PneumaCult™ Alveolar Organoid Media
Cell culture media for expansion and differentiation of human alveolar organoids
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration
-
 Animal Component-Free Cell Dissociation Kit
Animal Component-Free Cell Dissociation KitDissociation kit for human stem and progenitor cells
-
 CryoStor® CS10
CryoStor® CS10Animal component-free, defined cryopreservation medium with 10% DMSO
-
 Heparin Solution
Heparin SolutionCell culture supplement
-
 Flat-Bottom Plates, Non-Treated
Flat-Bottom Plates, Non-TreatedSterile flat-bottom, non-treated multiwell plate with lid; 6-, 12-, 24-, 48-, or 96-well formats
-
Labeling Antibodies
Compatible antibodies for purity assessment of isolated cells
Overview
Use PneumaCult™ Alveolar Organoid Expansion (AvOE) Medium to achieve a 10,000X cumulative-fold expansion of ATII cells as organoids within 10 passages; this high yield reduces the need to regularly source donor tissue. You can flexibly pause your cultures at any passage by cryopreserving organoids cultured in PneumaCult™ AvOE Medium as single cells. The medium’s three-component, serum-free format maximizes experimental reproducibility, and the alveolar organoids maintain properties indicative of the ATII cell phenotype, including the ability for self-renewal, expected marker expression (e.g. HT2-280 and SP-C), and lineage potential for differentiation to ATI cells. PneumaCult™ AvOE Medium can also be used to generate alveolar monolayers to facilitate air-liquid interface (ALI) culture as outlined in this protocol.
Use PneumaCult™ Alveolar Organoid Differentiation (AvOD) Medium to differentiate ATII cells expanded as organoids in PneumaCult™ AvOE Medium. The two-component PneumaCult™ AvOD Medium enables in-dome differentiation of ATII organoid cultures to ATI cells in as few as 10 days, with over 85% purity. Differentiated cells show decreased ATII marker expression and strong up-regulation of ATI cell markers (RAGE/AGER, HT1-56, and GPRC5a).
