Isolation of CD34+ Cells from Human Cord Blood
- Document # 27003
- Version 2.0.0
- Dec 2024
Background
The isolation of CD34+ cells from human umbilical cord blood (CB) is an important step in hematopoietic stem and progenitor cell (HSPC) research. Great care must be taken to isolate CD34+ cells with high purities and yields as the frequency of CD34+ cells in CB is low (typically 0.1 - 1% of nucleated cells), sample volumes are small, and cell quality and viability can be variable between different CB samples. This technical bulletin describes two new EasySep™ cell separation kits for the isolation of CD34+ cells from whole CB that can be used with samples containing different amounts of platelets.
Advantages
- Combine your standard density gradient centrifugation step with RosetteSep™ pre-enrichment, then isolate purified CD34+ cells with EasySep™.
- Perform RosetteSep™ separation in SepMate™ tubes to minimize variability between separations and increase sample throughput.
- Use on whole CB with or without platelet depletion.
Positive Selection of Cord Blood CD34+ Cells
CD34+ cells are isolated from whole CB using a simple, two-step procedure. First, standard density gradient centrifugation is combined with pre-enrichment of CD34+ cells using a RosetteSep™ Human Cord Blood CD34 Pre-Enrichment Cocktail containing antibodies that bind to mature blood cells. Two RosetteSep™ antibody cocktails have been developed for CD34+ cell pre-enrichment.
- Human Cord Blood CD34 Positive Selection Kit II (Catalog #17896) contains a RosetteSep™ antibody cocktail that binds to T cells, B cells, red blood cells (RBCs), myeloid cells, and platelets. This cocktail is recommended for pre-enrichment of CD34+ cells from CB samples that contain large amounts of platelets, which may affect the quality and purity of CD34+ cells if not depleted.
- Human Cord Blood CD34 Positive Selection Kit III (Catalog #17897) contains a RosetteSep™ antibody cocktail that binds to T cells, B cells, RBCs, and myeloid cells. This cocktail is recommended for pre-enrichment of CD34+ cells from CB samples containing few platelets or when platelet depletion is not desired.
RosetteSep™ antibody cocktails cross-link unwanted mature cells to RBCs present in a sample, forming dense immunorosettes. Cross-linked samples are then layered over standard density gradient medium (e.g. Lymphoprep™) and separated by centrifugation (Figure 1, steps 1 - 3). The fraction enriched for CD34+ cells is collected from the interface between the plasma and density gradient medium, while unwanted mature cells and RBCs stay behind in the pellet (Figure 1, steps 3 - 4).
Density gradient centrifugation can also be performed using a SepMate™ tube. The SepMate™ tube contains a unique insert that prevents the density gradient medium and blood sample from mixing. The density gradient medium is first added to the tube by pipetting through a central hole in the insert. The sample is then poured or rapidly pipetted on top of the insert. This eliminates the need to carefully layer the sample directly onto the density gradient medium, an otherwise time-consuming step. Only 10 minutes of centrifugation is required, and this step can be carried out with the centrifuge brake on, further reducing the total time necessary for separation. After centrifugation, the fraction enriched for CD34+ cells is simply poured into a new tube.
In the second step, the pre-enriched CD34+ cells are labeled by incubating with the EasySep™ isolation cocktail and magnetic EasySep™ Dextran RapidSpheres™ (Figure 1, steps 5 - 6). The CD34+ cells are then isolated by placing the tube containing the immunomagnetically-labeled cell suspension into an appropriate EasySep™ magnet. After a brief incubation period, unwanted CD34- cells are simply poured off, while the desired CD34+ cells remain in the tube (Figure 1, steps 7 - 8). Isolated CD34+ cells can then be used for downstream applications.

EasySep™ Human Cord Blood CD34 Positive Selection Kit and EasySep™ magnet (magnet sold separately)
Platelet Removal
Depletion of platelets during RosetteSep™ pre-enrichment, prior to EasySep™ CD34+ selection, allows for the improved recovery of higher quality purified cells than with traditional centrifugebased methods. However, in some cases platelet depletion may not be necessary or desired.
- The RosetteSep™ antibody cocktail in Kit II (Catalog #17896) labels platelets, depleting these cells during the pre-enrichment step. This kit is recommended for samples that are > 24 hours old and that may show variable amounts of platelet activation and/or aggregation. Results of typical experiments with Kit II are shown in Figure 2 and Table 1 (page 3).
- The RosetteSep™ antibody cocktail in Kit III (Catalog #17897) does not label platelets and is recommended for very fresh CB samples (< 24 hours after delivery), samples that contain very few platelets, and samples for which platelet depletion is not desired. Results of typical experiments with Kit III are shown in Figure 3 and Table 2 (page 3).
General Protocol for Pre-Enrichment and Positive Selection of CD34+ Cells
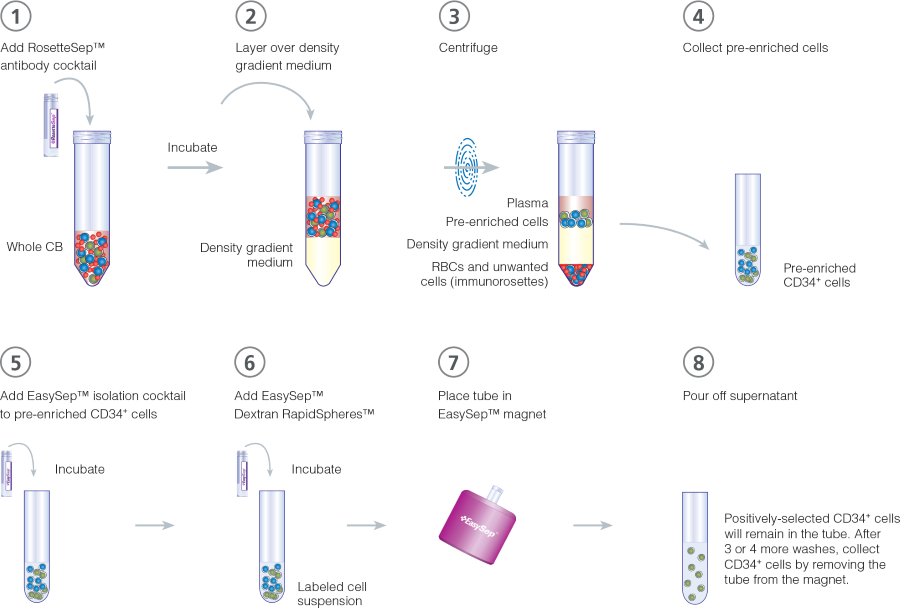
Figure 1. Combined RosetteSep™ and EasySep™ Protocol for Isolation of CD34+ Cells
CD34+ cells are isolated from whole CB using RosetteSep™ (steps 1 - 4) and EasySep™ (steps 5 - 8). Pre-enrichment with RosetteSep™ can be performed with SepMate™ tubes to allow faster layering of a sample over the density gradient medium as an insert within the tube prevents layers from mixing. After centrifugation, with the brake on when using SepMate™, or with the brake off for standard conical tubes, pre-enriched CD34+ cells are collected into a fresh tube (step 4). The EasySep™ isolation cocktail and Dextran RapidSpheres™ are then added to the pre-enriched cells to label the CD34+ cells (steps 5 - 6). The labeled cells are placed into a magnet to positively select for CD34+ cells. After isolation, purified cells are ready for downstream applications. For detailed procedures on manual cell separation methods using either the EasySep™ purple magnet (Catalog #18000), “The Big Easy” magnet (Catalog #18001), the EasyEights™ magnet (Catalog #18103), or for automated cell separation using RoboSep™ instruments (Catalog #21000/23000), please refer to the Product Information Sheet (PIS) for each kit.
Data
Human Cord Blood CD34 Positive Selection Kit II
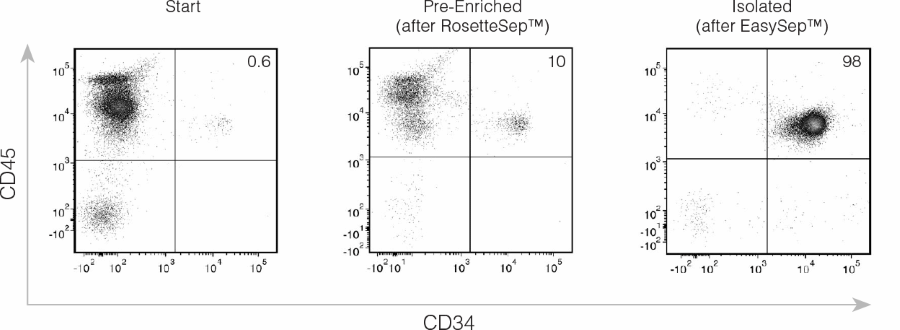
Figure 2. Isolation of CD34+ CB Cells Using EasySep™ Human Cord Blood CD34 Positive Selection Kit II
CD45 and CD34 expression of cells before separation (“Start”), after RosetteSep™ (“Pre-Enriched”), and after selection of CD34+ cells (“Isolated”) using EasySep™ Kit II (Catalog #17896). Results of a typical experiment are shown. CD45+CD34+ HSPCs (top right quadrant) have been enriched > 15-fold (from 0.6% to 10%) after RosetteSep™ pre-enrichment and > 200-fold (from 0.6% to 98%) after EasySep™ CD34+ selection (“Isolated”). The flow cytometry data shown are gated on cells with intermediate to high forward light scatter (FSC), and which are negative for propidium iodide (PI) staining to exclude debris, RBCs, platelets and dead cells. The results of cell separations with 15 different CB samples are summarized in Table 1.
Table 1. Purity of CD34+ Cells Isolated from Different CB Samples Using EasySep™ Human Cord Blood CD34 Positive Selection Kit II

Human Cord Blood CD34 Positive Selection Kit III
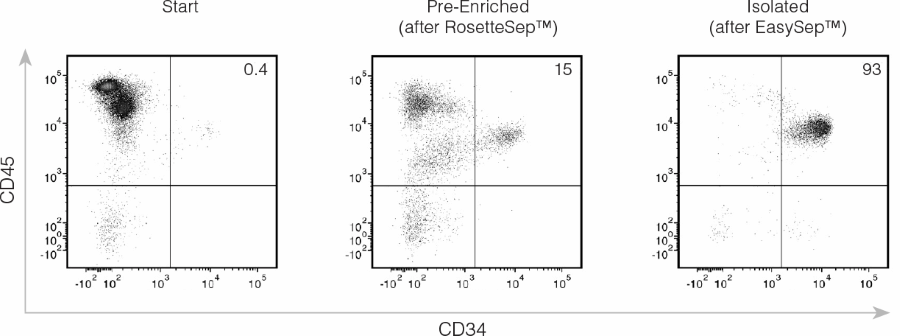
Figure 3. Isolation of CD34+ CB Cells Using EasySep™ Human Cord Blood CD34 Positive Selection Kit III
CD45 and CD34 expression of cells before separation (“Start”), after RosetteSep™ (“Pre-Enriched”), and after selection of CD34+ cells (“Isolated”) using EasySep™ Kit III (Catalog #17897). Results of a typical experiment are shown, gated as described in Figure 2. The results of cell separations with 10 different CB samples are summarized in Table 2.
Table 2. Purity of CD34+ Cells Isolated from Different Fresh CB Samples Using EasySep™ Human Cord Blood CD34 Positive Selection Kit III

Product Information

*Each kit contains reagents sufficient for processing up to 1000 mL of CB.
Try using these EasySep™ kits to isolate CD34+ cells from cord blood in your own lab.
Unsure which CD34 positive selection kit to choose? We can help. Contact us by phone or email or use the LiveChat function on this page to discuss your specific needs and applications.
Required Reagents*
- 50 mL conical tubes, e.g. SepMate™-50 tubes (Catalog #15450)
- 5 mL or 14 mL polystyrene round-bottom tubes
- EasySep™ Buffer (Catalog #20144), RoboSep™ Buffer (Catalog # 20104), or PBS containing 2% fetal bovine serum (FBS) and 1 mM EDTA
- Density gradient medium, e.g. Lymphoprep™ (Catalog #18060)
- EasySep™ Magnet (Catalog #18000/18001/18103) or RoboSep™ fully automated cell separator (Catalog #21000/23000)
*Not included in the kits
Other Kits for the Isolation of Human CD34+ Cells
- To isolate CD34+ cells from fresh adult peripheral blood or buffy coat cells, we recommend the Complete Kit for Human Whole Blood CD34+ Cells (Catalog #15086).
- To isolate CD34+ cells from other samples, including fresh or previously frozen mobilized peripheral blood or bone marrow mononuclear cells, or from previously frozen CB mononuclear cells, we recommend using the EasySep™ Human CD34 Positive Selection Kit II (Catalog #17856).
Culture of CD34+ Cells
After isolation, CD34+ cells may be cultured using specialized cell culture media such as StemSpan™ serum-free media supplemented with cytokines and/or other supplements. StemSpan™ expansion supplements contain optimized combinations of cytokines and supplements to either promote the expansion of HSPCs or their lineage-specific differentiation to generate large numbers of mature erythroid, megakaryocyte, or myeloid cells, dependant on the type of supplement used. Purified CD34+ cells can also be analyzed in colony-forming unit (CFU) assays using methylcellulose-based MethoCult™ medium, and in LTC-IC assays using MyeloCult™ medium.
Related Resources
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration