Enrichment of Circulating Tumor Cells
Using Fast and Easy Cell Isolation
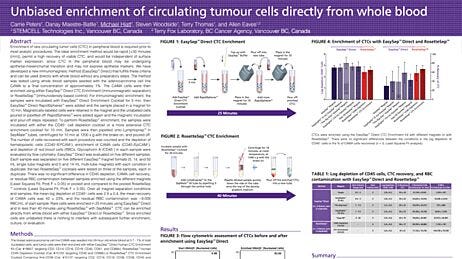
Isolate circulating tumor cells (CTCs) with our innovative cell separation platforms, EasySep™ and RosetteSep™, which provide an easy, fast and effective method for isolating rare cells. By reducing the number of processing steps, both isolation platforms result in better yields and high recovery compared to other cell isolation alternatives.
Enriched CTCs are untouched and immediately ready for
- Cell culture
- DNA/RNA isolation for genetic analyses
- Further purification using microfluidics or cell sorting
- Other downstream assays
How EasySep™ Works
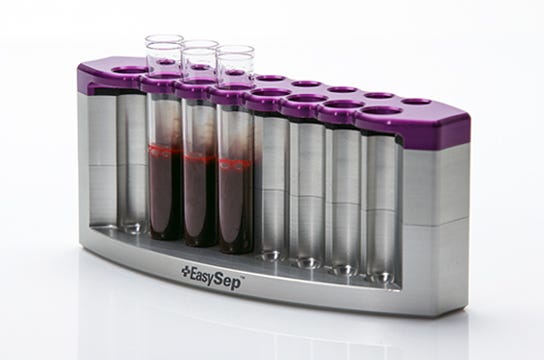
EasySep™ is an immunomagnetic cell isolation platform suitable for the enrichment of CTCs from whole blood, bone marrow and fresh or previously frozen human mononuclear cells. Unwanted cells are targeted for depletion using antibody complexes linked to magnetic particles. Unwanted cells are pulled to the sides of the tube when the sample is placed in an EasySep™ magnet. The enriched cells are then simply poured or pipetted off into a new tube. EasySep™ kits can be completely automated using RoboSep™, the fully automated cell isolation platform.
Quote from EasySep™ User
We adopted for our cell isolation protocols primarily because of the speed but we also benefit from time-savings when FACS-sorting cells thanks to the high purity of cells isolated with EasySep™. Additionally, the protocol is really straight-forward so it is easy for new group members to use the EasySep™ system.
Thomas Krausgruber, PhD, Postdoctoral Fellow, Dr. Christoph Bock's Lab
How RosetteSep™ Works
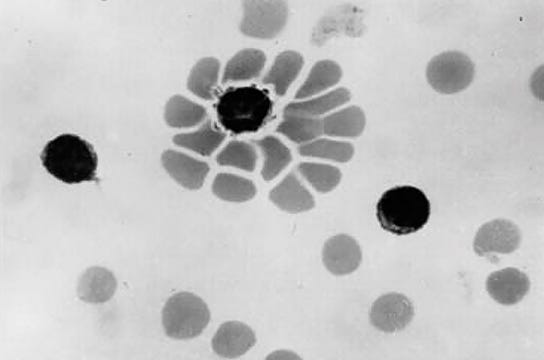
RosetteSep™ kits offer one-step enrichment of cells directly from human whole blood. By crosslinking unwanted cells to red blood cells (RBCs) present in the sample, CTCs are enriched during standard density gradient centrifugation. RosetteSep™ is easy to use, does not require additional equipment, reduces sample handling time and maximizes convenience. RosetteSep™ can be easily combined with SepMate™, a specialized isolation tube that standardizes and minimizes variability when isolating cells using density gradient centrifugation.
Explore These Resources
References
- Clezardin P et al. (1993) Expression of thrombospondin (TSP1) and its receptors (CD36 and CD51) in normal, hyperplastic, and neoplastic human breast. Cancer Res 53(6): 1421–30.
- Kuemmerle NB et al. (2011) Lipoprotein lipase links dietary fat to solid tumor cell proliferation. Mol Cancer Ther 10(3): 427–36.
- Kontogianni K et al. (2005) CD56: a useful tool for the diagnosis of small cell lung carcinomas on biopsies with extensive crush artefact. J Clin Pathol 58(9): 978–80.
- Naito Y et al. (2006) CD56 (NCAM) expression in pancreatic carcinoma and the surrounding pancreatic tissue. Kurume Med J 53(3–4): 59–62.
- Tiemann K et al. (2006) Solid pseudopapillary neoplasms of the pancreas are associated with FLI-1 expression, but not with EWS/FLI-1 translocation. Mod Pathol 19(11): 1409–13.