PneumaCult™ AvOE and AvOD Media are compatible with primary isolated fresh or cryopreserved ATII cells, as well as high-quality commercially available alveolar epithelial cell sources.
Learn more about lung organoid cultures in our learning center.
More Information
| Safety Statement | CA WARNING: This product can expose you to Progesterone which is known to the State of California to cause cancer. For more information go to www.P65Warnings.ca.gov |
|---|
Data Figures
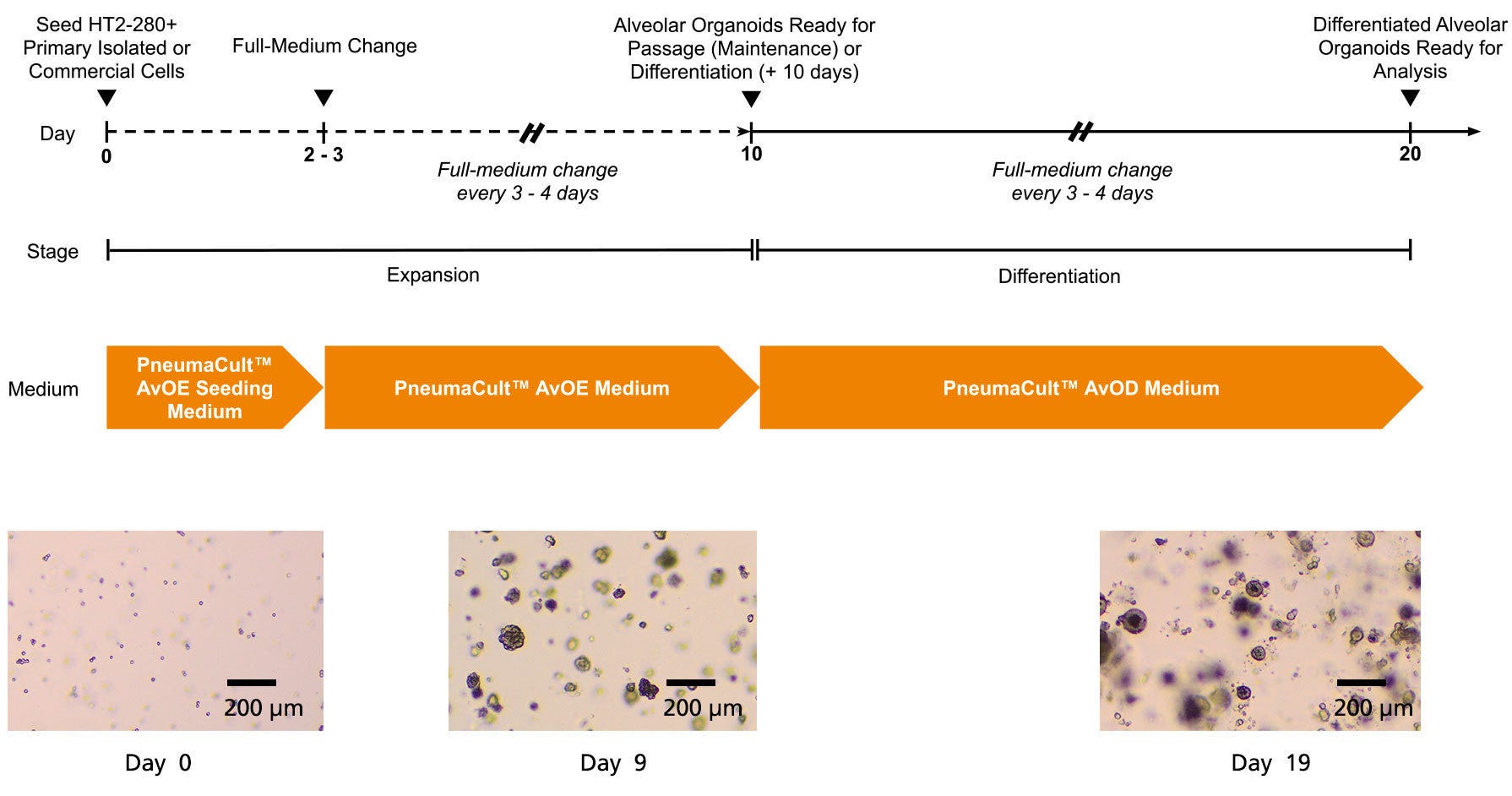
Figure 1. Generation of Alveolar Organoids Using PneumaCult™ Alveolar Organoid Media
The PneumaCult™ Alveolar Organoid Media workflow is a two-stage protocol. During the expansion stage, primary isolated or cryopreserved human ATII single cells are seeded in PneumaCult™ Alveolar Organoid Expansion (AvOE) Seeding Medium. On day 2 - 3, after a full-media change, cultures are expanded using PneumaCult™ AvOE Medium to obtain mature ATII organoids. In the differentiation stage, ATII organoids are cultured for 10 additional days using PneumaCult™ Alveolar Organoid Differentiation (AvOD) Medium to generate ATI organoids.
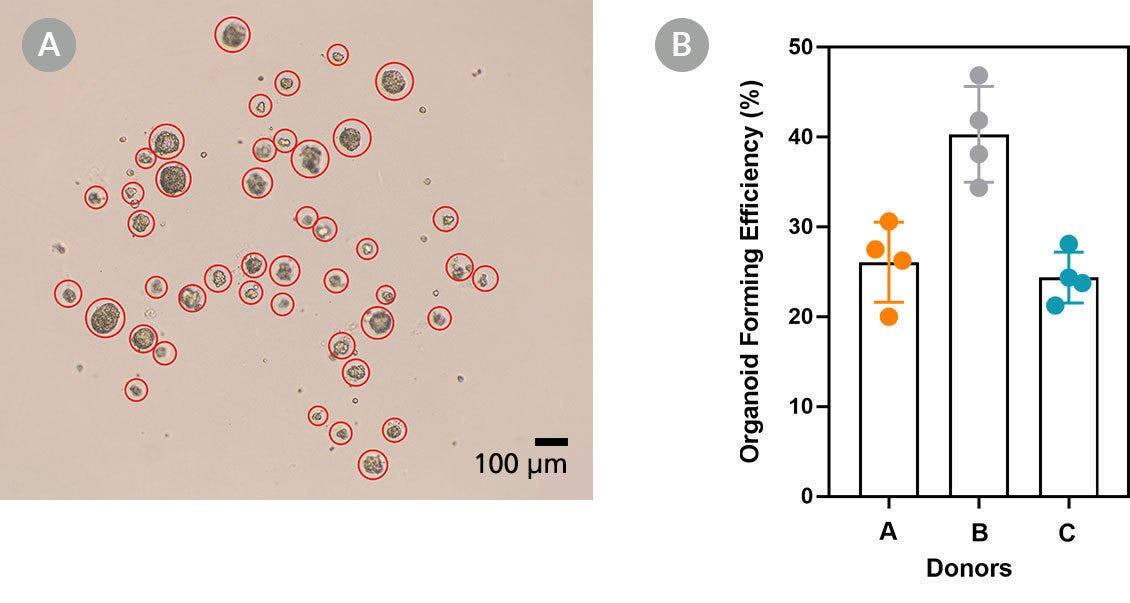
Figure 2. PneumaCult™ Alveolar Organoid Expansion Medium Supports High Organoid Forming Efficiency
Organoid forming efficiency (OFE) serves as a functional measure of the health of an organoid culture and the performance of the media. ATII organoids were expanded in PneumaCult™ AvOE Medium (160 cells seeded per dome). (A) Brightfield image taken at day 9 using a 4X objective. The entire dome was captured in the image, each organoid formed was counted, and indicated (total 44; OFE=27.5%). (B) Bar graph showing OFE of ATII organoid cultures derived from three different donors (means ± SD). OFE = (Organoids Formed/Cells Seeded) x 100%.
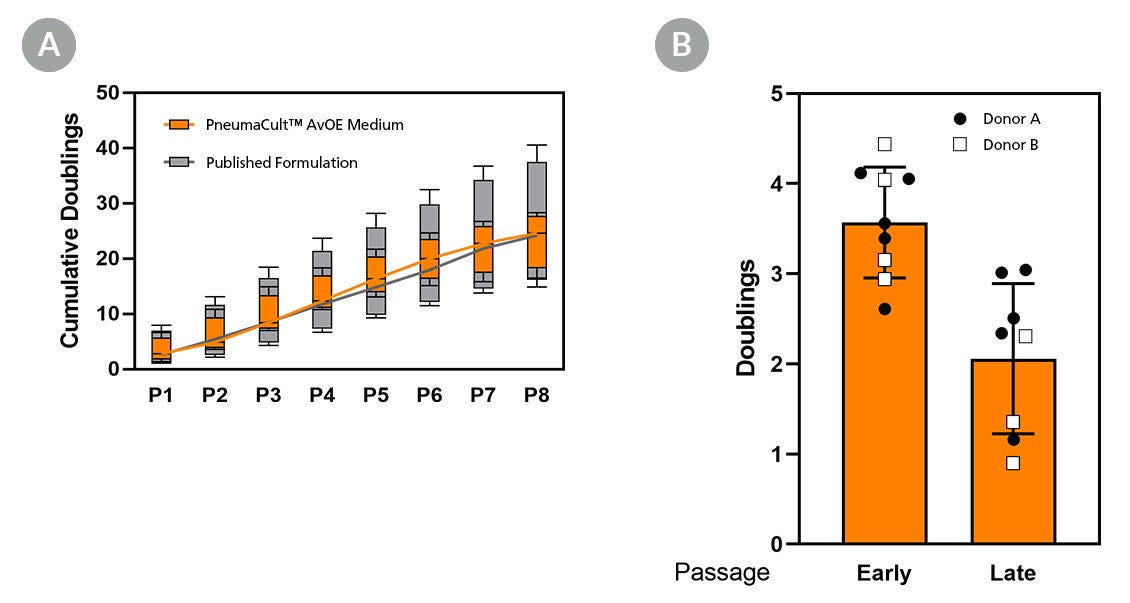
Figure 3. PneumaCult™ AvOE Medium Supports Long-Term Expansion and Cryopreservation For Biobanking
(A) Box and whiskers plot of the cumulative doublings at each passage (n=4 independent donors) for organoids expanded in PneumaCult™ AvOE Medium (orange) and published formulation (gray). The means are connected by a line. Long-term passaging of alveolar organoids and over 10000x fold expansion was achieved by both PneumaCult™ AvOE Medium and published formulation. (B) Bar graph showing population doublings (mean ± SD) of organoids generated from cells previously cryopreserved at early and late passages, and expanded in PneumaCult™ AvOE Medium. The cultured organoids retained their expansion potential after thawing.
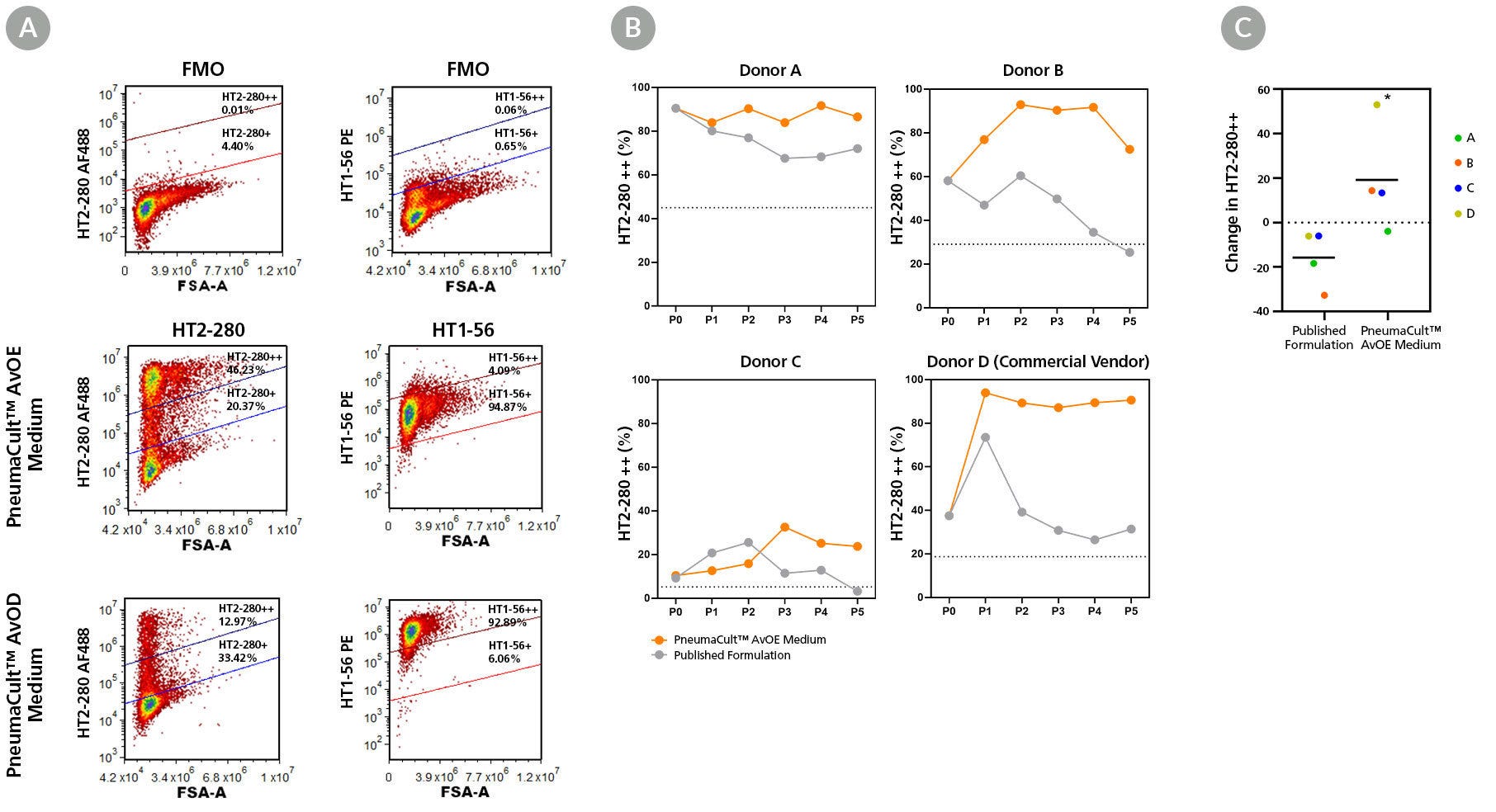
Figure 4. PneumaCult™ Alveolar Organoid Media Maintains High Levels of ATII and ATI Cell Marker Expression
(A) Flow cytometric gating of fluorescence minus one (FMO) controls for HT2-280 and HT1-56 markers (top), stained cells from organoids expanded in PneumaCult™ AvOE Medium (middle), and subsequently differentiated in PneumaCult™ AvOD Medium (bottom). PneumaCult™ AvOE Medium maintains both the ATII marker expression (HT2-280) and lineage potential to differentiate into ATI cells (HT1-56) in PneumaCult™ AvOD Medium. (B) Organoids derived from 4 independent donors, expressing high levels of HT2-280 were cultured in PneumaCult™ AvOE Medium or published formulation, and compared at each passage. Dashed line depicts 50% of starting HT2-280++%. (C) Scatter plot illustrating the percent change in HT2-280 expression between P0 and P5 from the same donors. Organoids cultured in PneumaCult™ AvOE Medium maintained higher levels of HT2-280 marker expression, compared to those cultured using published formulation. Mean is displayed as a solid line. * = p<0.05 in a one-tailed paired T-test (n=4).
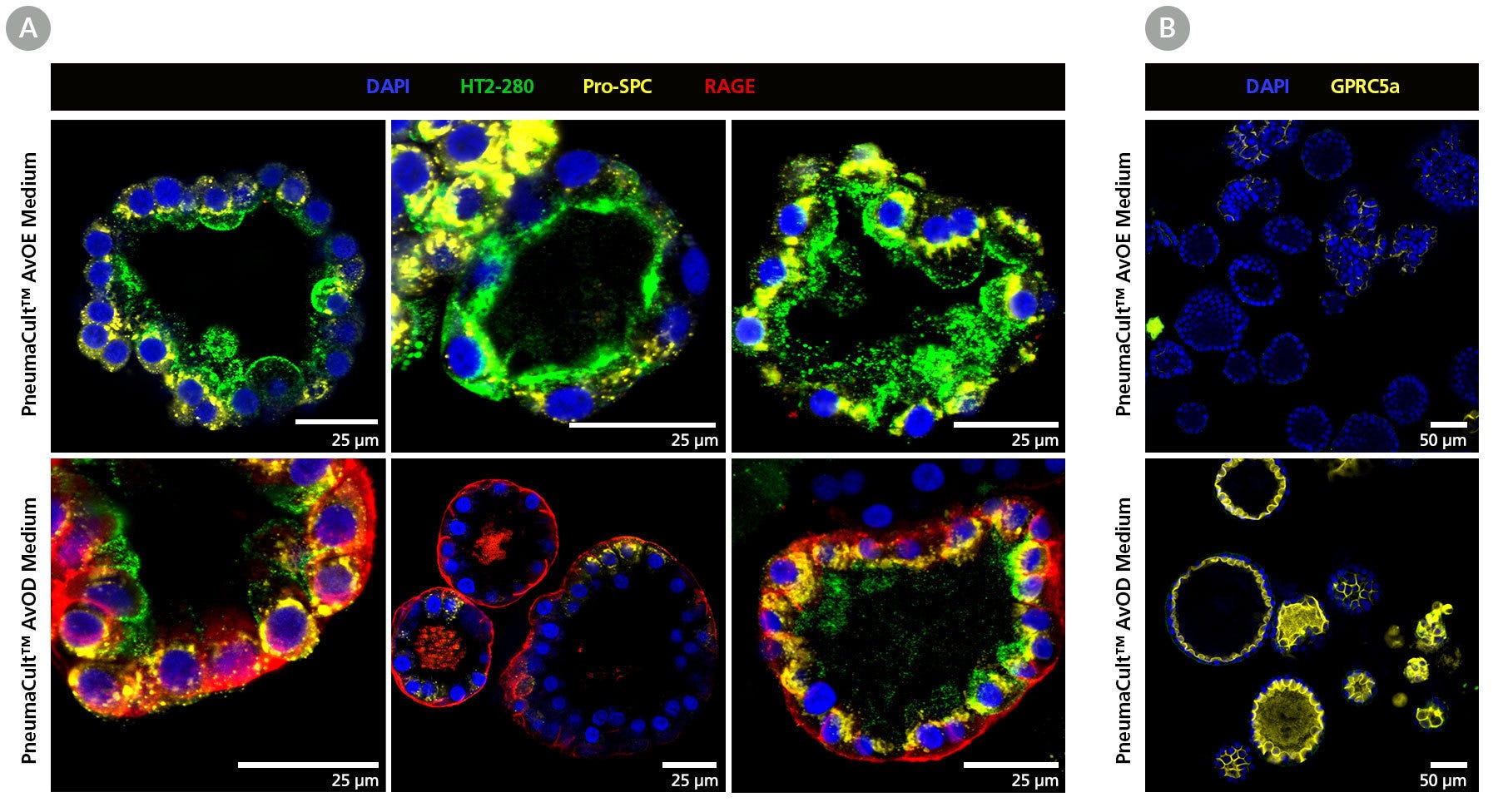
Figure 5. Immunohistochemistry Confirms Expression of ATII and ATI Cell Markers in Alveolar Organoids
(A) Organoids from three donors expanded in PneumaCult™ AvOE Medium (top) express the ATII markers, HT2-280 (green) and pro-surfactant protein C (pro-SPC, yellow). When further differentiated in PneumaCult™ AvOD Medium (bottom), the levels of these ATII markers are down-regulated, while the ATI marker RAGE (red) is up-regulated. (B) Differentiated organoids also express high levels of ATI marker GPRC5a (yellow).
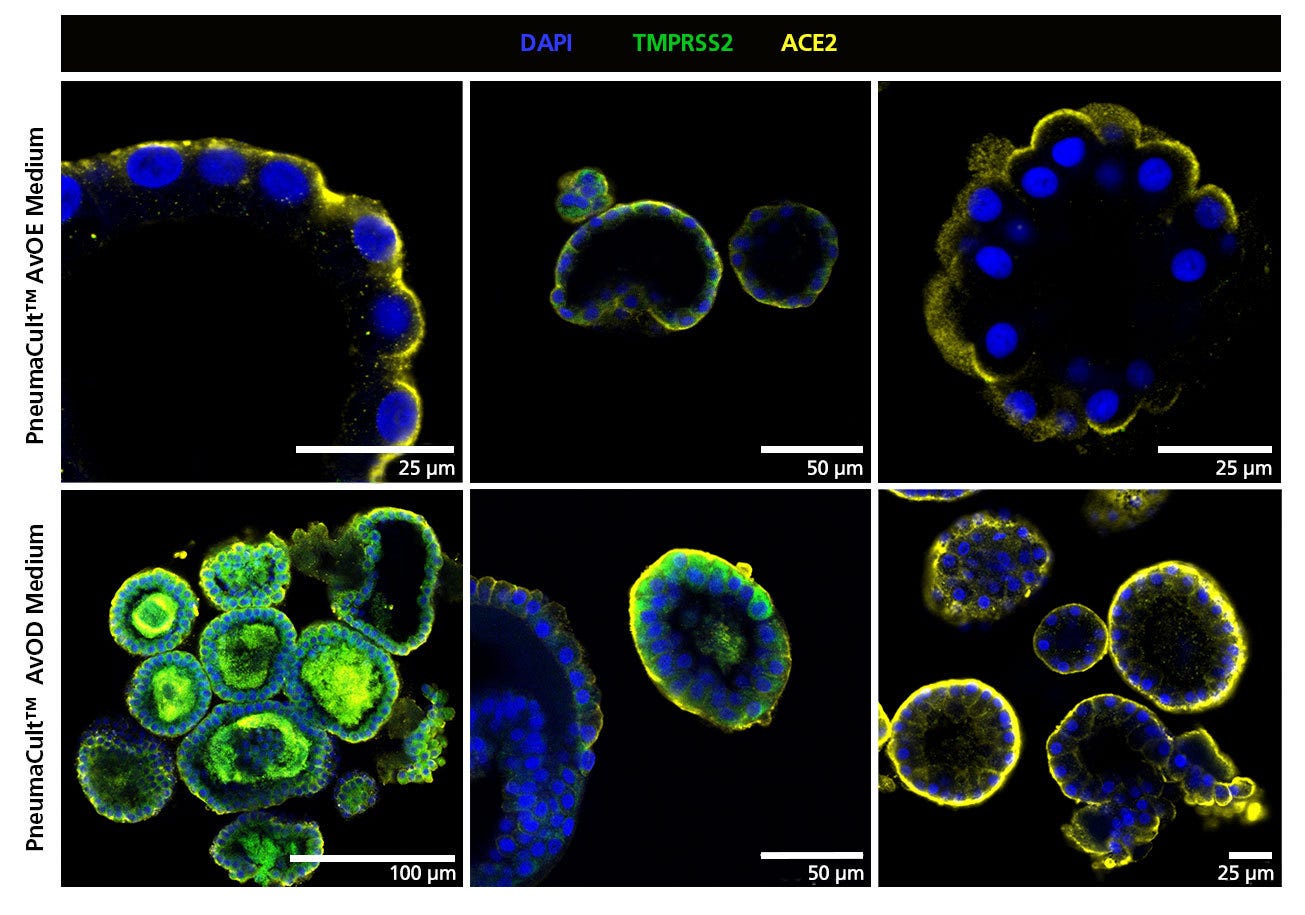
Figure 6. Organoids Cultured in PneumaCult™ Alveolar Organoid Media Express TMPRSS2 and ACE2, Key Markers for SARS-CoV-2 Entry
Organoids from three donors expanded in PneumaCult™ AvOE Medium (top) and differentiated in PneumaCult™ AvOD Medium (bottom) express proteins associated with SARS-CoV-2 entry, TMPRSS2 and ACE2. While ACE2 was expressed in all donors and conditions, TMRPSS2 expression was donor-dependent.
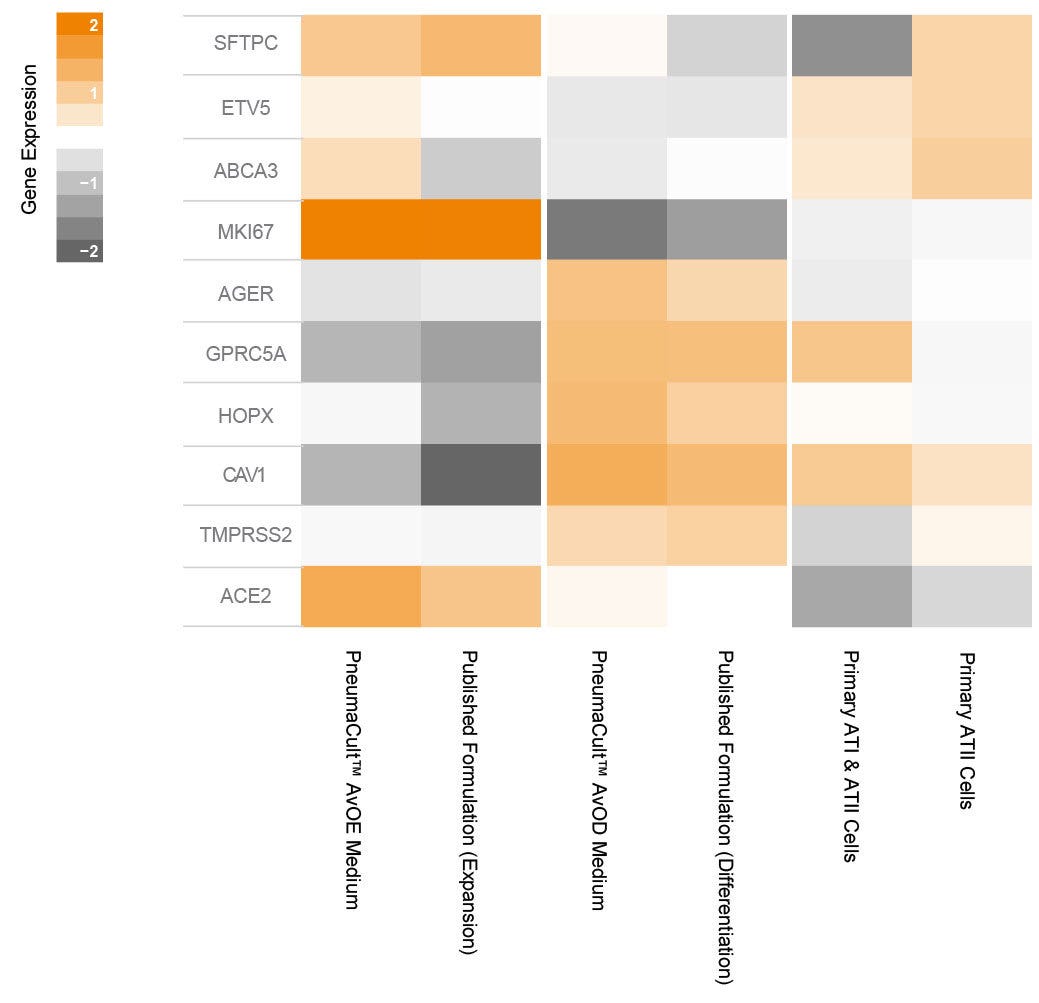
Figure 7. Gene Expression Analysis of Organoids Cultured in PneumaCult™ Alveolar Organoid Media Shows Presence of Alveolar Markers
Normalized gene expression heatmap of organoids expanded in PneumaCult™ AvOE Medium and differentiated in PneumaCult™ AvOD Medium compared to those grown using a published formulation. The presence of canonical ATII and ATI genes as well as SARS-CoV-2 key entry markers, ACE2 and TMPRSS2, are shown. Published RNA-Seq gene expression datasets of primary alveolar epithelial cells (including ATI and ATII cells, from Mikami et al. 2021, GSE186359) and primary ATII cells (Jacob and Kotton, 2017, GSE96642) were used as controls.
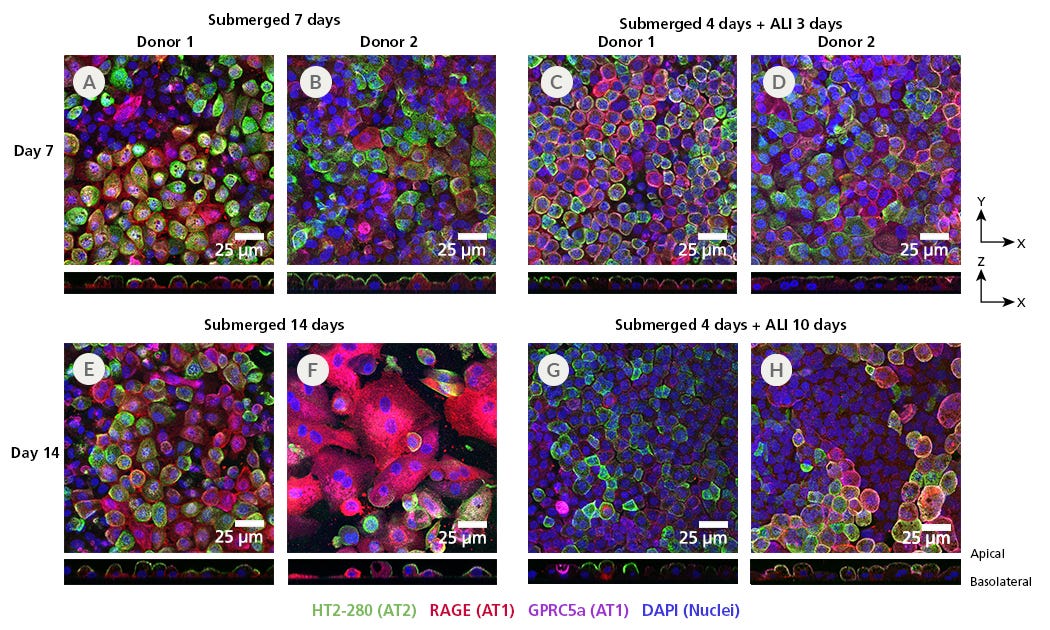
Figure 8. ATII Cells Show Optimal Marker Expression When Cultured for 7 Days As Submerged or ALI Cultures
The generation of alveolar type 2 (ATII) cultures at the air-liquid interface (ALI) on cell culture inserts is a two-stage protocol. In the first stage, ATII cells from two separate donors were seeded in PneumaCult™ Alveolar Organoid Expansion (AvOE) Medium as organoid cultures for 10 days. Single cells were then harvested and seeded into ECM-coated inserts and cultured to Day 7 or Day 14. Cultures were placed at ALI on day 4 and cultured for an additional (C, D) 3 or (G, H) 10 days. Cells were fixed on the membrane in 4% PFA and stained for the ATII-specific marker HT2-280 (green), as well as the ATI-specific markers RAGE (red) and GPRC5a (magenta) and counterstained with DAPI (blue). In both donors, cultures in (A, B) submerged and (C, D) ALI for 7 days express HT2-280 with some RAGE, and little GPRC5a. Additional growth for 7 days in (E, F) submerged cultures resulted in a decrease in HT2-280 and an increase in RAGE, indicating differentiation into ATI cells. (G, H) Growth for an additional 7 days at the ALI resulted in some decrease in HT2-280, but expression was more stable than in submerged culture conditions. This data suggests that the ideal culture conditions for ATII cells on cell culture inserts are for (C, D) 7 days, with 3 days at ALI; however these cultures can still be maintained for up to 14 days (10 days of ALI) with minimal loss of ATII markers. Note scale is the same for XY and ZX figures.
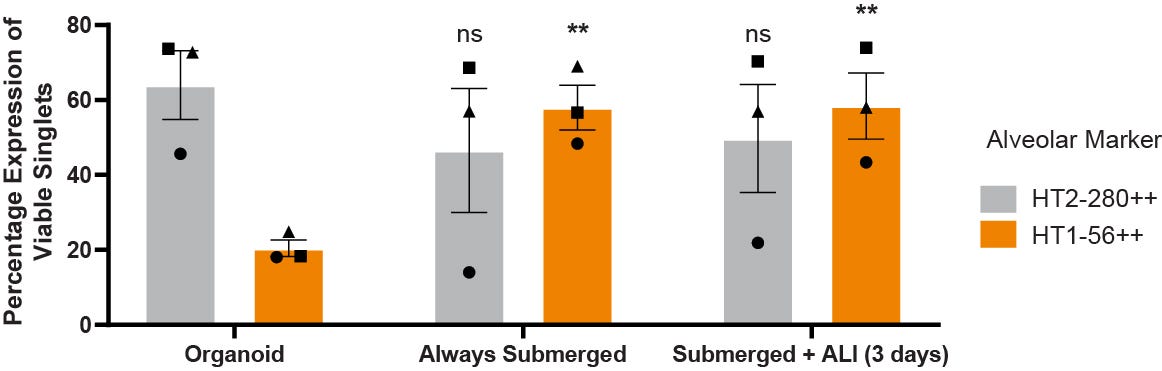
Figure 9. Alveolar Cells Grown on Cell Culture Inserts Maintain Expression of HT2-280 and Increase Expression of HT1-56
ATII cells from three separate donors were seeded from organoid domes onto ECM-coated cell culture inserts and cultured to Day 7. Expression of the ATII marker HT2-280 and the ATI marker HT1-56 was determined as the percentage of viable singlets by flow cytometry. The data demonstrates HT2-280 high positive expression decreases slightly but not significantly, while HT1-56 high positive expression increases significantly (ANOVA with Dunnett’s post hoc test, where ** = p < 0.01; n = 3) when alveolar cells are cultured on cell culture inserts compared to organoid cultures. There were no differences between ALI (3 days) and always submerged cultures in either HT2-280 or HT1-56 expression. Symbols represent different donors across the conditions.
Protocols and Documentation
Find supporting information and directions for use in the Product Information Sheet or explore additional protocols below.
Applications
This product is designed for use in the following research area(s) as part of the highlighted workflow stage(s). Explore these workflows to learn more about the other products we offer to support each research area.
Resources and Publications
Educational Materials (16)
Related Products
-
 PneumaCult™-ALI Medium
PneumaCult™-ALI MediumSerum- and BPE-free medium for human airway epithelial cells cultured at the air-liquid interface
-
 PneumaCult™-Ex Plus Medium
PneumaCult™-Ex Plus MediumSerum- and BPE-free medium for expansion of primary human airway epithelial cells
-
 PneumaCult™ Airway Organoid Kit
PneumaCult™ Airway Organoid KitSerum- and BPE-free medium for efficient establishment and differentiation of airway organoids
-
 STEMdiff™ Branching Lung Organoid Kit
STEMdiff™ Branching Lung Organoid KitSerum-free media for generation and maturation of branching lung organoids from human ES and iPS cells
Item added to your cart

PneumaCult™ Alveolar Organoid Media
PRODUCTS ARE FOR RESEARCH USE ONLY AND NOT INTENDED FOR HUMAN OR ANIMAL DIAGNOSTIC OR THERAPEUTIC USES UNLESS OTHERWISE STATED. FOR ADDITIONAL INFORMATION ON QUALITY AT STEMCELL, REFER TO WWW.STEMCELL.COM/COMPLIANCE.
CA WARNING: This product can expose you to Progesterone which is known to the State of California to cause cancer. For more information go to www.P65Warnings.ca.gov
















