Intestinal Organoids
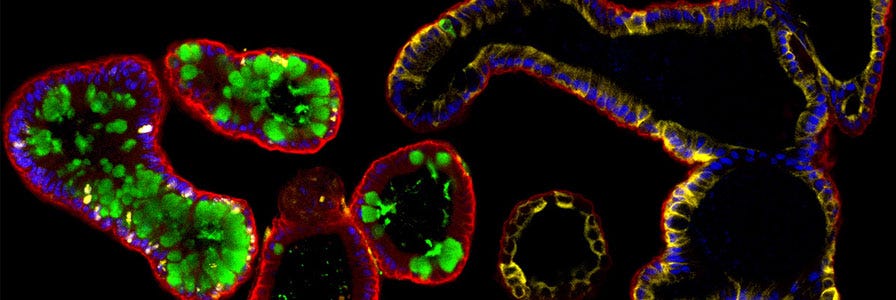
The intestinal epithelium is a rapidly renewing tissue that can undergo complete cellular replacement every four to five days. These regenerative properties make the intestinal epithelium an attractive system for researchers studying epithelial regeneration, adult stem cell biology, disease modelling and cancer biology, both within and outside the context of the intestine.
Explore the resources below for information to support your intestinal research and the growth of intestinal organoids.
E-Book: Proven Protocols for Intestinal Organoid Culture
From getting started with intestinal organoids to advanced assays, this e-book provides curated protocols for the isolation and culture of human-derived intestinal organoids. Find proven protocols for a wide variety of well-established assays from immunocytochemical staining, toxicity testing, genetic manipulation, and monolayer differentiation.
Download Now >Mini-Review: Intestinal Organoids
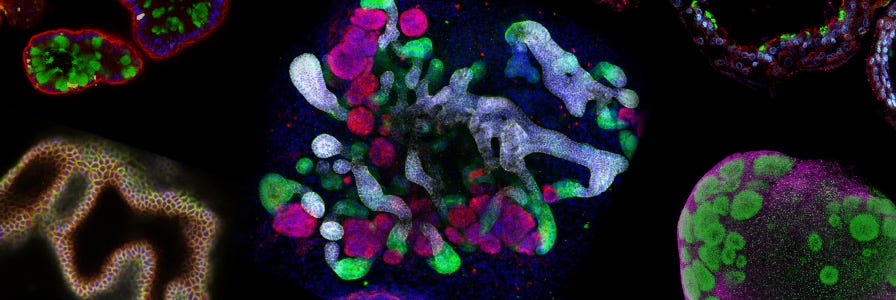
Learn more about intestinal organoids and how they are being used to model intestinal development, health, disease across a wide range of research areas.
E-book: Organoid Evolution and Applications
Download a copy of the Wiley e-book "Organoid Research Techniques: Evolution and Applications" to learn more about the development of these novel model systems.
Mini-Review: Organoids in Drug Discovery
Learn about the predictive power of organoids in drug discovery with collated research, data, and related educational resources.
Organoids: Nature Research Round Table
Global organoid experts discuss the current state of organoid technology, protocol standardization, clinical translation, and what questions a given organoid culture can and can’t answer.