12 (before CD34+ cell isolation) for two ES cell lines (H1 and H9) and three iPS cell lines (WLS-1C, STiPS-M001, and STiPS-F016) ranged between 31% and 51%. The average yield of CD34+ cells produced per well of a 6-well AggreWell™400 plate ranged between 4.0 x 105 and 6.7 x 105. Data are shown as mean ± SEM (n = 9 - 35). Results are representative of populations prior to magnetic cell separation.
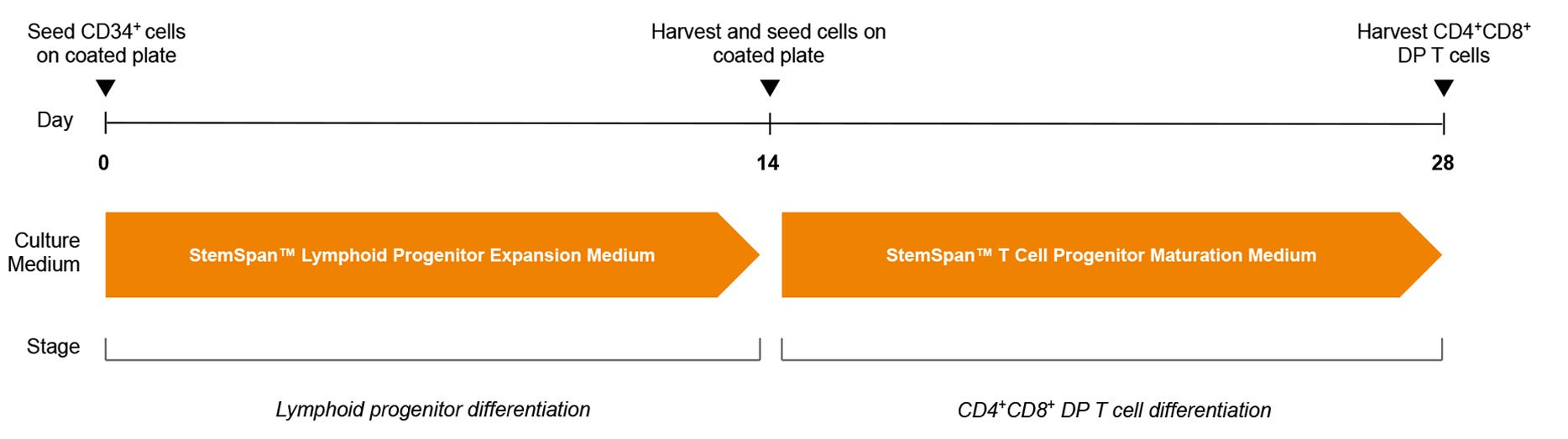
Figure 3. T Cell Generation Protocol
hPSC-derived CD34+ cells are seeded in StemSpan™ Lymphoid Progenitor Expansion Medium on plates coated with StemSpan™ Lymphoid Differentiation Coating Material. On day 14, cells at the lymphoid progenitor stage are harvested and reseeded in StemSpan™ T Cell Progenitor Maturation Medium onto coated plates for further differentiation to CD4+CD8+ DP T cells. The DP T cells are harvested after 28 days.
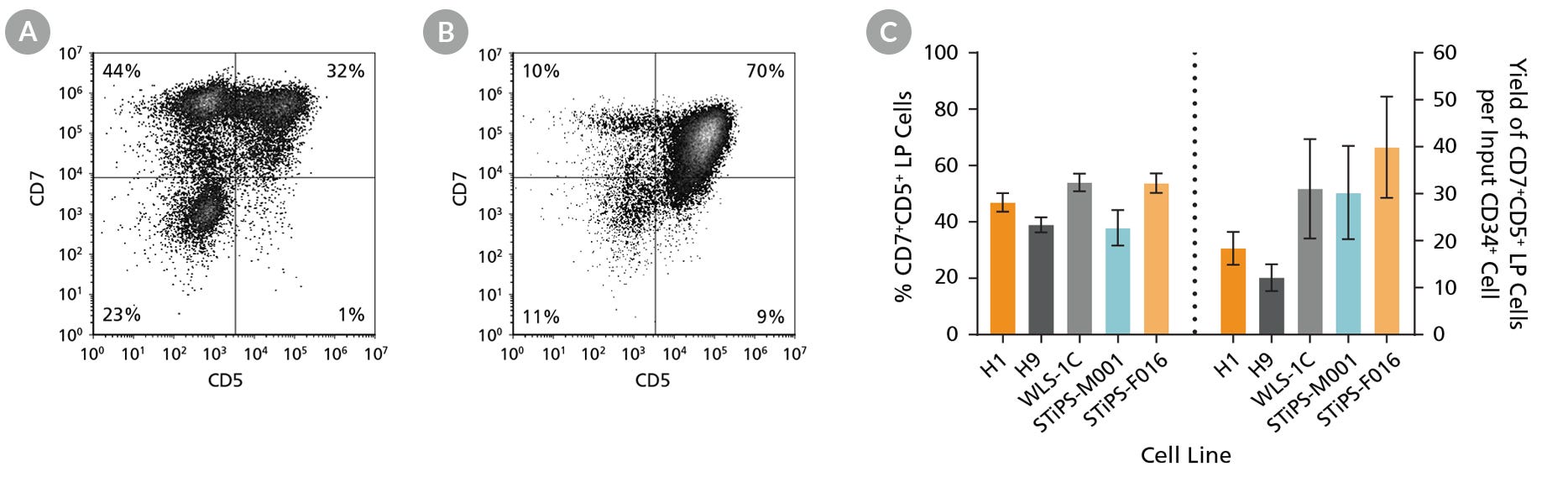
Figure 4. hPSC-Derived CD34+ Cells Differentiate to CD5+CD7+ Lymphoid Progenitor Cells Over 14 Days of Culture
hPSC-derived CD34+ cells were cultured for 14 days in StemSpan™ SFEM II + StemSpan™ Lymphoid Progenitor Expansion Supplement on plates coated with StemSpan™ Lymphoid Differentiation Coating Material (Figure 3). Cells were harvested and analyzed for CD7 and CD5 expression by flow cytometry. Representative flow cytometry plots for (A) ES (H1) cell-derived and (B) iPS (STiPS-F016) cell-derived cells are shown. (C) The average frequency of viable CD7+CD5+ lymphoid progenitor cells on day 14 ranged between 38% and 54%, and the average yield of lymphoid progenitor cells produced per input hPSC-derived CD34+ cell was between 12 and 40. Data are shown as mean ± SEM (n = 8 - 32).
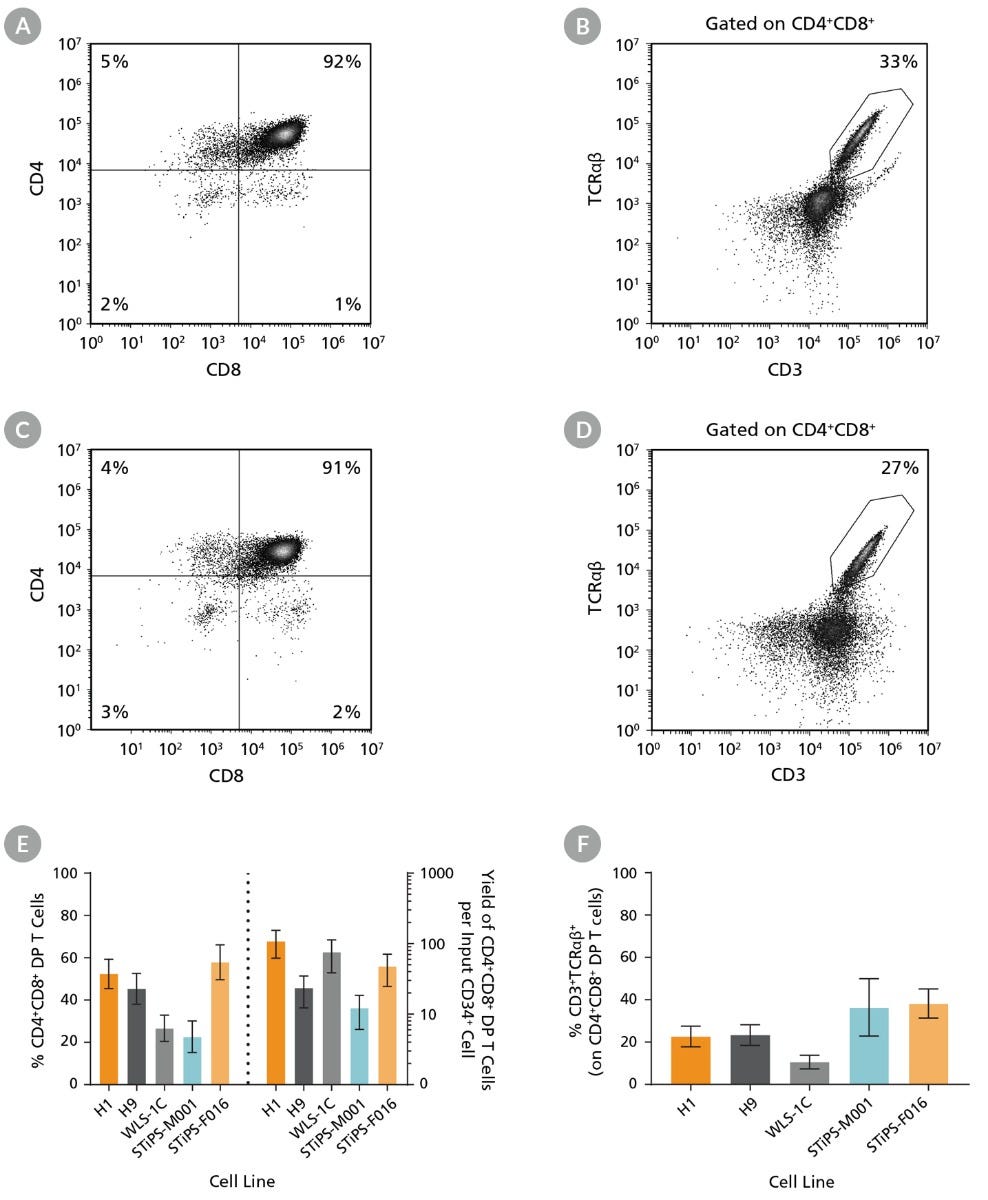
Figure 5. CD4+CD8+ DP T Cells Can Be Generated from Human hPSC-Derived CD34+ Cells After 28 Days of Culture
DP T cells were differentiated from hPSC-derived CD34+ cells as described (Figure 3). Cells were harvested and analyzed for expression of CD3, CD4, CD8, and TCRαβ by flow cytometry. Representative flow cytometry plots are shown for (A,B) ES (H1) cell-derived and (C,D) iPS (STiPS-F016) cell-derived cells. (E) The average frequency of viable CD4+CD8+ DP T cells on day 28 ranged between 23% and 58%, and the average yield of DP T cells produced per input hPSC-derived CD34+ cell was between 12 and 108. (F) The average frequency of CD3+TCRαβ+ expressed on DP T cells ranged between 11% and 38%. Data are shown as mean ± SEM (n = 6 - 17).
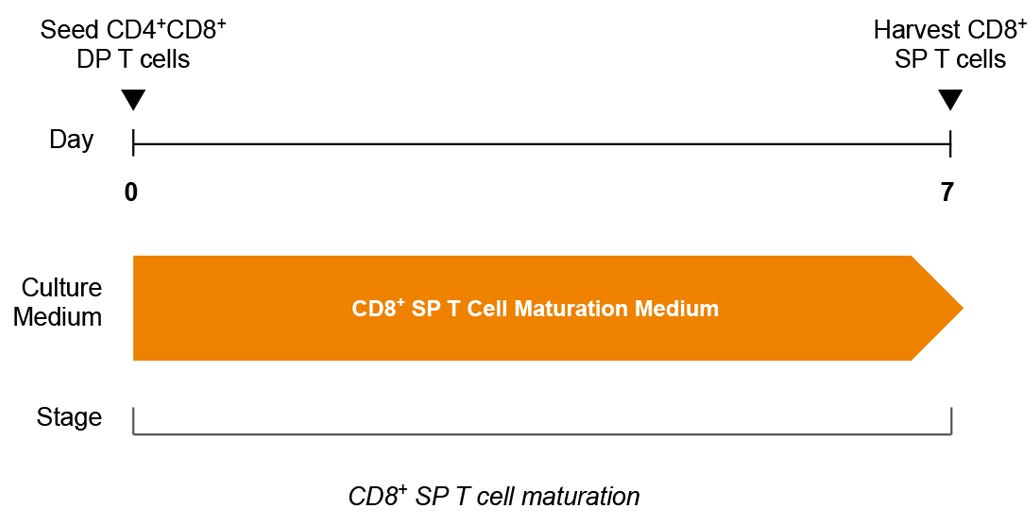
Figure 6. Optional CD8+ SP T Cell Maturation Protocol
DP T cells are seeded in CD8+ SP T Cell Maturation Medium supplemented with ImmunoCult™ T Cell Activator on plates coated with StemSpan™ Lymphoid Differentiation Coating Material. CD8+ SP T cells can be harvested after 7 days.
