ThawSTAR® CFT2 Automated Thawing System
Automated cell thawing system for consistent thawing performance
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration
Overview
During vial transport, protect your cells from transient warming events and ensure maximum cell health by using the ThawSTAR® CFT2 Transporter. It provides a portable solution for the handling and transport of frozen vials, from long-term storage in vapor phase liquid nitrogen or in a -80°C freezer, to downstream processing with the ThawSTAR® CFT2 Automated Thawing System.
ThawSTAR® CFT2 Automated Thawing System is compatible with a variety of 1.8 - 2.0 mL vials.
Note: For performance qualification documentation and accessories required to facilitate functional testing, use the ThawSTAR® CFT2 IOPQ Kit (available for separate purchase). Additional ThawSTAR® CFT2 Confirmation Vials to evaluate the performance of the instrument are also available for purchase.
Data Figures
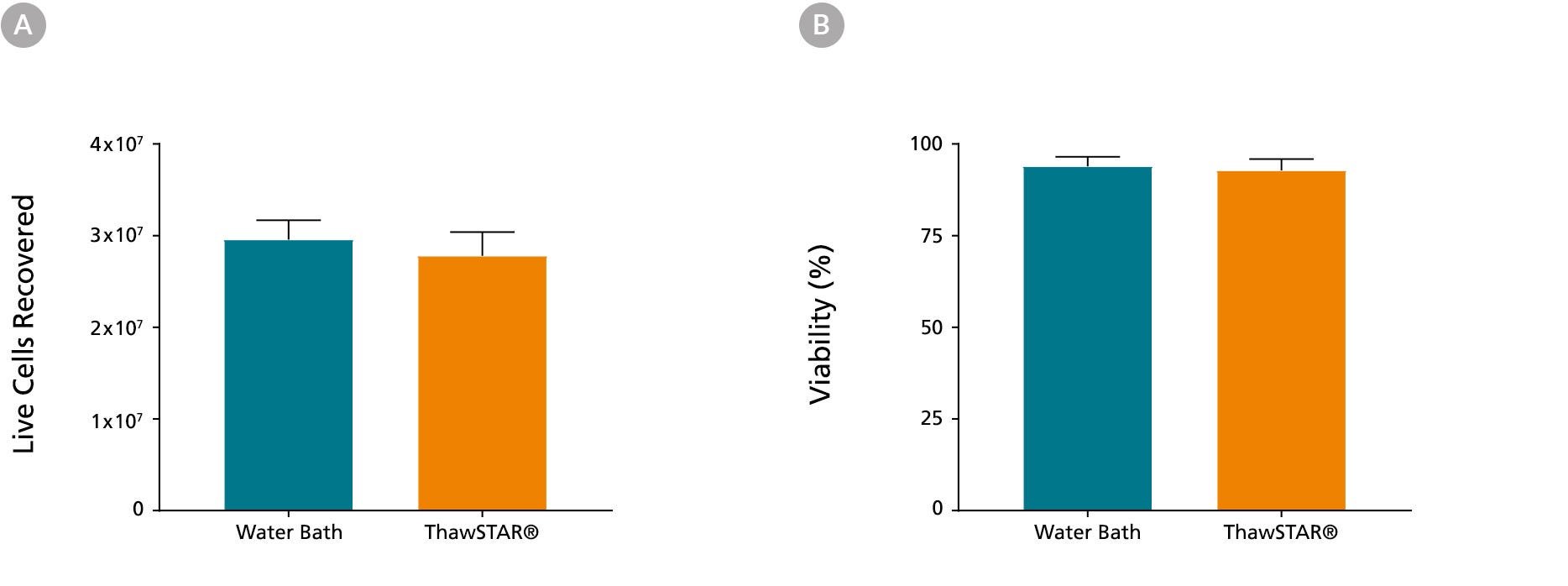
Figure 1. Frozen PBMCs Thawed Using the ThawSTAR® CFT2 Automated Thawing System Show High Recovery and Viability
Peripheral blood mononuclear cells (PBMCs) cryopreserved in CryoStor® CS10 (Catalog #07930) at a concentration of 3 x 107 cells/vial were retrieved from liquid nitrogen, one week after storage. When thawed using the ThawSTAR® CFT2 Automated Thawing System or a water bath, (A) the mean live cell recovery was 2.78 x 107 vs. 2.96 x 107 cells, respectively and (B) the mean viability was 92.8% vs. 93.9%, respectively. The cells were from 3 different donors and were tested in triplicates. Cell recovery and viability was assessed using a Nucleoview counter.
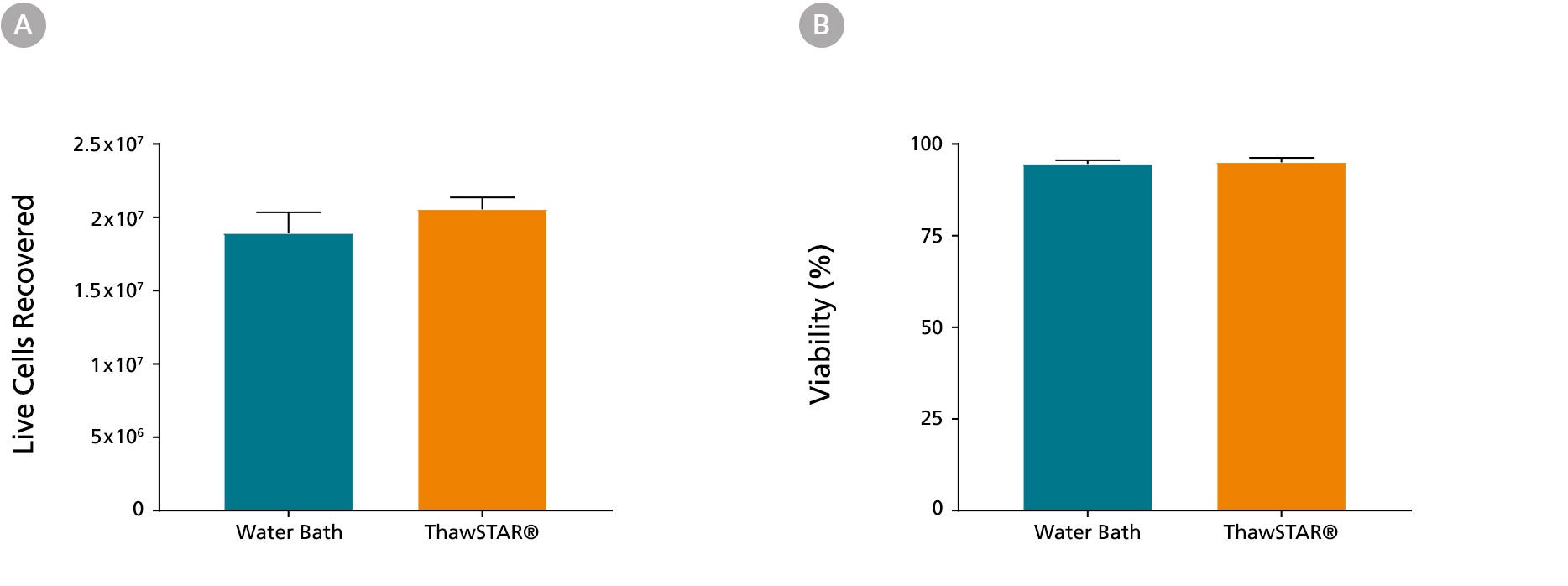
Figure 2. Frozen Monocytes Thawed Using the ThawSTAR® CFT2 Automated Thawing System Show High Recovery and Viability
Monocytes cryopreserved in CryoStor® CS10 (Catalog #07930) at a concentration of 2 x 107 cells/vial were retrieved from liquid nitrogen, one week after storage. When thawed using the ThawSTAR® CFT2 Automated Thawing System or a water bath, (A) the mean live cell recovery was 2.05 x 107 vs. 1.89 x 107 cells, respectively and (B) the mean viability was 95% vs. 94.6%, respectively. The cells were from 3 different donors and were tested in triplicates. Cell recovery and viability was assessed using a Nucleoview counter.
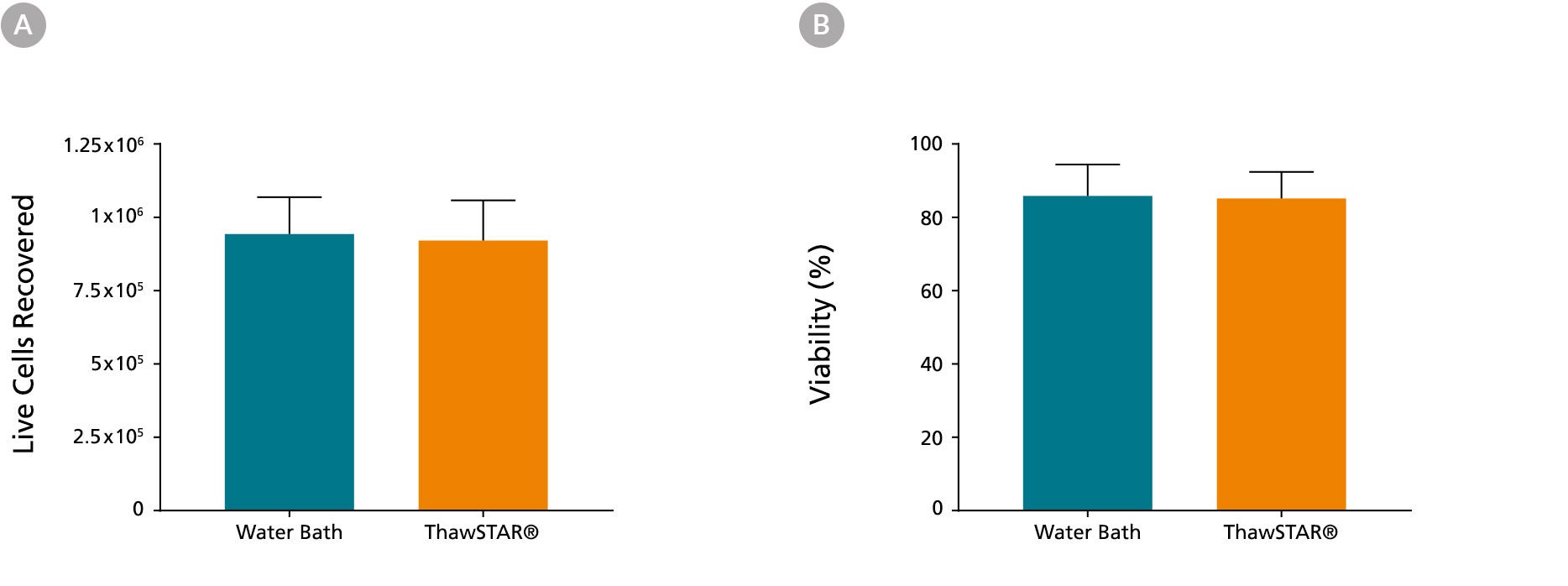
Figure 3. Frozen hPSCs Thawed Using the ThawSTAR® CFT2 Automated Thawing System Show High Recovery and Viability
Human pluripotent stem cells (hPSCs) cryopreserved in CryoStor® CS10 (Catalog #07930) at a concentration of 1 x 106 cells/vial were retrieved from liquid nitrogen, one week after storage. When thawed using the ThawSTAR® CFT2 Automated Thawing System or a water bath, (A) the mean live cell recovery was 9.05 x 105 vs. 9.35 x 105 cells, respectively and (B) the mean viability was 83.04% vs. 82.93%, respectively. The hPSCs were from 3 different cell lines (M001, 1C, and H9) and were tested in triplicates. Cell recovery and viability was assessed using a Nucleoview counter.
Protocols and Documentation
Find supporting information and directions for use in the Product Information Sheet or explore additional protocols below.
Applications
This product is designed for use in the following research area(s) as part of the highlighted workflow stage(s). Explore these workflows to learn more about the other products we offer to support each research area.
Resources and Publications
Educational Materials (9)
Related Products
-
 ThawSTAR® CFT2 Transporter
ThawSTAR® CFT2 TransporterPortable solution for the handling and transport of frozen vials from liquid nitrogen or -80°C freezer
-
 ThawSTAR® CFT2 Confirmation Vials
ThawSTAR® CFT2 Confirmation VialsFor use before and after each thaw session to document instrument performance and audit trail
-
 ThawSTAR® CFT2 IOPQ Kit
ThawSTAR® CFT2 IOPQ KitInstallation, operational, and performance qualification documentation and accessories to facilitate functional testing of ThawSTAR® CFT2
-
 Corning® Cryogenic Vials with Orange Caps
Corning® Cryogenic Vials with Orange CapsSterile, self-standing, round-bottom, internally threaded, polypropylene cryogenic vial with cap
Item added to your cart

ThawSTAR® CFT2 Automated Thawing System
PRODUCTS ARE FOR RESEARCH USE ONLY AND NOT INTENDED FOR HUMAN OR ANIMAL DIAGNOSTIC OR THERAPEUTIC USES UNLESS OTHERWISE STATED. FOR ADDITIONAL INFORMATION ON QUALITY AT STEMCELL, REFER TO WWW.STEMCELL.COM/COMPLIANCE.










