Genetic Basis of Autoimmunity
Immunology Feature
In 2007, human genetic variation was named “Breakthrough of the Year” by Science. The ability to detect variations in the human genome has allowed scientists to identify genes and variations associated with a multitude of human diseases, including autoimmunity, leading to subsequent breakthroughs in how we predict, treat and prevent these diseases.
Autoimmune diseases affect ~10% of the population, and both genetic and environmental factors play a role in their development1. With emerging genomic technologies, many research studies have provided great insight into the genetic causes of autoimmune diseases. For example, the Hafler and Bernstein labs have joined forces to identify causal variants for twenty one autoimmune diseases2. The following three studies reveal that genetic variants of CTLA4, PTPN22, IL2RA and IL23R can contribute to the susceptibility to autoimmune diseases.
CTLA4
Autosomal dominant immune dysregulation syndrome in humans with CTLA4 mutations3
Nature Medicine
Cytotoxic T-lymphocyte protein 4 (CTLA-4) is a co-inhibitory receptor and a negative regulator of immunity. The expression of CTLA-4 is critical for the suppressive function of regulatory T cells (Tregs) and the prevention of autoimmunity. Mechanistically, CTLA-4 has been shown to both intrinsically dampen T cell signaling and extrinsically remove CD80 and CD86 from antigen-presenting cells via transendocytosis4,5. In this article, Schubert et al. identified a CTLA4 mutation that causes an autosomal dominant immune dysregulation and autoimmunity in humans. The authors further found that this mutation impairs CTLA-4 expression and the suppressive function of Tregs.
Cell separation: EasySep™ Human CD4+ T Cell Isolation Kit
PTPN22
The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep degradation associated with lymphocyte and dendritic cell hyperresponsiveness6
Genetics
Protein tyrosine phosphatase, non-receptor type 22 (PTPN22) encodes for Lyp/Pep, a tyrosine phosphatase expressed on hematopoietic cells that negatively regulates immune cell signaling. The existence of mutations in the PTPN22 gene is a major risk factor for autoimmune diseases, including type 1 diabetes and rheumatoid arthritis. In this article, Zhang et al. showed that disease-associated variants of PTPN22 causes the over-activation of lymphocytes and dendritic cells by increasing the susceptibility of Lyp/Pep to calpain- and proteasome-mediated degradation. By increasing this immune cell hyperresponsiveness, disease-associated variants of PTPN22 reduce immune tolerance and promote the development of autoimmunity.
Cell separation: EasySep™ Mouse CD11c Positive Selection Kit
IL2RA and IL23R
Intersection of population variation and autoimmunity genetics in human T cell activation7
Science
CD4+ T cells can be categorized into several T helper subsets, and the over-activation of these subsets is associated with autoimmune diseases. For example, increased Th1 and Th17 responses have been associated with rheumatoid arthritis and multiple sclerosis8,9. These studies suggest that interindividual variations in T cell activation and polarization can affect how prone an individual is to mounting Th1- or Th17-biased responses. In this paper, Ye et al. sought to determine how genetic factors associated with different ancestries contribute to variations in the expression levels of cytokines, chemokines and their receptors in activated T cells. The authors were able to identify genetic variants associated with increased expressions of IL2RA and IL23R, cytokine receptors that are critical for the activation and maintenance of CD4+ T cell subsets. Interestingly, variants of both of these genes have previously been associated with autoimmune diseases, such as type 1 diabetes in the case of IL2RA10.
Cell separation: RosetteSep™ Human CD4+ T Cell Enrichment Cocktail
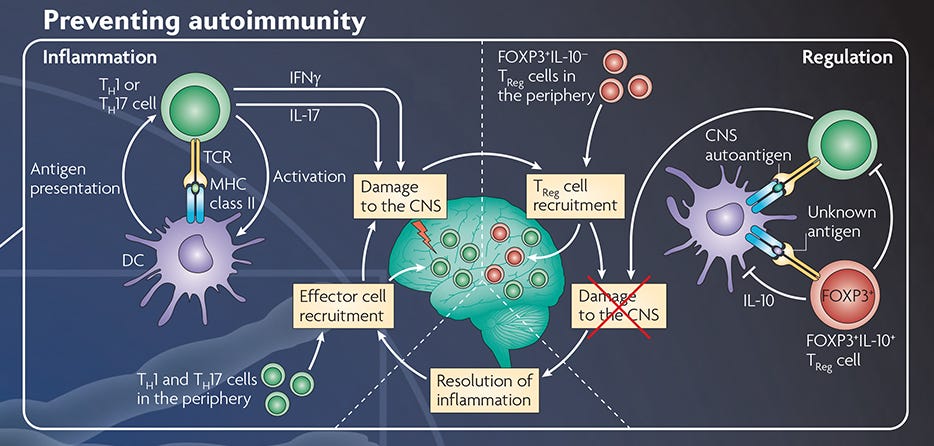
Figure 1. Preventing Th1/Th17-Driven Autoimmunity
Adapted from the Nature Reviews Immunology Regulatory T Cell wallchart, created in partnership with STEMCELL Technologies. Request a copy of the full Regulatory T Cell wallchart for your lab.
The ability to detect disease-associated variants has allowed us to implement early monitoring and treatment in affected individuals. Researchers are investigating ways to effectively apply these discoveries for disease prevention. Perhaps regulatory T cells can be used to prevent autoimmunity (Figure 1), or immune cell engineering can in the future be used to specifically and safely modify disease-associated variants in humans. Regardless, continued research aimed at understanding the genetic basis of autoimmunity will allow scientists to develop more effective strategies for the prevention, early detection and therapeutic intervention of autoimmune diseases.
Explore More Immunology Features
From the Authors
"I have been using EasySep™ to isolate human T cell subsets and monocytes for 5 years. Compared to other cell isolation products, EasySep™ kits save time and are more cost effective. I get very high purities of my target cells. STEMCELL's technical experts have also helped me customize cell isolation kits and design custom protocols."
Tiezheng Hou (MD, PhD), Research Fellow, David Sansom Lab, University College London
To help immunologists like Tiezheng isolate highly purified immune cells faster and easier, we've been developing innovative cell isolation technologies for over 20 years.
Resources for Your Autoimmunity Research
References
- Marson A et al. (2015) J Clin Invest 125(6): 2234–2241.
- Farh KK et al. (2015) Nature 518(7539): 337–343.
- Schubert D et al. (2014) Nat Med 20(12):1410-1416.
- Walker LSK et al. (2011) Nat Rev Immunol 11: 852-863.
- Hou TZ et al. (2015) J Immunol 194: 2148-2159.
- Zhang J et al. (2011) Nat Genet 43(9): 902-907.
- Ye CJ et al. (2014) Science 345(6202): 1254665.
- Burkett PR et al. (2015) J Clin Invest 125(6): 2211-2219.
- Damsker JM et al. (2010) Ann N Y Acad Sci 1183: 211-221.
- Lowe CE et al. (2007) Nat Genet 39(9): 1074-1082.