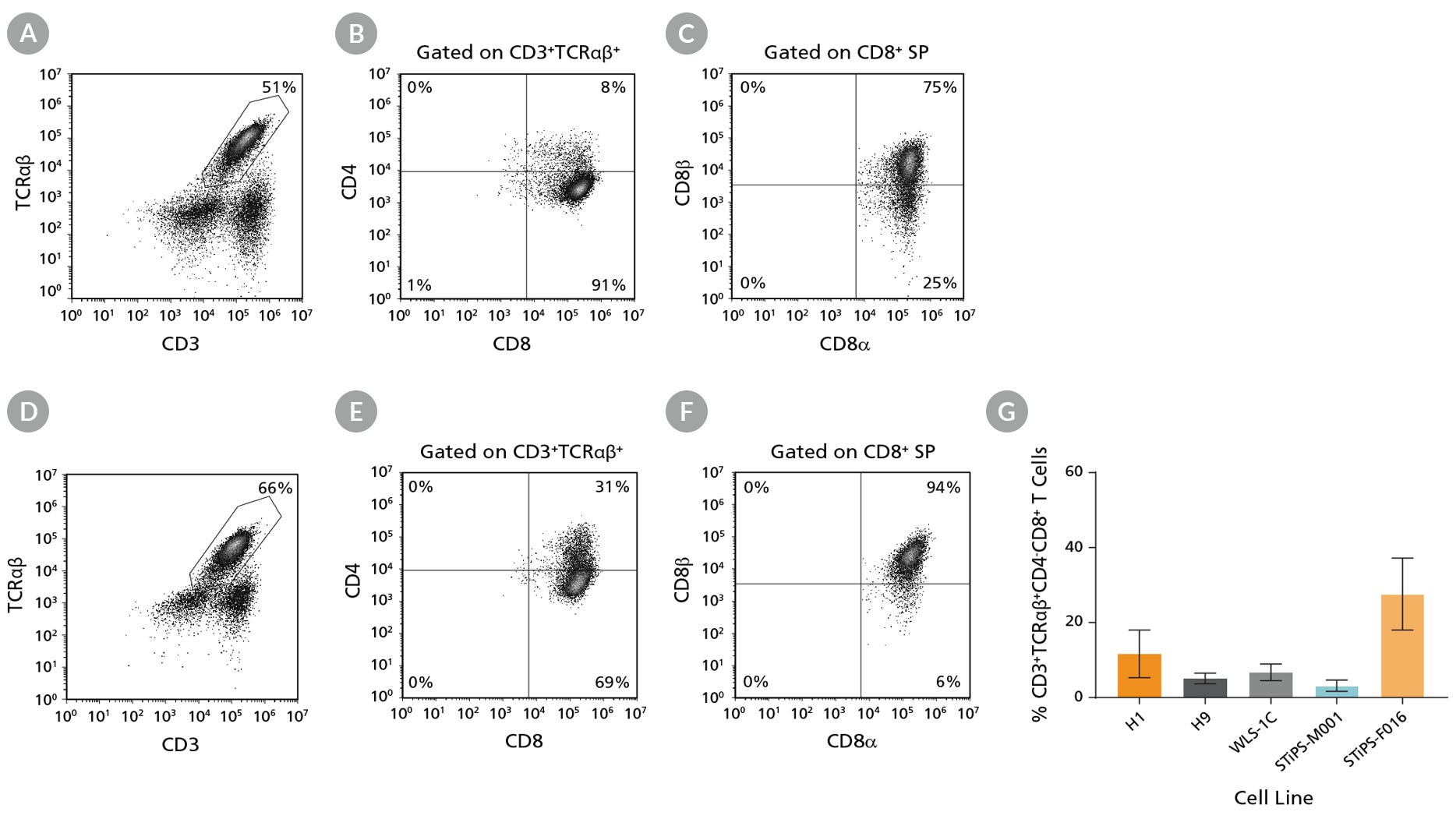
Figure 7. hPSC-Derived CD4+CD8+ DP T Cells Are Able to Mature to CD8+ SP T Cells
hPSC-derived CD34+ cells were first differentiated to DP T cells during 28 days of culture (Figure 3), and were then matured to CD8+ SP T cells using an additional 7-day maturation protocol (Figure 6). Cells were harvested and analyzed by flow cytometry for expression of (A,D) CD3 and TCRαβ, (B,E) CD4 and CD8 (gated on CD3+TCRαβ+), and (C,F) CD8α and CD8β (gated on CD8+ SP). Representative results for (A,B,C) ES (H1) cell-derived and (D,E,F) iPS (STiPS-F016) cell-derived cells are shown. (G) The average frequency of CD3+TCRαβ+CD4-CD8+ (CD8+SP) T cells was between 3% and 28%. Data are shown as mean ± SEM (n = 2 - 9).
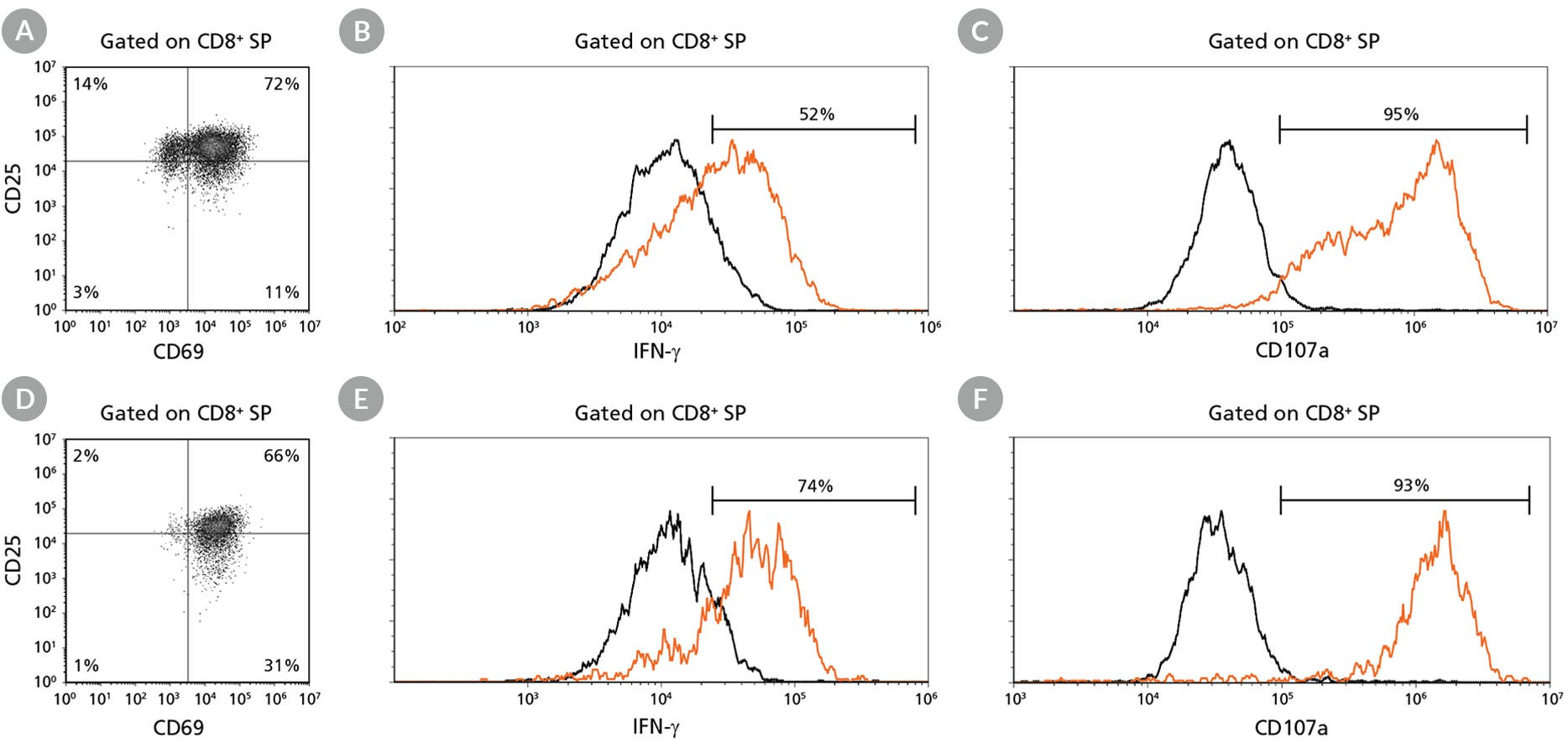
Figure 8. hPSC-Derived CD8+ SP T Cells Can Be Induced to Express Activation Markers CD25 and CD69, Degranulate, and Produce IFN-γ
hPSC-derived CD34+ cells were first differentiated to DP T cells during 28 days of culture and then matured to CD8+ SP T cells using an additional 7-day maturation protocol (Figures 3 and 6). CD8+ SP T cells were sorted using fluorescence-activated cell sorting and cultured for 7 days in ImmunoCult™-XF T Cell Expansion Medium supplemented with IL-2 and stimulated with ImmunoCult™ Human CD3/CD28 T Cell Activator. Cells were harvested and analyzed by flow cytometry for expression of (A,D) CD25 and CD69. For assessment of degranulation and cytokine production, at 4 hours prior to harvest, some cells were also stimulated with phorbol 12-myristate 13-acetate (PMA) and ionomycin. After one hour, monensin and brefeldin A were added to each well to inhibit protein transport. Control (unstimulated; black histogram) and PMA + ionomycin-stimulated cells (orange histogram) were additionally analyzed by flow cytometry for surface expression of (C,F) CD107a (lysosomal-associated membrane protein 1 or LAMP-1) indicating degranulation and intracellular expression of (B,E) IFN-γ (gated on CD8+ SP). Representative results for (A,B,C) ES (H1) cell-derived and (D,E,F) iPS (STiPS-F016) cell-derived cells are shown.
Protocols and Documentation
Find supporting information and directions for use in the Product Information Sheet or explore additional protocols below.
Applications
This product is designed for use in the following research area(s) as part of the highlighted workflow stage(s). Explore these workflows to learn more about the other products we offer to support each research area.
Resources and Publications
Educational Materials (12)
Related Products
-
 StemSpan™ SFEM II
StemSpan™ SFEM IISerum-free medium for culture and expansion of hematopoietic cells
-
 StemSpan™ Lymphoid Progenitor Expansion Sup...
StemSpan™ Lymphoid Progenitor Expansion Sup...Supplement for the expansion and differentiation of human CD34+ cells to lymphoid progenitor cells
-
 StemSpan™ Lymphoid Differentiation Coating ...
StemSpan™ Lymphoid Differentiation Coating ...Coating material for the expansion and differentiation of lymphoid cells
-
 StemSpan™ T Cell Progenitor Maturation Supp...
StemSpan™ T Cell Progenitor Maturation Supp...Supplement for the maturation of lymphoid progenitor cells to T cells
-
 StemSpan™ T Cell Generation Kit
StemSpan™ T Cell Generation KitComplete kit for expansion and differentiation of human CD34+ hematopoietic progenitor cells to T cells
Item added to your cart

STEMdiff™ T Cell Kit
PRODUCTS ARE FOR RESEARCH USE ONLY AND NOT INTENDED FOR HUMAN OR ANIMAL DIAGNOSTIC OR THERAPEUTIC USES UNLESS OTHERWISE STATED. FOR ADDITIONAL INFORMATION ON QUALITY AT STEMCELL, REFER TO WWW.STEMCELL.COM/COMPLIANCE.












