Cancer Immunology
Immunology Feature
In 2013, the promising results of cancer immunotherapy trials led to this field of research being chosen as the "Breakthrough of the Year" by Science. With recent advances in cancer immunotherapy, researchers are increasingly interested in harnessing immune mechanisms for cancer therapy.
Immune cells have the capability to eliminate tumor cells; however, tumor cells can develop the ability to evade immune responses. For example, malignant cells can modify their antigens to avoid immune recognition, or express factors that modulate or suppress immune cells1. To add to the complexity, certain immune cells, including certain subsets of regulatory T cells and macrophages, have been shown to contribute to tumorigenic environment or cancer metastasis.2,3 Current research efforts have been placed on developing personalized strategies for cancer treatment and developing new therapies that bolster the immune response in the fight against tumor cells.
Novel Therapies Against Cancer
Cancer therapy has come a long way since the days when surgery and radiation therapy were the only treatment options. While small molecules and antibodies have long been a focus for researchers trying improve cancer treatment, cellular therapy has emerged more recently as a serious contender in the race to cure cancer.
Small Molecule Therapy
Smac mimetics and innate immune stimuli synergize to promote tumor death4
Nature Biotechnology
For years, researchers have worked diligently to target signaling nodes that contribute to tumorigenesis using small molecules (Figure 1). One example of a small molecule drug for cancer is Smac mimetic compounds (SMCs), which have been proven safe in phase 1 clinical trials. SMCs promote the apoptosis of tumor cells by antagonizing inhibitor of apoptosis proteins (IAPs) expressed on tumor cells. Inflammatory cytokines and pro-apoptotic proteins can synergize with SMCs in tumor killing in vitro. In this article, Beug et al. found that the combinatorial therapy of SMCs and poly(I:C) or CpG resulted in enhanced tumor killing, which was dependent on the stimulation of IFN-B, TNF-a and/or TNF-related apoptosis-inducing ligand (TRAIL) production. Notably, these combinatorial therapies were able to cure mouse models of cancer.
Cell separation: EasySep™ Mouse CD11b Positive Selection Kit and EasySep™ Mouse CD49b Positive Selection Kit
Smac mimetics synergize with immune checkpoint inhibitors to promote tumour immunity against glioblastoma5
Nature Communications
This study shows that combinatorial therapy using SMCs and innate immune stimulants or checkpoint inhibitors result in long-lasting anti-tumor immunity in a mouse model of glioblastoma. Beug et al. further described that this synergistic effect is dependent on TNF-α-producing cytotoxic T cells. Overall, these two studies from the Korneluk lab illustrate that small molecules directed towards direct tumor killing can complement immunotherapeutic approaches that aim to boost immune responses against tumors.
Cell separation: EasySep™ Mouse CD8 T Cell Isolation Kit
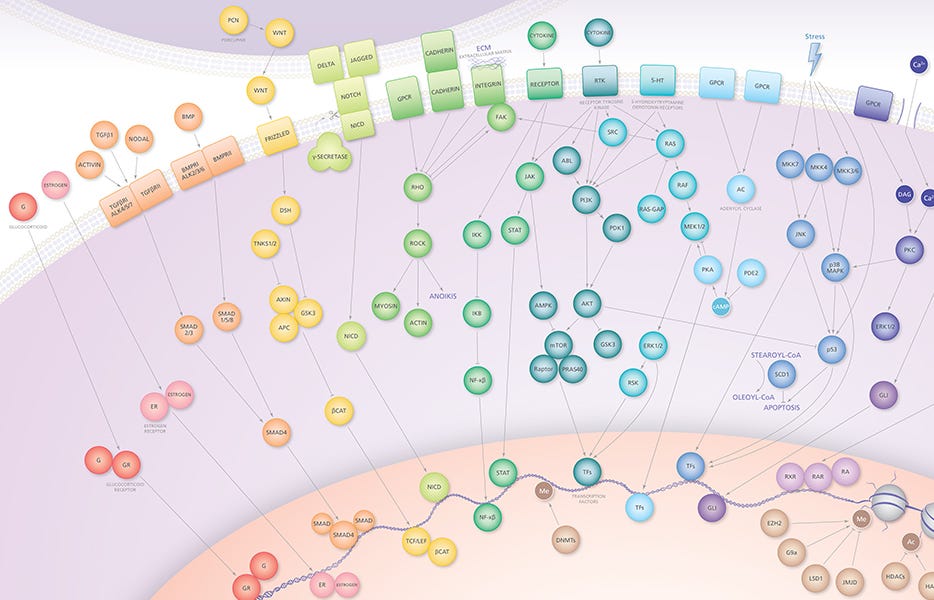
Figure 1. Small Molecule Targets in Cancer
Adapted from the Small Molecules, Big Impact in Cancer Research wallchart, created by STEMCELL Technologies. Request a copy of the Small Molecules in Cancer Research wallchart for your lab.
Antibody Therapy
CD47 blockade triggers T cell-mediated destruction of immunogenic tumors5
Nature Medicine
Antibody-based therapy has proven to be one of the most successful strategies for cancer treatment. Monoclonal antibodies can either be directed towards malignant cells to promote their inhibition and antibody-dependent cell-mediated cytotoxicity, or directed towards immunosuppressive molecules that prevent effective immune responses toward tumor cells6. CD47 is a protein that inhibits phagocytosis, and its expression has been observed in various tumor cells. Therapies that block CD47 have proven efficacious in mouse models of cancer, and antibody-mediated CD47 blockade is currently being evaluated in clinical trials. Macrophage-mediated phagocytosis of tumor cells was previously hypothesized as the mechanism behind CD47 blockade therapies; however, these early studies were performed in T cell-deficient mice. Here, Liu et al. showed that the therapeutic effect of CD47 blockade largely depends on T cell activation, and also relies on dendritic cells, interferons and CD8+ T cells. Overall the authors conclude that components of both innate and adaptive immunity are required for CD47 blockade therapy in cancer.
Cell separation: EasySep™ Mouse CD8+ T Cell Isolation Kit and EasySep™ Mouse CD8ɑ Positive Selection Kit
Dendritic cells but not macrophages sense tumor mitochondrial DNA for cross-priming through Signal Regulatory Protein α signaling8
Immunity
Tumor-derived double-stranded DNA (dsDNA) can be taken up by antigen-presenting cells (APCs) to initiate DNA-sensing pathways (ex. STING), which contributes to heightened anti-tumor responses. In this study, Xu et al. showed that CD47 blockade preferentially increases cytosolic DNA sensing in DCs compared to macrophages. The ability of DCs to take up and sense cytosolic tumor-derived DNA allows them to produce type I IFN and cross-prime cytotoxic T cells, which are crucial for the therapeutic effects of CD47 blockade. This study, along with the previous, suggests that therapeutic strategies that enhance phagocytosis and tumor-derived DNA sensing will improve adaptive immune responses against tumors.
Cell separation: EasySep™ Mouse CD8+ T Cell Isolation Kit and EasySep™ Mouse CD11c Positive Selection Kit II
Oncolytic adenoviral delivery of an EGFR-targeting T cell engager improves antitumor efficacy9
Cancer Research
Bispecific T cell engagers (BiTE) are two single chain antibodies (scFV) connected by a peptide linker. One scFV is specific for a T cell co-receptor, and the other scFV is specific for a tumor-associated antigen such as epidermal growth factor receptor (EGFR). Mechanistically, BiTEs can simultaneously bind to T cells and target cells, causing T cell activation and specific killing of target cells. In this study, Fajardo et al. developed oncolytic adenoviruses that produce EGFR-targeting BiTEs in cancer cells upon viral replication. Oncolytic adenoviruses are therapeutic agents that infect tumor cells to promote their lysis; however, their efficacy is limited by infiltrating anti-virus T cells. Coupling these viruses with BiTEs allow for these infiltrating T cells to be redirected towards tumor cells, thus enhancing the therapeutic efficacy. This study highlights the strength and novel possibilities of combinatorial therapy against cancer.
Cell separation: RosetteSep™ Human T Cell Enrichment Kit
Cellular Therapy
Effector T cells abrogate stroma-mediated chemoresistance in ovarian cancer10
Cell
Adoptive cellular therapy is now being touted by many as the future of cancer therapy. T cells that are able to enter the tumor microenvironment can promote inflammatory signals and elimination of tumor cells. However, it is still unclear how T cells interact with other components of the tumor microenvironment, and how they affect the efficacy of other forms of treatment, such as chemotherapy.
Fibroblasts are a major component of the tumor microenvironment that help to promote tumor progression. Interestingly, this study showed that fibroblasts can promote resistance to platinum-based chemotherapy drugs such as cisplatin, by reducing nuclear accumulation of platinum in tumor cells. This effect is mediated by products of cysteine metabolism in fibroblasts. Furthermore, Wang et al. showed that effector T cells inhibit cysteine metabolism in fibroblasts via production of the inflammatory cytokine IFN-γ, thereby counteracting the effect of fibroblasts on chemoresistance. As a result, T cell therapies may not only directly eliminate tumor cells, but also enhance their sensitivity to conventional chemotherapeutic agents.
Cell separation: EasySep™ Human CD8+ T Cell Isolation Kit
Although researchers are simultaneously working on all of the above strategies for cancer therapy, cellular therapy using engineered immune cells have gained increasing popularity due to recent successes of chimeric antigen receptor (CAR) T cells.
Learn more about CAR T cell therapy research:
Brief Review of CAR T Cell Research
A mini-review of our selection of key advancements in CAR T cell therapy research.
Production of CAR T Cells Wallchart
Free Nature Protocols Wallchart summarizing the processes involved in producing CAR T cells for therapy.
Personalized Medicine
Due to the heterogeneity of cancer, one promising approach for cancer treatment is personalized medicine, where genetic and phenotypic profiling of tumor cells helps to determine the best course of therapeutic intervention for each individual.11
Tumorigenicity and genetic profiling of circulating tumor cells in small-cell lung cancer12
Nature Medicine
In this study, Hodgkinson et al. profiled circulating tumor cells (CTCs) in small-cell lung cancer (SCLC), an aggressive form of cancer with a five-year survival rate of only 5%. CTCs are prevalent in SCLC, and Hodgkinson et al. demonstrated that isolated CTCs from chemotherapy-naïve SCLC patients are tumorigenic in immunocompromised mice. Furthermore, the response of these CTC-derived explants to different forms of treatment mirror the donor’s response, indicating that the molecular analysis of CTCs could provide a useful tool in facilitating the delivery of personalized medicine for SCLC.
Cell separation: RosetteSep™ CTC Enrichment Cocktail
From the Authors
“The RosetteSep™ CTC Isolation Kit is an instrumental part of developing our CTC-derived explants mouse models. Its rapid and simple protocol is essential to the success of our work."
Kristopher Frese, Postdoctoral Fellow, Caroline Dive Lab, University of Manchester
To help immunologists like Kristopher isolate highly purified immune cells faster and easier, we've been developing innovative cell isolation technologies for over 20 years.
Resources for Your Cancer Immunology Research
References
- Vinay DS et al. (2015) Immune evasion in cancer: mechanistic basis and therapeutic strategies. Semin Cancer Biol 35 Suppl: S185-98.
- Halim L et al. (2017) An atlas of human regulatory T helper-like cells reveals features of Th2-like Tregs that support a tumorigenic environment. Cell Rep 20(3): 757-770.
- Li R et al. (2016) Macrophage-secreted TNFα and TGFβ1 influence migration speed and persistence of cancer cells in 3D tissue culture via independent pathways. Cancer Res 77(2): 279-290.
- Beug ST et al. (2014) Smac mimetics and innate immune stimuli synergize to promote tumor death. Nat Biotechnol 32(2): 182-190.
- Beug ST et al. (2017) Smac mimetics synergize with immune checkpoint inhibitors to promote tumour immunity against glioblastoma. Nat Commun 8 doi:10.1038/ncomms14278.
- Liu X et al. (2015) CD47 blockade triggers T cell-mediated destruction of immunogenic tumors. Nat Med 21(10): 1209-1215.
- Scott AM et al. (2012). Monoclonal antibodies in cancer therapy. Cancer Immun 12: 14.
- Xu MM et al. (2017) Dendritic cells but not macrophages sense tumor mitochondrial DNA for cross-priming through Signal Regulatory Protein α signaling. Immunity 7(2): 363-373.
- Fajardo CA et al. (2017) Oncolytic adenoviral delivery of an EGFR-targeting T-cell engager improves antitumor efficacy. Cancer Res 77(8): 2052-2063.
- Wang W et al. (2016). Effector T cells abrogate stroma-mediated chemoresistance in ovarian cancer. Cell 165(5): 1092-1105.
- Hanash SM et al. (2011) Emerging molecular biomarkers--blood-based strategies to detect and monitor cancer. Nat Rev Clin Oncol 8(3): 142-150.
- Hodgkinson CL et al. (2014) Tumorigenicity and genetic profiling of circulating tumor cells in small-cell lung cancer. Nat Med 20(8): 897-903.