How to Generate Human Intestinal Organoid-Derived Monolayers Using IntestiCult™
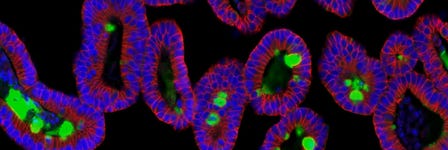
Intestinal organoids provide researchers with a physiologically relevant cell model and constitute a valuable experimental tool for probing intestinal epithelial cell biology and modeling disease. However, intestinal organoid cultures have the limitation of being a closed luminal compartment—a physical characteristic that presents a challenge for experiments requiring access to the apical surface.
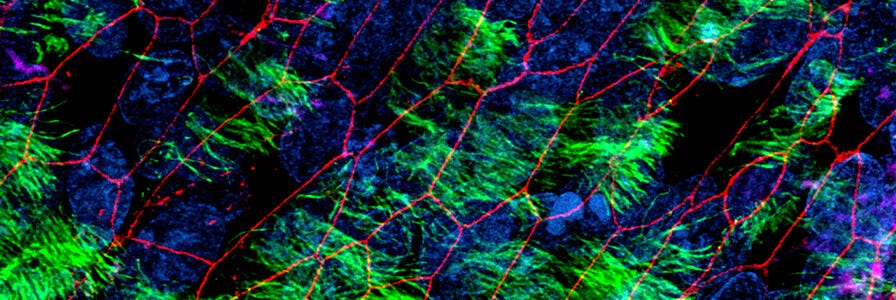
Researchers can generate monolayers from intestinal organoids to enable easy access to the apical surface, facilitating studies such as those involving apical cell surface receptors, interaction with commensal or pathogenic microorganisms, or modeling the effects of potentially beneficial or harmful compounds in intestinal contents.
In this protocol, we describe a method to generate human intestinal organoid-derived monolayer cultures using IntestiCult™ Organoid Differentiation Medium (ODM) (Human). Using this procedure, cells expanded as human intestinal organoid cultures are seeded onto a dilute culture matrix, where they form a confluent monolayer and differentiate to model the intestinal epithelium.
Intestinal epithelial monolayer cultures can be established on glass coverslips that enable high-quality imaging or on Transwell® membrane inserts that enable measurement of active and passive transport of substances across the epithelial layer and electrical barrier function assays. Monolayers can be generated using a range of cultureware, giving flexibility in experimental throughput.
Researchers working with specific applications may need to further optimize this protocol.
Materials
- IntestiCult™ Organoid Differentiation Medium (Human) (Catalog #100-0214)
- Costar® 6.5 mm Transwell® inserts (Catalog #38024) or Costar® 12 mm Transwell® inserts (Catalog #38023)
- Corning® Matrigel® Matrix, Growth Factor Reduced (GFR), Phenol Red-Free (Corning® #356231)
- D-PBS (e.g. Catalog #37350)
- 15 mL conical tubes (e.g. Falcon® Conical Tubes, Catalog #38009)
- Y-27632 (Catalog #72302)
- Antibiotics (e.g gentamicyn or penicillin/streptomycin)
Protocol
Note: Researchers who need large numbers of organoid monolayers may wish to use the organoid suspension culture protocol (see section VI below) to facilitate the generation of a higher number of organoids per well.
I. Coating Cultureware with Corning® Matrigel®
For monolayer organoid culture, cultureware must be coated with Corning® Matrigel®, as described below. For optimal results, Costar® 6.5 mm or 12 mm Transwell® inserts (Catalog #38023/38024) are recommended; however, standard tissue culture-treated plates may be used. If ALI culture is desired to increase differentiation of the epithelial layer (see section V), Transwell® inserts must be used for monolayer culture.
Note: Defined matrices such as Collagen I (e.g. Catalog #04902), Collagen IV, or Vitronectin XF™ (Catalog #07180) can be used instead of Matrigel®, but protocols may require further optimization.
- Thaw one aliquot of Corning® Matrigel® on ice.
- Dispense an appropriate amount of cold (2 - 8°C) D-PBS into a 15 mL conical tube and place on ice. Refer to Table 1 below for recommended volumes.
- Add thawed Matrigel® to the cold D-PBS at a ratio of 1 µL Matrigel® to 49 µL D-PBS. Mix thoroughly.
- Immediately coat cultureware with diluted Matrigel®. Swirl the cultureware to spread the solution evenly across the surface.
- Incubate at 37°C for at least 1 hour before use. Do not let the Matrigel® solution evaporate.
Note: If not used immediately, seal the cultureware with Parafilm® to prevent evaporation; store at 2 - 8°C for up to 1 week after coating. Allow stored coated cultureware to warm to room temperature (15 - 25°C) for 30 minutes before proceeding to the next step.
- Gently tilt the cultureware to one side and allow the excess Matrigel® solution to collect at the edge. Remove the excess Matrigel® solution using a serological pipette or by aspiration. Ensure that the coated surface is not scratched.
Table 1. Recommended Volumes of Diluted Matrigel® for Various Cultureware
Cultureware Volume of Diluted Matrigel® Per Well 6.5 mm Transwell® insert 100 µL (top) 12 mm Transwell® insert 250 µL (top) 6-well plate 1000 µL 24-well plate 250 µL 96-well plate 100 µL
II. Media Preparation
Use sterile technique to prepare IntestiCult™ Monolayer Growth Medium (IntestiCult™ ODM Human Basal Medium + Organoid Supplement + Y-27632). The following example is for preparing 100 mL of complete medium. If preparing other volumes, adjust accordingly.
- Prepare a 10 mM stock solution of Y-27632 in DMSO. Store at -20°C until ready to use.
- Thaw Basal Medium and Organoid Supplement at room temperature (15 - 25°C) or at 2 - 8°C overnight. Mix thoroughly.
Note: If not used immediately, aliquot and store at -20°C for up to 3 months. After thawing the aliquots, use immediately. Do not refreeze.
- Add 50 mL of Organoid Supplement to 50 mL of Basal Medium. Mix thoroughly.
- Add 100 µL of 10 mM Y-27632 (final concentration 10 µM). Mix thoroughly.
Note: If not used immediately, store at 2 - 8°C for up to 1 week.
- Add desired antibiotics immediately before use (e.g. 50 µg/mL gentamicin or 100 units [100 µg/mL] penicillin/streptomycin).
III. Dissociation of Human Intestinal Organoids
For complete instructions for growing human intestinal organoids using IntestiCult™ Organoid Growth Medium (Human), refer to the corresponding Product Information Sheet.
The following protocol is for dissociating 2 - 3 wells of human intestinal organoids grown in IntestiCult™ Organoid Growth Medium (Human) in a 24-well plate for 7 - 8 days in 50 µL 50% Matrigel® domes. If seeding monolayer cultures on a larger scale, we recommend using organoids grown in suspension culture to increase organoid density per well (see section VI).
Intestinal organoids should be passaged no more than 15 times if being used for monolayer cultures; seeding efficiency may vary with organoid passage number and the number of days of growth. This protocol assumes at least 100 - 150 organoids per well. Matrigel® domes may contain fewer but larger organoids, or a larger number of smaller organoids, but will yield similar results. The number of organoids required may vary with donor and culture quality and may need to be further optimized.
- Refer to Table 2 below for the recommended number of wells of intestinal organoids to harvest for various cultureware.
Note: If ALI culture is desired to increase differentiation of the epithelial layer (section V), Transwell® inserts must be used for monolayer culture.
- Aspirate all medium from the organoid cultures without disturbing the organoids within the Matrigel® domes.
- Add 1 mL of Gentle Cell Dissociation Reagent to each well of organoids to be harvested.
- Incubate at room temperature (15 - 25°C) for 1 minute.
- Using a 1 mL pipettor, vigorously pipette up and down to disrupt the Matrigel® dome and resuspend the organoids.
- Pool the harvested wells in a 15 mL conical tube. Incubate at room temperature for 10 minutes with gentle agitation or rocking.
- Centrifuge at 200 x g for 5 minutes at 2 - 8°C.
- Remove and discard the supernatant. Add 5 mL ice-cold DMEM/F-12 with 15 mM HEPES to resuspend organoids. Centrifuge at 200 x g for 5 minutes at 2 - 8°C.
- Aspirate supernatant, removing as much as possible, being careful not to disturb the pellet. Add 1 mL of warm (37°C) Trypsin-EDTA (0.05%) to resuspend organoids.
Note: If pooling larger numbers of organoid wells, increase the volume of Trypsin-EDTA to ensure efficient dissociation of the organoids.
- Using a 1 mL pipettor, pipette up and down to mix thoroughly. Incubate at 37°C for 5 - 10 minutes.
- Mix thoroughly by vigorous pipetting or vortexing to disrupt the organoids as much as possible. Use a microscope to check the organoids for sufficient disruption. Organoids should be dissociated into either individual cells or small fragments. If many large fragments or whole organoids remain, repeat pipetting/vortexing until fragments are sufficiently disrupted.
Note: Perform the remaining steps as quickly as possible, as cells will start to clump together.
- Add an equal volume of DMEM/F-12 (e.g. 1 mL DMEM/F-12 per mL Trypsin-EDTA) and pipette up and down to mix thoroughly. Centrifuge fragments at 200 x g for 5 minutes at 2 - 8°C.
Note: DMEM/F-12 supplemented with 5 - 10% FBS can also be used to neutralize Trypsin-EDTA if needed. Carefully remove and discard the supernatant.Note: If a buoyant mucus layer is present, steps 11 and 12 may need to be repeated to properly pellet the organoid fragments.
Table 2. Recommended Number of Wells of Intestinal Organoids to Harvest
Cultureware Number of Wells of Intestinal Organoids to Harvest
(per well to be seeded)6.5 mm Transwell® insert 2 - 3 wells 12 mm Transwell® insert 3 - 4 wells 6-well plate 6 - 8 wells 24-well plate 3 - 4 wells 96-well plate 1 - 2 wells
IV. Plating Organoid Fragments for Monolayer Culture
- Coat cultureware with Corning® Matrigel® and prepare IntestiCult™ Monolayer Growth Medium (see section II).
- Add IntestiCult™ Monolayer Growth Medium (prepared in section II) to organoid fragments and mix gently. Refer to Table 3 below for the volume required for various cultureware.
- Slowly and gently add the fragment suspension to each Matrigel®-coated well. Incubate at 37°C and 5% CO2. Monitor growth daily. Perform a full-medium change every 2 - 3 days.
Note: Monolayers will usually reach 100% confluency within 2 - 3 days; however, if attachment is poor, this may take longer. Cells will remain viable and the monolayer will remain confluent for at least 3 weeks, with continued full-medium changes every 2 - 3 days.
- For air-liquid interface (ALI) culture, proceed to section V.
Table 3. Recommended Resuspension Volume of IntestiCult™ Monolayer Growth Medium for Various Cultureware
Cultureware Volume of IntestiCult™ Monolayer Growth Medium Per Well 6.5 mm Transwell® insert 100 µL (top), 500 µL (bottom) 12 mm Transwell® insert 500 µL (top), 1.5 mL (bottom) 6-well plate 1.5 mL 24-well plate 500 µL 96-well plate 100 µL
V. Air-Liquid Interface (ALI) Differentiation (Optional)
The following instructions are for using an ALI culture to increase differentiation of the epithelial layer. This is an optional step that is not necessary for all applications. The result will be a significant increase in the number of secretory and other differentiated cell types at the expense of stem cells and transit-amplifying cells. Organoid-derived human intestinal epithelial cell monolayer cultures should be established in a Transwell® insert. When the monolayer has been 100% confluent for at least 4 days, proceed with the protocol below.
- Remove medium from the top and bottom wells. Add fresh IntestiCult™ Monolayer Growth Medium to the bottom well; leave the top well empty. Incubate at 37°C and 5% CO2.
- Every 2 - 3 days, perform a full-medium change in the bottom well using fresh IntestiCult™ Monolayer Growth Medium. Allow the monolayer to differentiate for at least 1 week. The ALI culture can be maintained for 2 weeks or longer, with full-medium changes every 2 - 3 days.
Note: The upper well can be washed with 100 µL D-PBS to rinse away excess mucus; however, this is not necessary and the presence of mucus may be desirable to better represent the physiology of the intestinal epithelium.
VI. Organoid Suspension Culture Protocol for Establishing Large Numbers of Organoid Monolayers (Optional)
The protocol below is an alternative to the protocol in the Product Information Sheet (PIS) for IntestiCult™ Organoid Growth Medium (Human), in which organoids are suspended in Matrigel® domes. This protocol describes how to passage organoids from Matrigel® domes into a suspension culture, 7 days prior to establishing monolayer cultures. This alternative method for organoid growth will facilitate a higher number of organoids per well, reducing the amount of work needed when establishing larger numbers of organoid monolayers.
- Start with organoid cultures grown in 50 µL 50% Matrigel® domes in a 24-well plate as described in the IntestiCult™ Organoid Growth Medium (Human) PIS. Each well of a 6-well suspension plate will require 4 - 6 wells of organoids (600 - 1000 organoids).
- Prepare IntestiCult™ Organoid Growth Medium (Human) and warm to room temperature (15 - 25°C).
Note: For each well of a 6-well suspension plate, 3 mL of medium will be required.
- Add 3 mL of IntestiCult™ Organoid Growth Medium (Human) per well of a 6-well Ultra-Low Adherent Plate for Suspension Culture. Place in a 37°C incubator for at least 1 hour.
- Thaw Corning® Matrigel® on ice; for each well of a 6-well plate, 500 µL of Matrigel® will be required. Place DMEM/F-12 with 15 mM HEPES on ice.
- Carefully remove and discard medium from each well to be passaged, without disturbing the Matrigel® dome.
- Add 1 mL of room temperature Gentle Cell Dissociation Reagent (GCDR) on top of the exposed dome in each well. Incubate at room temperature for 1 minute.
- Pre-wet a 1 mL pipette tip with GCDR; use this pipette tip to thoroughly scrape the Matrigel® dome free of the bottom of the well. Pipette the GCDR in the well up and down 2 - 3 times to break up the dome and the organoids; avoid touching the bottom of the well with the pipette tip. Ensure all pieces of Matrigel® have been rinsed free of the plate.
- Using the same pipette tip, transfer the organoid mixture to a 15 mL conical tube.
- Add 1 mL GCDR to the newly emptied well. Using a pipette tip pre-wetted with GCDR, pipette the GCDR up and down 2 - 3 times to rinse the well. Transfer the contents of the well to the 15 mL conical tube from step 8.
- Repeat steps 7 - 9 for each well to be passaged.
- Incubate the tubes at room temperature on a rocking platform set at medium speed (~40 rpm) for 10 minutes.
- Centrifuge the tubes at 290 x g for 5 minutes at 2 - 8°C. Gently pour off and discard the supernatant.
- Add 5 mL of cold DMEM/F-12 to each tube. Using a pre-wetted 1 mL pipette tip, vigorously pipette up and down 15 times to resuspend and fragment organoids.
Note: Avoid touching the side/bottom of the tube with the pipette tip.
- Centrifuge at 200 x g for 5 minutes. Aspirate as much supernatant as possible.
- Resuspend the organoid fragments in each tube in 500 µL cold Matrigel®. Mix thoroughly by gently pipetting up and down 10 - 15 times (try to avoid introducing air bubbles). Always keep the Matrigel® suspension on ice.
Note: Add 10 µM Y-27632 to increase organoid yield.
- Remove the 6-well suspension plate from the incubator (prepared in step 3). Slowly pipette the organoid-Matrigel® suspension into the center of the medium in one well. The Matrigel® should immediately solidify and remain suspended in the medium. Repeat for each tube of organoid-Matrigel® suspension.
- Incubate at 37°C and 5% CO2. Cultures can be maintained for up to 7 days. Perform a half-medium change every 2 - 3 days, as follows:
a. Tilt the plate and use a pipette tip to gently push the Matrigel® suspension away from a corner of the well.
b. Use the pipettor to gently remove 1.5 mL of medium (do not aspirate). Be careful not to remove any of the Matrigel® suspension.
c. Add 1.5 mL fresh IntestiCult™ Organoid Growth Medium (Human).Note: Each well of a 6-well suspension plate should yield at least an equivalent number of organoids as 18 wells of a 24-well plate with dome cultures (1800 - 2400 organoids). - To establish a monolayer culture, transfer the organoid suspension from one well of the 6-well suspension plate to a 15 mL conical tube. Centrifuge at 200 x g for 5 minutes. Aspirate as much supernatant as possible.
- Add 5 mL GCDR to the organoid pellet and mix gently to resuspend. Incubate at room temperature for 10 minutes with gentle agitation or rocking.
- Proceed to section III, step 7 to dissociate organoids for monolayer culture.
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration