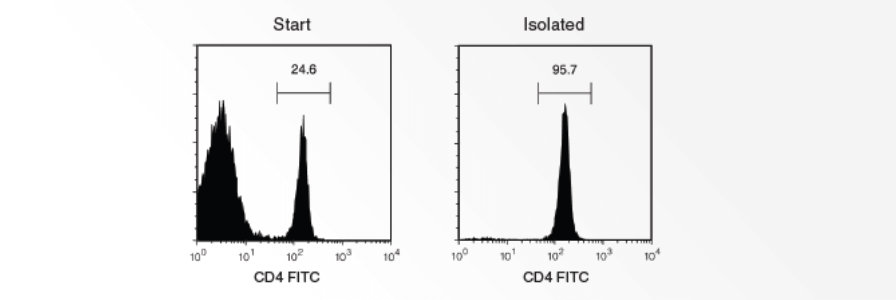
How to Isolate Specific Cell Subsets by Immunodensity Cell Separation
Using a RosetteSep™ Antibody Cocktail to isolate a particular cell type of interest; specific conditions may vary according to the cell type being enriched
The following procedure provides guidelines for isolating cells from whole blood using RosetteSep™, an immunodensity cell separation platform that isolates specific cell subsets during density gradient centrifugation. RosetteSep™ first crosslinks unwanted cells to red blood cells (RBCs) present in the sample to form immunorosettes. When the sample is centrifuged over a density gradient medium, the immunorosettes pellet, leaving highly purified cells at the interface between the plasma and the density gradient medium. This is a general procedure for using a RosetteSep™ Antibody Cocktail to isolate a particular cell type of interest; specific conditions may vary according to the cell type being enriched.
Materials
- Whole blood sample
- RosetteSep™ antibody cocktail for your desired cell subset
- Phosphate-buffered saline containing 2% fetal bovine serum (PBS + 2% FBS; e.g. Dulbecco's Phosphate Buffered Saline with 2% Fetal Bovine Serum, Catalog #07905)
- Density gradient medium (e.g. Lymphoprep™, Catalog #18060)
Protocol
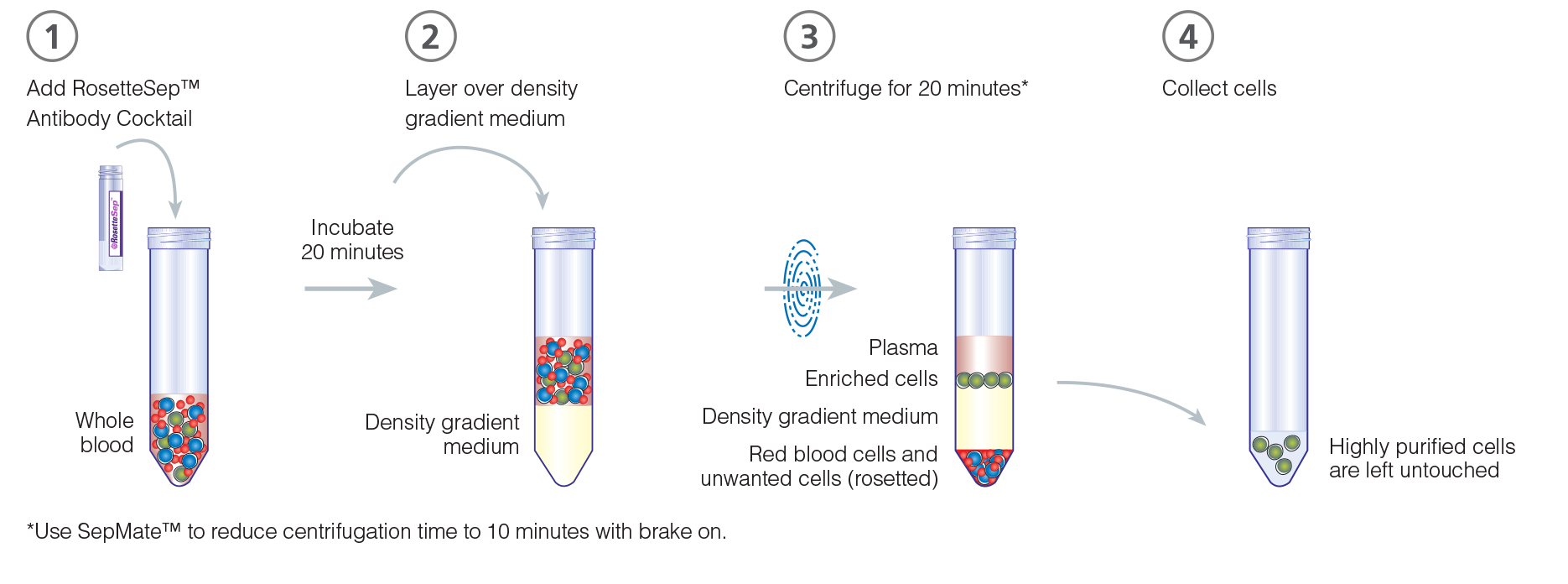
- Add RosetteSep™ cocktail to the sample and mix well. Incubate at room temperature (15 - 25°C) for 20 minutes.
-
Dilute sample with an equal volume of PBS + 2% FBS and mix gently. Layer the diluted sample on top of the density gradient medium using the recommended volumes (Table 1). Be careful to minimize mixing of the density gradient medium and the sample.
Table 1. Recommended Volumes and Tube Sizes for Immunodensity Cell Separation.
Sample Size (mL)Tube Size (mL)PBS + 2% FBS (mL)Density Gradient Medium (mL)1511.52142331433414445505151050101515501515
-
Centrifuge at 1200 x g for 20 minutes at room temperature, with the brake off.
Tip: Use SepMate™ to reduce centrifugation time to 10 minutes with the brake on. -
Remove the enriched cells from the density medium:plasma interface. Wash enriched cells with PBS + 2% FBS. Repeat.
Note: It may be difficult to see cells at the interface, especially when very rare cells are enriched. The following protocol extension can be used to maximize recovery in these instances.Optional Protocol Extension to Maximize Recovery of Rare Cells:
a. Remove some of the density gradient medium along with the enriched cells.
b. Add PBS + 2% FBS. Centrifuge at 300 x g for 10 minutes with the brake on low. Discard supernatant. Repeat. Enriched cells are now ready for use.
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration