The Role of Memory B Cells in Infectious Diseases
Immunology Feature
B cells have an important and complex role in the immune response to infectious diseases, which poses a challenge to understanding infection risks and developing vaccines.
Following an infection or vaccination, memory B cells and high-affinity antibody-secreting plasma cells are produced to confer immunological memory. Interestingly, memory B cell responses can differ across different memory B cell subpopulations and the persistence of plasma cells varies greatly for reasons that are not well understood. Consequently, some pathogens and vaccines do not elicit lasting protective immunity.1
The gaps in our understanding of the complex role of memory B cells in infectious diseases necessitate ongoing research in this field. For example, studying memory B cell biology and mobilization to sites of infection can help in the development of more efficacious vaccines. More recently, memory B cells have been the focus of therapeutic development as a new tool for clinical intervention. Below, we will focus on the complexity of memory B cells and their role in infectious diseases, and will highlight recent advancements in the field.
Understanding B Cell Responses in Chronic Infections
Vaccines typically function by providing the host with one or more antigens specific to the targeted pathogen. Antigen-presenting cells, such as dendritic cells or macrophages, will then process and present the antigen(s) on MHC class I or class II molecules, which subsequently leads to T cell activation. Activated helper T cells provide co-stimulatory signals to mature and primed B cells, which will induce the proliferation and differentiation of B cells into antibody-producing plasma cells and into memory B cells for long-lasting immunity.
While the above outlines the typical immune response to vaccinations, not all infectious diseases elicit the same B cell responses. This can make developing effective vaccines challenging. The following papers describe investigations into a couple of these cases.
Migrant memory B cells secrete luminal antibody in the vagina2
Nature
Serum antibody levels are often an indication of the protection conferred by a prophylactic vaccine, but this may not always be the case for viruses that enter at mucosal surfaces.3 In this paper, the Iwasaki lab studied HSV-2 infection in mice to determine the extent to which antibodies were delivered to type II mucosa in the lower female reproductive tract lumen. The study shows that high levels of antiviral antibodies in sera do not correspond to the presence of antibodies in the vaginal lumen, as circulating antibodies fail to enter the female reproductive tract lumen. Instead, the researchers found that circulating memory B cells are recruited to the female reproductive tract upon secondary challenge with HSV-2 in a CXCR3-dependent manner. Here, memory B cells secreted antibodies locally to help protect mice from HSV-2 infection.
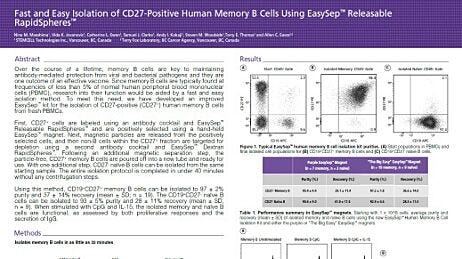
Cell Separation: EasySep™ Mouse B Cell Isolation Kit
Malaria-induced interferon-γ drives the expansion of Tbethi atypical memory B cells4
PLoS Pathogens
Several chronic infectious diseases, including HIV and malaria, are associated with an expansion of CD21−CD27− “atypical” memory B cells. These atypical memory B cells display reduced B cell receptor signaling and decreased effector functions, and may present challenges when developing effective vaccines for chronic infections. In this paper, Obeng-Adjei et al. found that atypical B cells express high levels of T-bet, a transcription factor key in influencing the fate of other immune cell types. The authors found that children who frequently got malaria had more T-bethi B cells than children who rarely got malaria, and that naïve B cells expressed T-bet more efficiently in the presence of Th1-polarized Tfh (Tfh-1) cells. The paper offers further insights on the mechanisms involved in atypical memory B cell differentiation and the importance of this on improving antibody responses following vaccination.
Cell Separation: EasySep™ Human B Cell Enrichment Kit
Studying Antibody Production When Preparing for Future Pandemics
Specific memory B cell response in humans upon infection with highly pathogenic H7N7 avian influenza virus5
Scientific Reports
H7 avian influenza viruses cause yearly worldwide outbreaks among poultry populations. These viral outbreaks have also led to severe cases in humans, and as such, have the potential to become future pandemic threats. Studying the memory B cell response to avian influenza virus infection in humans could provide the key to understanding human infection risks and how to retrospectively detect the exposure of human populations to the H7N7 influenza virus. In this study, Westerhuis et al. determined the frequency and specificity of memory B cells by characterizing antibodies produced following infection with influenza H7N7 in humans, using peripheral blood from cases and controls as sample sources. This was done by stimulating oligoclonal B cell cultures in vitro and detecting subtype-specific antibodies in the culture supernatants. The study serologically confirmed the H7 avian influenza virus infections and the results suggest that H7 infection prompts the production of strain-specific and cross-reactive antibodies.
Cell Separation: EasySep™ Human CD19 Positive Selection Kit II
Investigating Neutralizing Antibodies to Better Understand Viral Infections
Neutralizing antibodies (NAbs) are antibodies that bind to pathogens and neutralize their biological activity, preventing the pathogen from infecting cells. Successful vaccines need to trigger the production of NAbs, which are produced by B cells upon exposure to an antigen. The following publications show how studying NAbs can help with responding to emerging infectious diseases, such as SARS-CoV-2, and provide insights into controlling chronic viral infections.
Potent neutralizing antibodies against SARS-CoV-2 identified by high-throughput single-cell sequencing of convalescent patients’ B cells6
Cell
In this study, Cao et al. performed high-throughput single-cell RNA and VDJ sequencing of antigen-enriched B cells from 60 convalescent patients to identify neutralizing antibodies against SARS-CoV-2. The researchers identified fourteen potent SARS-CoV-2 NAbs and reported that the most potent antibody, BD-368-2, displayed strong therapeutic and prophylactic efficacy in infected mice. Overall, the study showcases that high-throughput single B-cell sequencing can be used to efficiently identify human NAbs, which can prove useful in responding to emerging infectious diseases.
Cell Separation: EasySep™ Human Memory B Cell Isolation Kit, EasySep™ Human B Cell Enrichment Kit
Quantitative and qualitative analysis of humoral immunity reveals continued and personalized evolution in chronic viral infection7
Cell Reports
Neutralizing antibodies are produced during chronic lymphocytic choriomeningitis virus (LCMV) infection, but it is unclear why neutralizing antibodies are largely absent in acute infection and how the antibody response evolves during ongoing infection. In this study, Kräutler et al. investigate how the humoral immune response evolves during acute and chronic LCMV infection using quantitative functional assays and time-resolved antibody repertoire sequencing. The findings uncover sustained germinal center reactions and plasma cell differentiation during chronic infection. The study also suggests a correlation between maintained B cell diversity and the development of infection-resolving antibodies.
Cell Separation: EasySep™ Mouse Pan-B Cell Isolation Kit
Explore More Immunology Features.
Vaccine Development for COVID-19 and Infectious Diseases
Learn about tools and assays for developing vaccines, including products for sourcing and isolating immune cells, assessing vaccine candidates, and developing production lines for recombinant vaccines.
Therapeutics Development for COVID-19 and Viral Diseases
Explore technologies, applications, and research tools for developing and assessing therapeutics to combat viral diseases such as COVID-19.
Additional B Cell Resources
References
- Wong et al. (2019) Basics of memory B‐cell responses: lessons from and for the real world. Immunology 156(2): 120–9.
- Oh et al. (2019) Migrant memory B cells secrete luminal antibody in the vagina. Nature 571(7763): 122–6.
- Iwasaki (2016) Exploiting mucosal immunity for antiviral vaccines. Annu Rev Immunol 34: 575-608.
- Obeng-Adjei et al. (2017) Malaria-induced interferon-γ drives the expansion of Tbethi atypical memory B cells. PLoS Pathog 13(9): e1006576.
- Westerhuis et al. (2020) Specific memory B cell response in humans upon infection with highly pathogenic H7N7 avian influenza virus. Sci Rep 10(1): 3152.
- Cao et al. (2020) Potent neutralizing antibodies against SARS-CoV-2 identified by high-throughput single-cell sequencing of convalescent patients’ B cells. Cell 182(1): 73–84.e16.
- Kräutler et al. (2020) Quantitative and qualitative analysis of humoral immunity reveals continued and personalized evolution in chronic viral infection. Cell Rep 30(4):997-1012.e6.