Forskolin-Induced Swelling of Human Intestinal Organoids Grown in IntestiCult™
- Document # 27179
- Version 1.0.1
- Feb 2020
Below we describe a protocol for forskolin-induced swelling of human intestinal organoids derived from healthy individuals and cultured in IntestiCult™ Organoid Growth Medium (Human) (Catalog #06010). For complete culturing instructions, use this document in conjunction with the relevant Product Information Sheet (Document #DX21423).
Introduction
Cystic fibrosis (CF) is a genetic disease caused by defects in the gene encoding the cystic fibrosis transmembrane conductance regulator (CFTR), a chloride anion channel expressed in mulitple epithelial tissues, including the intestinal epithelium. Defects in CFTR cause aberrant ion transport and mucus build-up in various organs, leading to difficulty breathing, recurring lung infections, and digestive issues. The recent characterization of intestinal organoid cultures has provided a new tool for studying CFTR function in vitro.1 Intestinal organoids can be efficiently established from colorectal biopsies, expanded, and maintained in long-term in vitro culture. These organoids maintain the genotype and phenotype of the parental tissue and retain the expected CFTR function when cultured in vitro.
Exposing intestinal organoids to forskolin causes the cells to rapidly increase their levels of cyclic adenosine monophosphate (cAMP), resulting in the opening of the CFTR channel.1,2 As chloride ions move through the channel and into the lumen, the organoids increase in size due to water following by osmosis, allowing for the assay of CFTR activity through organoid swelling. Because the swelling is CFTR-dependent, wild-type organoids increase in size while those carrying a CFTR mutation, or treated with a CFTR inhibitor, do not.
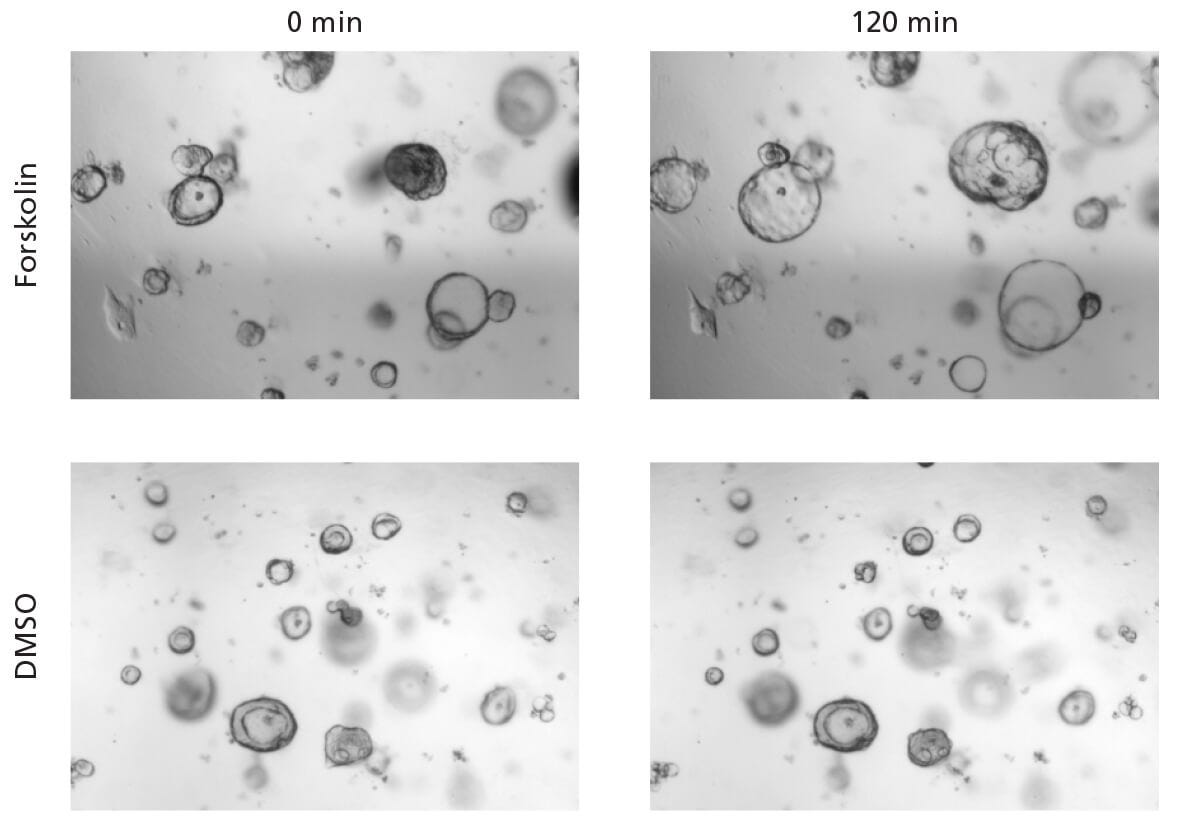
Figure 1. Forskolin-Induced Swelling of Intestinal Organoids Cultured in IntestiCult™ Organoid Growth Medium (Human)
The lumen of intestinal organoids became enlarged in response to treatment with 10 μM of forskolin after 120 minutes. This increase in size was not observed in intestinal organoids treated with a DMSO control.
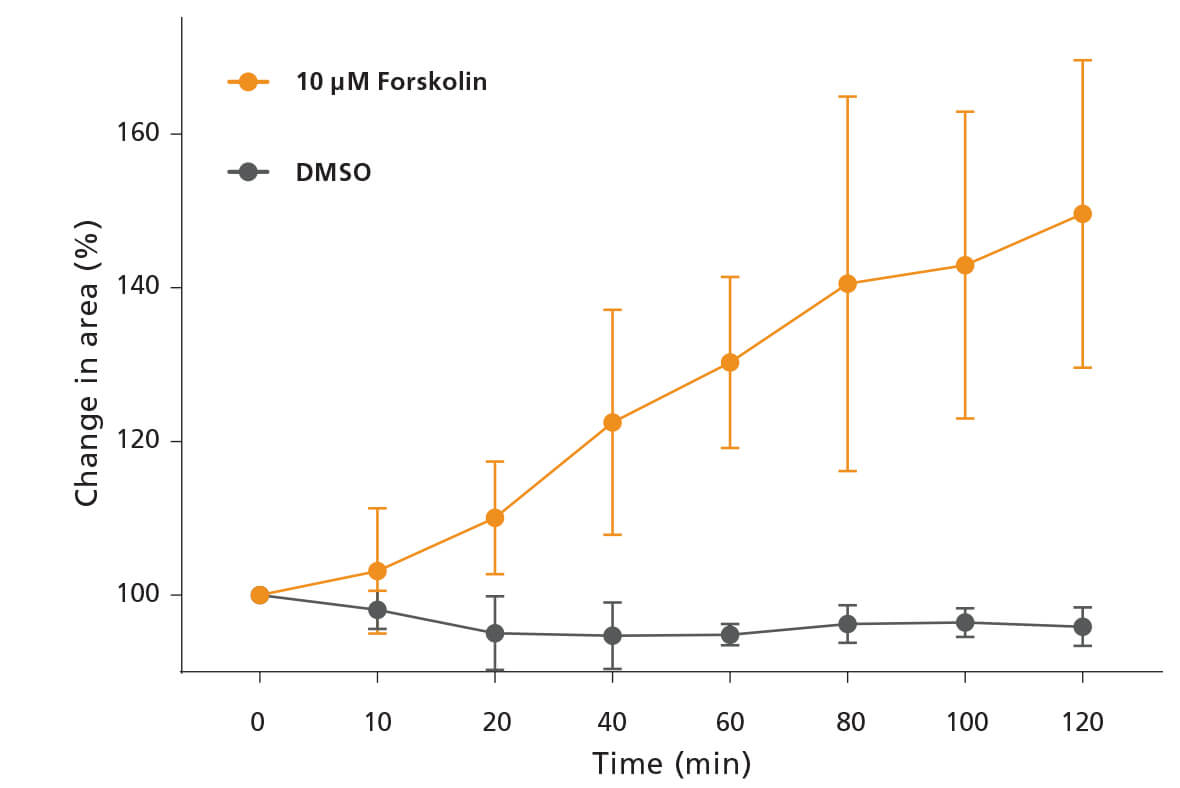
Figure 2. Quantification of Forskolin-Induced Swelling of Intestinal Organoids
The swelling of intestinal organoids was quantified by brightfield microscopy. The percentage change in area at later time points was calculated relative to the average area of the organoids observed at time = 0. At each time point, the whole well was imaged and the average from three technical replicates was calculated. Data is representative of one normal colonic donor at passage 3.
Protocol
Materials
- Establish organoid cultures as described in the Product Information Sheet (Document #DX21423). Grow organoids for 3 - 5 days after passage in IntestiCult™ Organoid Growth Medium (Human) (Catalog #06010) prior to assessing forskolin-induced swelling.
-
Remove media from each well and replace with an equal volume of Krebs-Ringer Bicarbonate (KBR) buffer. Allow the buffer to equilibrate
with the Corning®Matrigel® domes for 30 minutes at 37°C.
NOTE: Forskolin-induced swelling can be performed and assayed in the culture medium, however, swelling is more pronounced if using KBR buffer. - OPTIONAL: If using CFTR inhibitors as an additional control (e.g. CFTRINH172 or GlyH-101), add the appropriate amount of inhibitor to each well as necessary (50 μM for CFTRINH172 or GlyH-101) and incubate for 3 hours at 37°C. If inhibitors are not being used, proceed directly to step 4.
- Capture time = 0 images before proceeding as organoid swelling may begin immediately after adding forskolin.
- Remove the buffer from all wells being assayed and replace with fresh buffer, inhibitors (if applicable), and forskolin to a final concentration of 10 μM forskolin.
- Image each well at regular intervals until the swelling in uninhibited wells ceases (usually 80 - 120 minutes).
- To quantify forskolin-induced swelling, brightfield images at each time point are analyzed using ImageJ for average organoid area compared to the time = 0 images.
Related Products
Organoid E-Book
Download a copy of "Organoid Research Techniques: Evolution and Applications", the organoid e-book developed in collaboration with Wiley Publishing. Get an overview of the development of organoid technologies, how they are used in practice, and considerations for the future development of organoids.
References
- Dekkers, JF et. al (2013) A functional CFTR assay using primary cystic fibrosis intestinal organoids. Nat. Med. 19(7): 939–47.
- Boj, SF et. al (2017) Forskolin-induced swelling in intestinal organoids: An in vitro assay for assessing drug response in cystic fibrosis patients. Journal of Visualized Experiments. 120(e55159).
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration


