PBS-MINI MagDrive Bioreactor
Compact bioreactor with single-use vessels for high-throughput, 3D suspension culture of hPSCs and other cell types
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration
Overview
PBS-MINI 0.1 L and 0.5 L MAG Single-Use Vessels are sold separately in packs of 4. These disposable vessels are animal component-free and meet the requirements for USP Class VI Testing for Plastics <88> and ISO 10993. PBS Biotech Inc. provides an 18-month warranty for the PBS-MINI MagDrive Bioreactor Base Unit, valid from date of shipment. This product is for Research Use Only.
The PBS-MINI MagDrive Bioreactor Base Unit includes a power adapter that is compatible with Type A electrical outlets and 120V or 240V A/C inputs. A separate wall adaptor is needed for use outside North America; to request a region-specific power cord, please contact techsupport@stemcell.com.
Data Figures
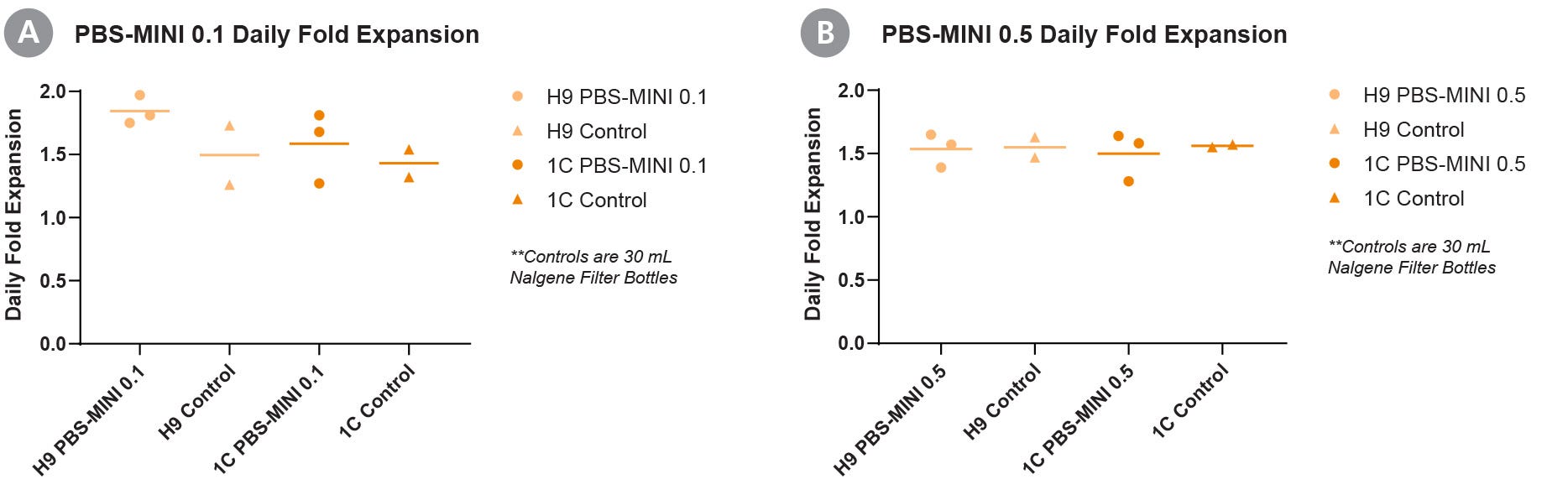
Figure 1. Consistently High Daily Fold Expansion of Human Pluripotent Stem Cells (hPSCs) Is Achieved with the PBS-MINI 0.1 and 0.5 MAG Single-Use Vessels
Daily fold expansions of a human embryonic stem (ES) cell line (H9) and human induced pluripotent stem (iPS) cell line (WLS-1C), cultured in PBS-MINI (A) 0.1 and (B) 0.5 MAG Single-Use Vessels, show consistently high growth. The cells were seeded at a density of 8 x 104 viable cells/mL in 100 mL (for PBS-MINI 0.1 MAG) and 3 - 8 x 104 viable cells/mL in 500 mL (for PBS-MINI 0.5 MAG), and cultured for 4 days using mTeSR™3D (Catalog #03950). Fold expansion of each cell line was comparable to their growth in Nalgene™ filter bottles (Control), when seeded at a density of 1 x 105 viable cells/mL in 30 mL. Error bars are +/- SD (n = 3 for PBS-MINI 0.1 and 0.5 conditions, and n = 2 for control).
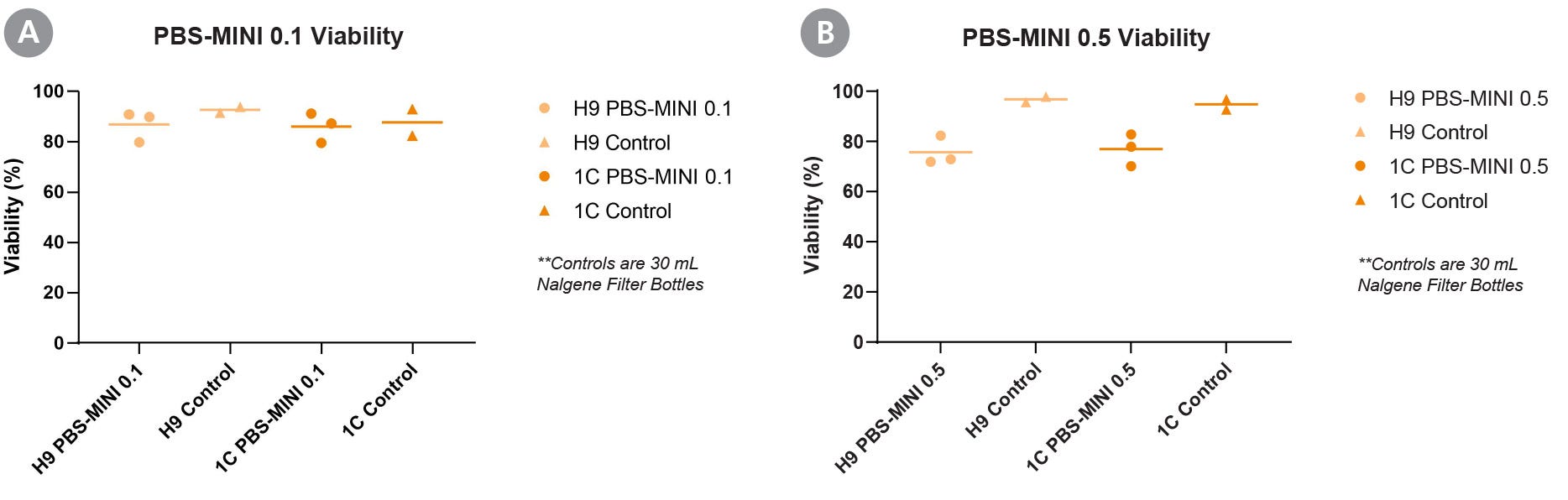
Figure 2. Culturing hPSCs in the PBS-MINI 0.1 and 0.5 MAG Single-Use Vessels Achieves High Cell Viability
End-of-passage viabilities of a human ES cell line (H9) and human iPS cell line (WLS-1C) cultured in mTeSR™3D show that the PBS-MINI 0.1 and 0.5 MAG Single-Use Vessels support expansion of hPSCs at (A) 100 mL (with PBS-MINI 0.1 MAG) and (B) 500 mL (with PBS-MINI 0.5 MAG) culture volumes. Error bars are +/- SD (n = 3 for PBS-MINI 0.1 and 0.5 conditions, and n = 2 for control).
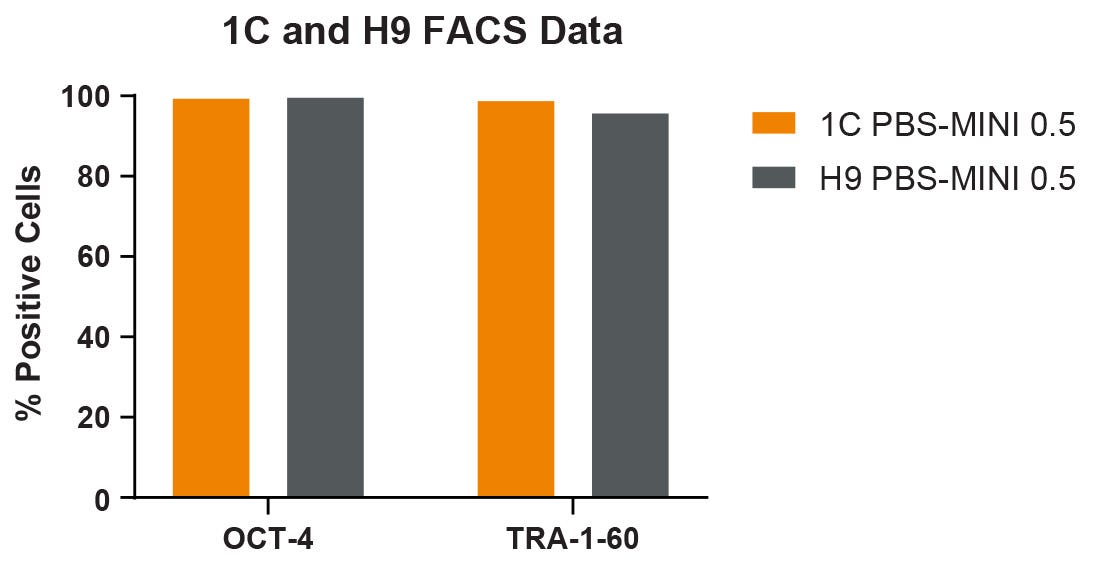
Figure 3. Markers of Undifferentiated hPSCs Harvested from the PBS-MINI 0.5 MAG Single-Use Vessels
1C and H9 hPSC lines after 9 passages in the PBS-MINI 0.5 MAG Single-Use Vessels using mTeSR™3D show high OCT-4 and TRA-1-60 expression. Error bars are +/- SD (n = 1).
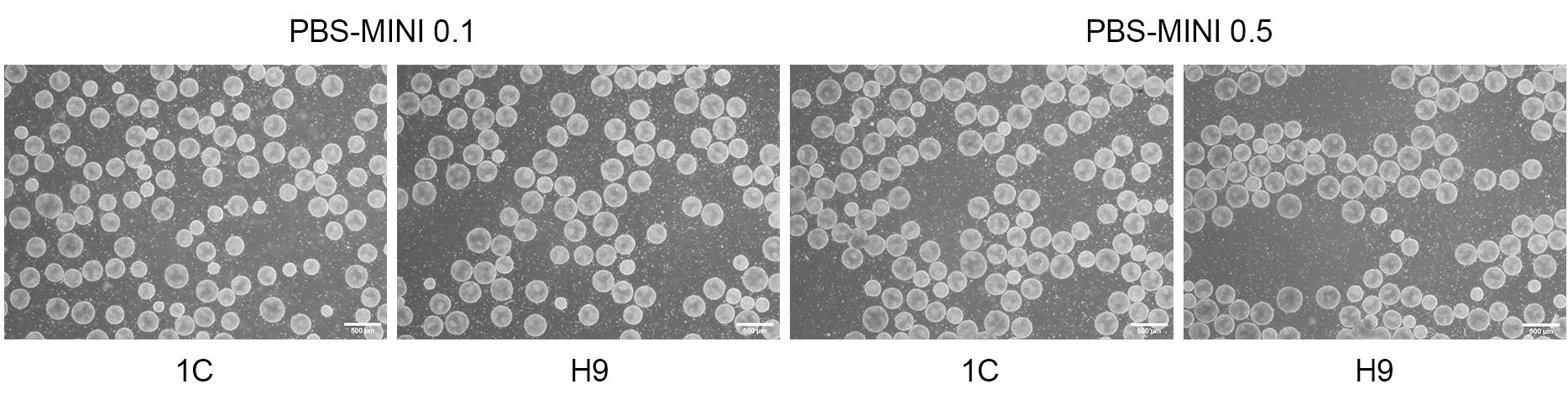
Figure 4. Morphology of hPSC Aggregates Is Optimal When Using the PBS-MINI 0.1 and 0.5 MAG Single-Use Vessels
hPSCs, expanded in suspension using mTeSR™3D in the PBS-MINI 0.1 and 0.5 MAG Single-Use Vessels, exhibit the typical morphological characteristics of hPSC aggregates in suspension culture, including a mostly spherical shape, clear edges (not smooth or shiny), an even color, and a dimpled or pock-marked appearance. Aggregates are typically 350-400 µm in size. Scale bar = 500 µm.
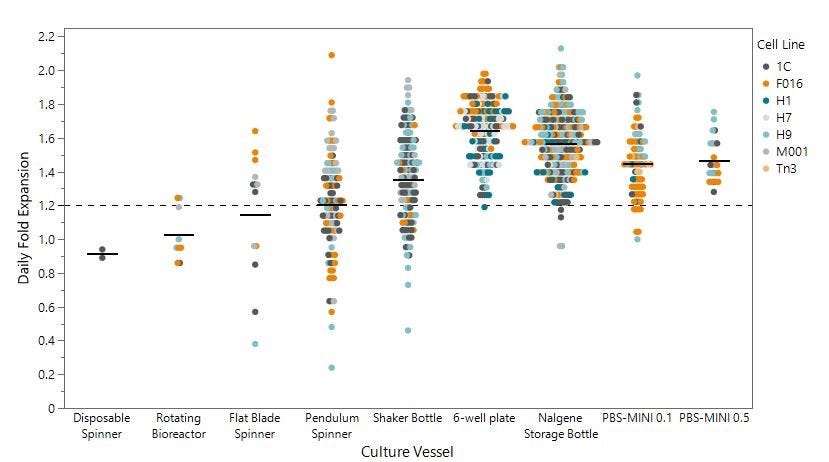
Figure 5. PBS-MINI 0.1 and 0.5 MAG Single-Use Vessels Demonstrate Consistently High Daily Fold Expansion Compared to Common Alternative Culture Vessels
Daily fold expansion of 7 cell lines in suspension culture vessels, tested from 2019 - 2022. Of the 9 vessels tested, those recommended for robust suspension culture scale-up are the Non Tissue Culture-Treated 6-Well Plate, Nalgene™ Filter Storage Bottle, PBS-MINI 0.1, and PBS-MINI 0.5. These vessels have a consistent mean daily fold expansion above 1.2.
Protocols and Documentation
Find supporting information and directions for use in the Product Information Sheet or explore additional protocols below.
Applications
This product is designed for use in the following research area(s) as part of the highlighted workflow stage(s). Explore these workflows to learn more about the other products we offer to support each research area.
Resources and Publications
Educational Materials (17)
Related Products
-
 mTeSR™3D
mTeSR™3DSerum-free media for suspension culture of human ES and iPS cells
-
 PBS-MINI Single-Use Vessel with Bottom Port
PBS-MINI Single-Use Vessel with Bottom PortSingle-use vessels with bottom port
-
 Extended 4-Gang Power Distribution Cable
Extended 4-Gang Power Distribution CableFour-way split cable to connect up to four PBS-MINI MagDrive Bioreactor Base Units to one power source
Item added to your cart

PBS-MINI MagDrive Bioreactor
PRODUCTS ARE FOR RESEARCH USE ONLY AND NOT INTENDED FOR HUMAN OR ANIMAL DIAGNOSTIC OR THERAPEUTIC USES UNLESS OTHERWISE STATED. FOR ADDITIONAL INFORMATION ON QUALITY AT STEMCELL, REFER TO WWW.STEMCELL.COM/COMPLIANCE.


















