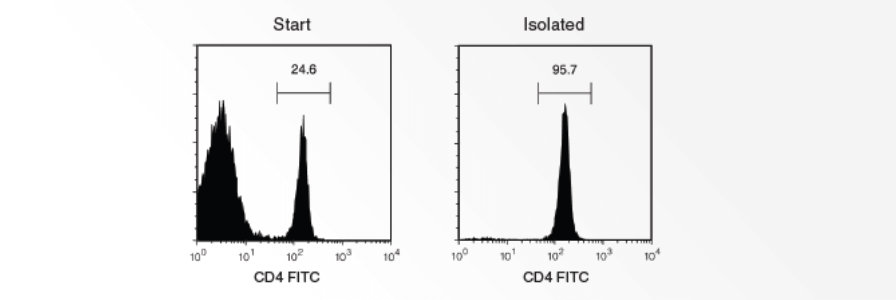
Isolate Virtually Any Cell Type Using EasySep™ or EasySep™ Release Indirect Selection Kits
Use any biotinylated, FITC-, PE- or APC-conjugated antibody to label your desired cells and mix with EasySep™ reagents to obtain highly purified cells
*For use with other species, we recommend adding a species-specific blocker to cell the suspension to minimize non-specific binding
Please refer to the following tips to ensure optimal isolation of your cell type of interest:
- Ensure cells are in single cell suspension.
- Add species-specific blocker to cell suspension to minimize non-specific binding. For more information on using Fc receptor (FcR) blockers, please refer to our Technical Tip: Importance of Using an FcR Blocker During Cell Separation Procedures .
- We recommend titrating your primary Biotin/FITC/PE/APC-conjugated antibody. The desired final concentration will be the lowest concentration of your primary antibody that gives the greatest fluorescence intensity shift by flow cytometry (for best results, aim to achieve 2 log staining intensity above background). We suggest using a final concentration of primary antibody between 0.3 - 3.0 µg/mL of cells, the optimal concentration will vary depending on the starting frequency of your cells of interest.
If the starting percentage of your desired cell type is:
- <10%, use a final concentration of antibody between 0.3 - 0.5 µg/mL
- 10-40%, use a final concentration of antibody between 0.5 - 1.0 µg/mL
- 40-60%, use a final concentration of antibody between 1.0 - 2.0 µg/mL
- >60% use a final concentration of antibody between 2.0 - 3.0 µg/mL *
- Some antibodies used to assess purity of the isolated cells will be blocked by the initial selection antibody, in which case you should stain with an alternate antibody. You could use an antibody for a different cell surface antigen (i.e. assess purity using alternate marker expressed on your target cells), or use a fluorochrome-conjugated antibody that will recognize the primary antibody (i.e. a goat anti-mouse antibody, such as GAM-FITC).
For further assistance and information, please contact techsupport@stemcell.com.
Related Resource
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration