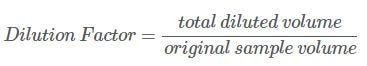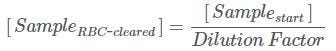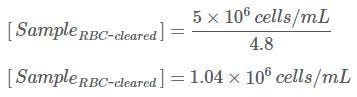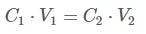How to Calculate Cell Concentration for Plating CFU Assays Following RBC Clearance with HetaSep™
How to calculate the cell concentration after a blood sample has been RBC-cleared with HetaSep™, and how to determine the volume of your RBC-cleared sample to achieve your desired plating density for a CFU assay
When preparing small volume samples of fresh blood for colony-forming unit (CFU) assays, red blood cells (RBCs) can be removed from your sample prior to plating via sedimentation. Such RBC-cleared samples may be subsequently used for CFU assays to assess the proliferation and differentiation of hematopoietic stem and progenitor cells. The protocol and formulas below describe how to calculate the cell concentration after a blood sample has been RBC-cleared with HetaSep™, and how to determine the volume of your RBC-cleared sample to achieve your desired plating density for a CFU assay.
Materials
- 3% Acetic Acid with Methylene Blue (Catalog #07060)
- HetaSep™ (Catalog #07806)
- Dulbecco's Phosphate Buffered Saline with 2% Fetal Bovine Serum (Catalog #07905)
- Iscove's MDM with 2% FBS (Catalog #07700)
Protocol
Please refer to the following definitions for notations used in the calculations below:
- [ Sample ] = concentration of cells in the sample (i.e. cells/mL)
- C = concentration
- V = volume
- Perform a total nucleated cell count using 3% Acetic Acid with Methylene Blue to obtain the cell concentration of your starting sample. For more information, see this protocol for How to Count Cells with a Hemocytometer.
-
Calculate the Dilution Factor by dividing the total diluted sample volume by the original sample volume using this dilution factor formula:
Example: If you are following this small-volume HetaSep™ protocol, start with a blood sample, and add 150 µL of Phosphate Buffered Saline (PBS) with 2% Fetal Bovine Serum (FBS) and 40 µL of HetaSep™. In this case, you would calculate the Dilution Factor as follows:
-
Calculate the nucleated cell concentration of your sample after RBC clearance using HetaSep™ ([SampleRBC-cleared]). This is calculated by dividing the cell concentration of your starting sample by the Dilution Factor. See the formula below:
Example: If the concentration of your starting sample was 5.0 x 106 cells/mL, and your Dilution Factor is 4.8, then the concentration of the sample after RBC clearance using HetaSep™ will be:
-
You are now ready to prepare your sample for the CFU assay using MethoCult™ medium. As per the standard protocol for CFU assays, prepare a 10X concentration of the desired plating density for dilution in the MethoCult™ medium. Use the RBC-cleared sample to prepare the 10X concentration, using the following formula:
Example: If the required plating density for your sample is 1 x 104 cells per 35 mm culture dish, then you will need to prepare a 10X concentration of 1 x 105 cells/mL as follows:
Therefore, you will need to add 96 µL of the RBC-cleared sample to 904 µL of IMDM with 2% FBS, to prepare the desired 10X concentrated sample for your CFU assay.
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration