Frequently Asked Questions on SepMate™
Find answers to frequently asked questions (FAQ) about SepMate™ isolation tubes, which allow for hassle-free peripheral blood mononuclear cell (PBMC) isolation in just 15 minutes.
Can I use SepMate™ with samples other than whole blood and bone marrow (e.g., LRSC, buffy coat, leukopak, cell suspension from tissue) or with non-human whole blood samples?
The intended use for SepMate™ products does not include leukopaks, buffy coat, leukocyte reduction system chamber (LRSC), cell suspensions from other tissues, or non-human samples. However, if you are interested in processing these samples using SepMate™, please contact Product and Scientific Support for additional information.
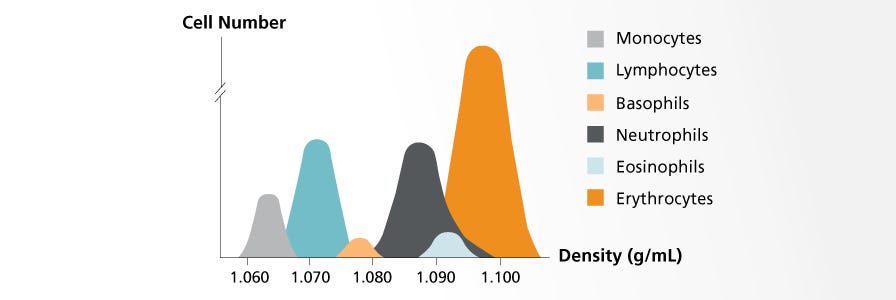
Which density gradient medium can be used with SepMate™?
A medium with a density of 1.077 g/mL that is designed for the separation of mononuclear cells, such as Lymphoprep (Catalog #18060), is recommended for use with SepMate™ products. For questions regarding SepMate™'s compatibility with other density media, please contact Product and Scientific Support.
The Principles of Using Density Gradient Centrifugation to Isolate Cells
Learn more about density gradient centrifugation and the several types of density gradient media.
Does it matter what type of rotor the centrifuge has?
Swinging bucket rotors are recommended for use with SepMate™ products during density centrifugation-based isolation procedures as they provide the highest particle (cell) separation resolution.
Which brake settings on the centrifuge should I use?
While the SepMate™ product information sheet specifies use of the brake, aggressive deceleration force may perturb the mononuclear cell (MNC) band at the plasma-Lymphoprep™ interface, resulting in a “cloudy” or diffuse suspension of cells. Different makes and models of centrifuge may provide different rates of deceleration when braking. If there is no visible layer of MNCs following centrifugation with the brake set to high, reduce the rate of deceleration (braking) to medium or low for the next separation.
Is it normal to not see a distinct MNC band at the interface of plasma and density gradient medium, above the insert? Instead, I see a ”cloudy” or dispersed suspension of cells in the entire volume above the insert.
A distinct MNC band should be seen at the plasma-density gradient medium interface following centrifugation. A “cloudy” or diffuse suspension of cells typically indicates insufficient separation (centrifuge speed/time) or sample mixing due to shaking/vibrations/shuddering (centrifuge imbalance) during braking; sudden stopping or aggressive deceleration force during braking are also known to contribute to a “cloudy” or diffuse MNC band, and an intermediate or low brake setting may help minimize this effect. However, a “cloudy” or diffuse MNC band will not negatively affect isolation performance as the entire supernatant is harvested via pour-off.
I observed some RBC contamination after MNC isolation; is this normal?
Residual RBC contamination is typically sample-dependent and is most commonly observed in older blood samples (>24 hours old); if significant RBCs are present above the insert, the SepMate™ tube can be spun at 1200 x g for an additional 10 minutes with the brake on. Alternatively, if the presence of residual RBCs is a concern, they may be lysed by performing one of the wash steps with Ammonium Chloride Solution (Catalog #07800).
Can I isolate neutrophils from whole blood using SepMate™?
SepMate™ is not recommended for the isolation of neutrophils from whole blood. When used in conjunction with Lymphoprep™ or Ficoll, the density of neutrophils is such that they will sediment through the density gradient medium solution during centrifugation. This means that the neutrophils will become trapped below the insert.
If you want to isolate human neutrophils from human whole blood, learn more about immunomagnetic isolation using EasySep™ Direct Human Neutrophil Isolation Kit (Catalog # 19666).
Isolate Neutrophils Directly from Whole Blood with EasySep™ Direct
EasySep™ Direct immunomagnetically depletes red blood cells and unwanted cells in a single step without density gradient centrifugation, RBC lysis, or other pre-processing steps that can alter cellular function or increase cell isolation time.
Can I use 1X PBS instead of 1X PBS + 2% FBS?
1X PBS can be used in lieu of 1X PBS + 2% FBS when using SepMate™ products. However, 2% FBS is typically included to maintain cell viability in addition to minimizing non-specific binding of antibodies in downstream assays.
Which anticoagulant should be used and why?
SepMate™ is compatible for use with blood collected using all types of anticoagulants including, but not limited to, heparin, Anticoagulant Citrate Dextrose Solution A (ACDA), and ethylenediamine tetraacetic acid (EDTA).
Can I automate PBMC isolations using SepMate™?
SepMate™ is a great tool for consistent and hassle-free peripheral blood mononuclear cell (PBMC) isolation. It reduces the procedure to just 15 minutes, but it can’t be used to automate the process. If you are interested in automation of PBMC isolation, we recommend exploring using EasySep™ Direct Human PBMC Isolation Kit together with RoboSep™ instruments. With these tools, you can automate PBMC isolation to save hands-on time, minimize sample handling, and increase sample processing throughput.
PBMCs: Everything You Need to Know
Find protocols and tools to help you isolate, source, freeze, and thaw human PBMCs in your research.
Additional Resources
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration