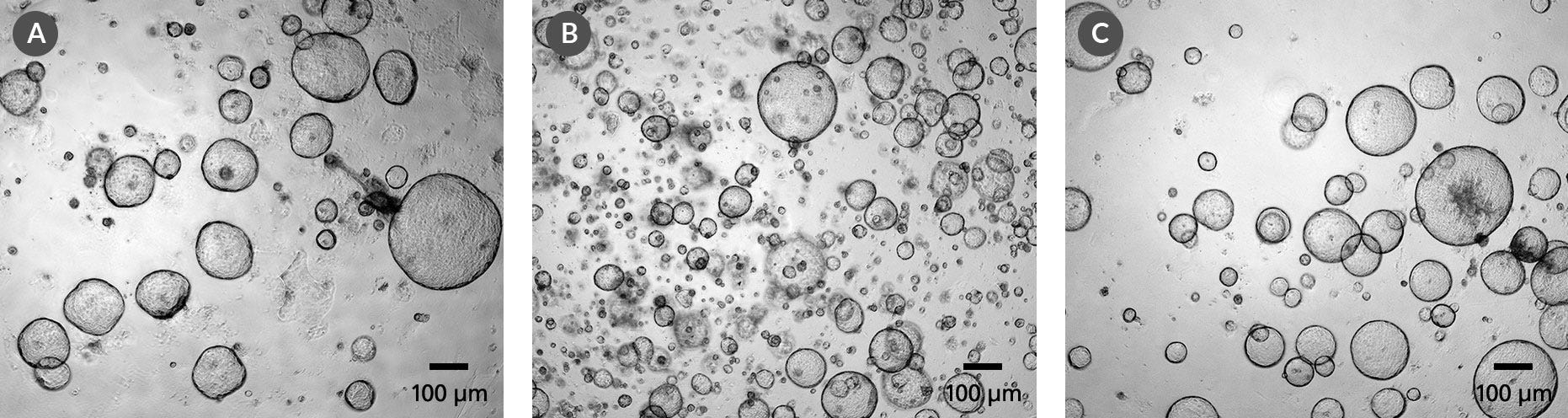
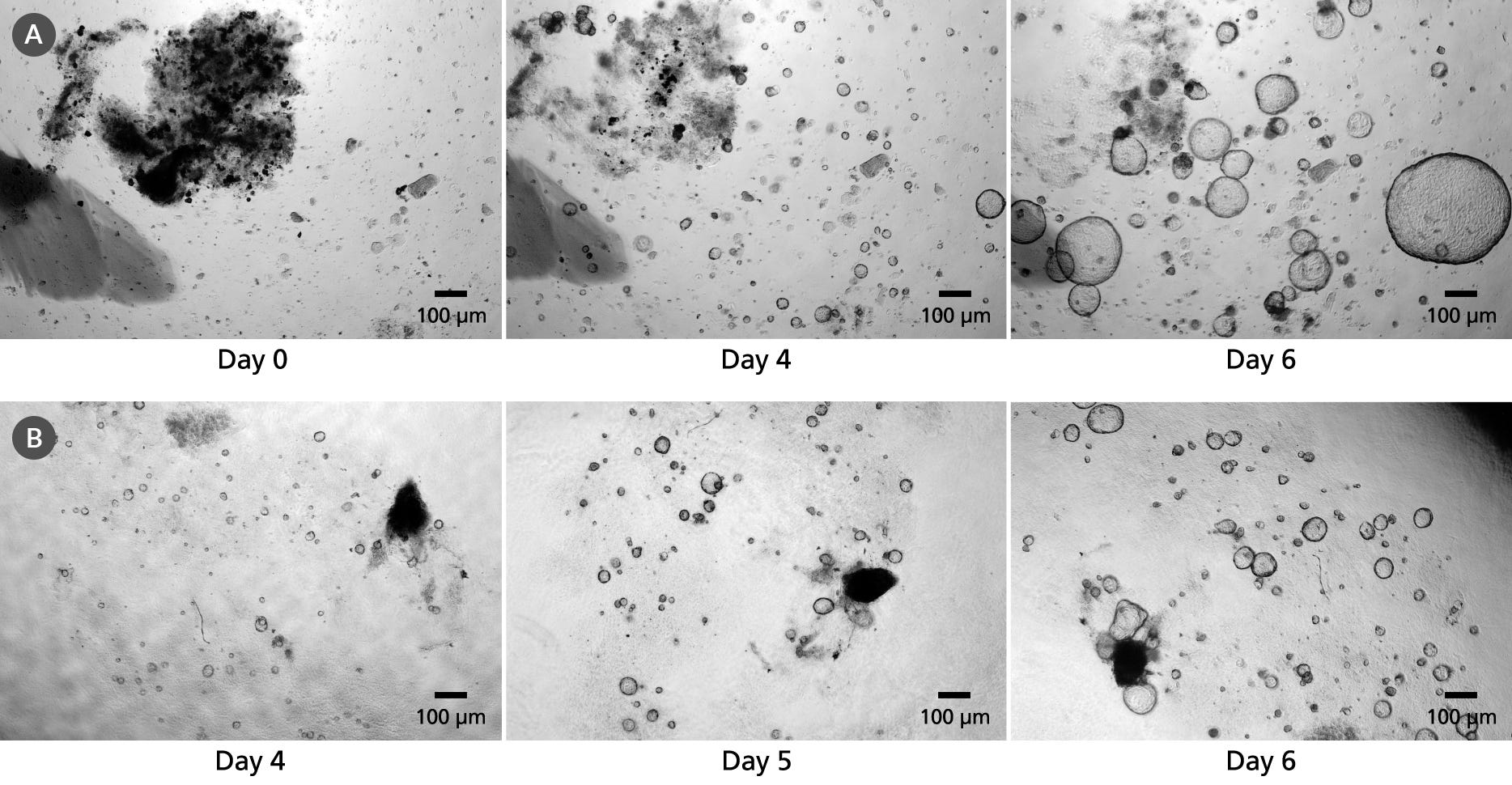
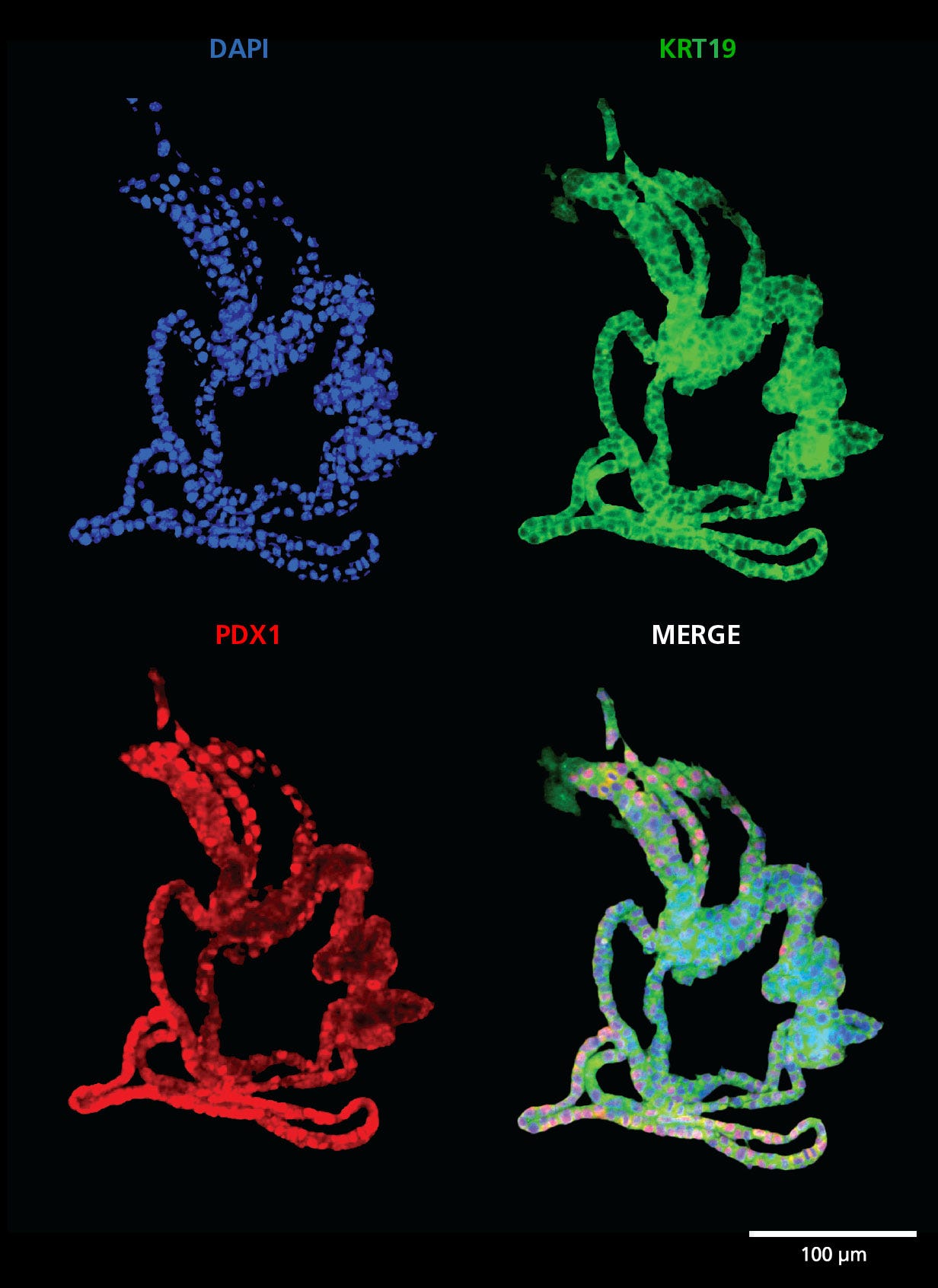
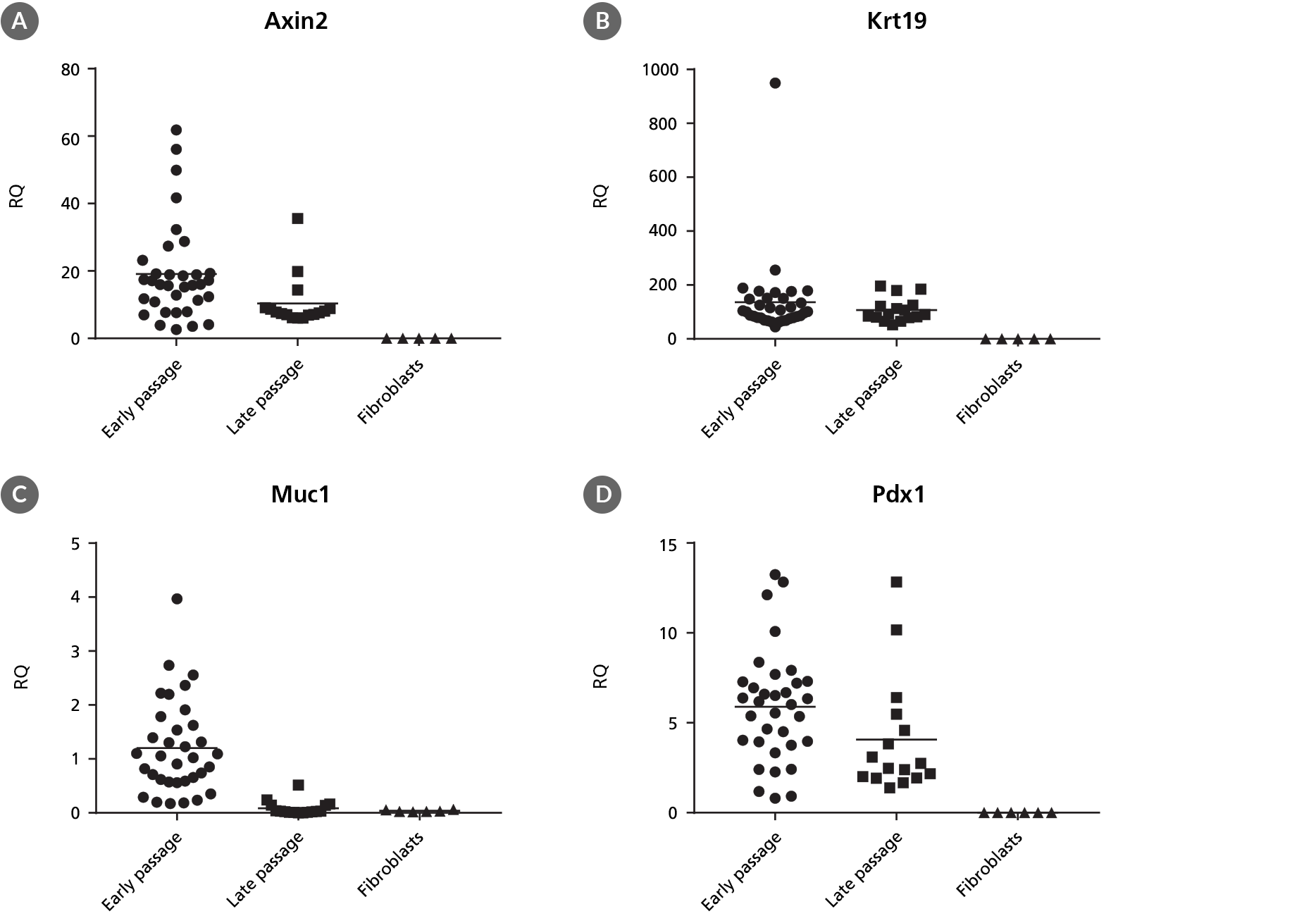
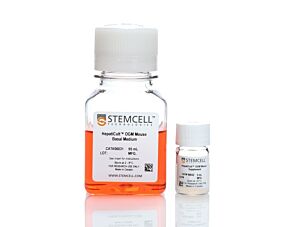
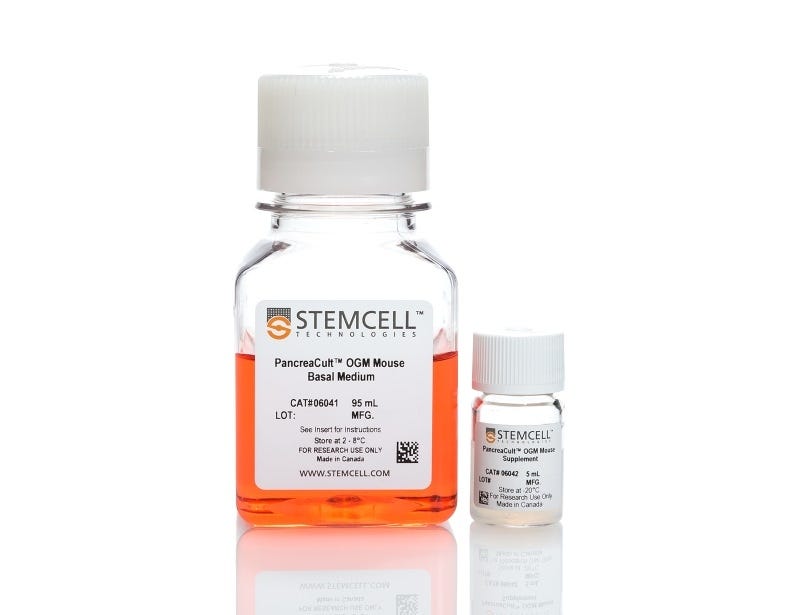
PancreaCult™ Organoid Growth Medium (Mouse)
Cell culture medium for establishment and maintenance of mouse pancreatic exocrine organoids
Interested in trying STEMCELL's organoid products? Fill out this form to request information about introductory offers.
Overview
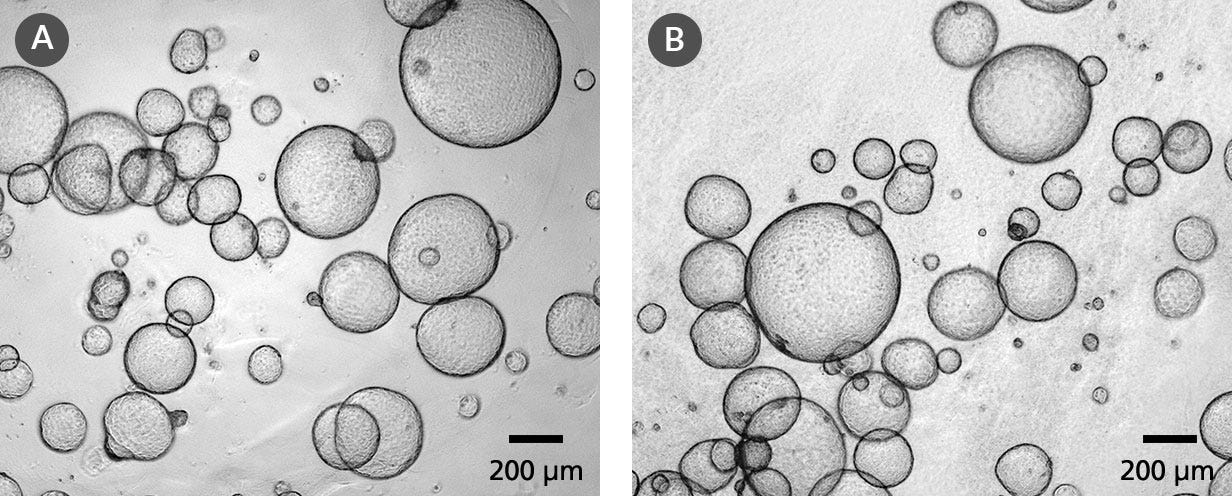
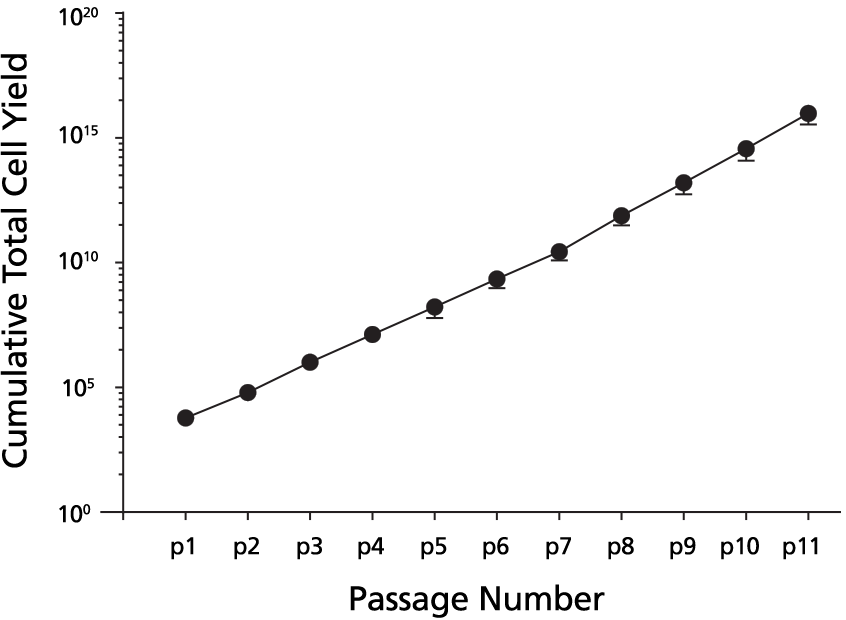
Data Figures
Protocols and Documentation
Find supporting information and directions for use in the Product Information Sheet or explore additional protocols below.
Applications
This product is designed for use in the following research area(s) as part of the highlighted workflow stage(s). Explore these workflows to learn more about the other products we offer to support each research area.
Resources and Publications
Educational Materials (15)
Publications (2)
The CellRaft AIR? system: A novel system enabling organoid imaging, identification, and isolation
SLAS Discovery 2022 4
Abstract
Three-dimensional (3D) culture systems have been developed that can re-capitulate organ level responses, simulate compound diffusion through complex structures, and assess cellular heterogeneity of tissues, making them attractive models for advanced in vitro research and discovery. Organoids are a unique subtype of 3D cell culture that are grown from stem cells, are self-organizing, and closely replicate in vivo pathophysiology. Organoids have been used to understand tissue development, model diseases, test drug sensitivity and toxicity, and advance regenerative medicine. However, traditional organoid culture methods are inadequate because they are low throughput and ill-suited for single organoid imaging, phenotypic assessment, and isolation from heterogenous organoid populations. To address these bottlenecks, we have adapted our tissue culture consumable and instrumentation to enable automated imaging, identification, and isolation of individual organoids. Organoids grown on the 3D CytoSort? Array can be reliably tracked, imaged, and phenotypically analyzed using brightfield and fluorescent microscopy as they grow over time, then released and transferred fully intact for use in downstream applications. Using mouse hepatic and pancreatic organoids, we have demonstrated the use of this technology for single-organoid imaging, clonal organoid generation, parent organoid subcloning, and single-organoid RNA extraction for downstream gene expression or transcriptomic analysis. The results validate the ability of the CellRaft AIR? System to facilitate efficient, user-friendly, and automated workflows broadly applicable to organoid research by overcoming several pain points: 1) single organoid time-course imaging and phenotypic assessment, 2) establishment of single cell-derived organoids, and 3) isolation and retrieval of single organoids for downstream applications.
A Multiscale Map of the Stem Cell State in Pancreatic Adenocarcinoma.
Cell 2019
Abstract
Drug resistance and relapse remain key challenges in pancreatic cancer. Here, we have used RNA sequencing (RNA-seq), chromatin immunoprecipitation (ChIP)-seq, and genome-wide CRISPR analysis to map the molecular dependencies of pancreatic cancer stem cells, highly therapy-resistant cells that preferentially drive tumorigenesis and progression. This integrated genomic approach revealed an unexpected utilization of immuno-regulatory signals by pancreatic cancer epithelial cells. In particular, the nuclear hormone receptor retinoic-acid-receptor-related orphan receptor gamma (ROR$\gamma$), known to drive inflammation and T cell differentiation, was upregulated during pancreatic cancer progression, and its genetic or pharmacologic inhibition led to a striking defect in pancreatic cancer growth and a marked improvement in survival. Further, a large-scale retrospective analysis in patients revealed that ROR$\gamma$ expression may predict pancreatic cancer aggressiveness, as it positively correlated with advanced disease and metastasis. Collectively, these data identify an orthogonal co-option of immuno-regulatory signals by pancreatic cancer stem cells, suggesting that autoimmune drugs should be evaluated as novel treatment strategies for pancreatic cancer patients.
Interested in trying STEMCELL's organoid products? Fill out this form to request information about introductory offers.
Legal Statement:
This product was developed under a license to intellectual property owned by Hubrecht Organoid Technology (HUB). This product is sold for research use only. Purchase of this product does not include the right to use it for drug screening aiming for commercial gain, equipment validation, biobanking, or for other commercial purposes. Purchasers wishing to use the product for purposes other than basic research use should contact HUB at www.huborganoids.nl to obtain a further license. Purchasers may apply for a License from HUB, which will not be unreasonably withheld by HUB.
This product was developed under a license to intellectual property owned by Hubrecht Organoid Technology (HUB). This product is sold for research use only. Purchase of this product does not include the right to use it for drug screening aiming for commercial gain, equipment validation, biobanking, or for other commercial purposes. Purchasers wishing to use the product for purposes other than basic research use should contact HUB at www.huborganoids.nl to obtain a further license. Purchasers may apply for a License from HUB, which will not be unreasonably withheld by HUB.
Quality Statement:
PRODUCTS ARE FOR RESEARCH USE ONLY AND NOT INTENDED FOR HUMAN OR ANIMAL DIAGNOSTIC OR THERAPEUTIC USES UNLESS OTHERWISE STATED. FOR ADDITIONAL INFORMATION ON QUALITY AT STEMCELL, REFER TO WWW.STEMCELL.COM/COMPLIANCE.
PRODUCTS ARE FOR RESEARCH USE ONLY AND NOT INTENDED FOR HUMAN OR ANIMAL DIAGNOSTIC OR THERAPEUTIC USES UNLESS OTHERWISE STATED. FOR ADDITIONAL INFORMATION ON QUALITY AT STEMCELL, REFER TO WWW.STEMCELL.COM/COMPLIANCE.