ReproRNA™-OKSGM
A non-integrating, self-replicating RNA reprogramming vector for generating iPS cells
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration
-
 Trypsin-EDTA (0.25%)
Trypsin-EDTA (0.25%)Enzymatic cell dissociation reagent
-
 DMEM with 1000 mg/L D-Glucose
DMEM with 1000 mg/L D-GlucoseDulbecco's Modified Eagle's Medium (DMEM) with 1000 mg/L D-glucose
-
 Puromycin
PuromycinAntibiotic; Protein synthesis inhibitor
-
Labeling Antibodies
Compatible antibodies for purity assessment of isolated cells
Overview
Data Figures
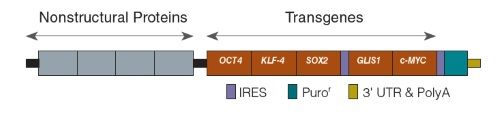
Figure 1. Schematic of ReproRNA™-OKSGM, a Single‑Stranded RNA Replicon Vector
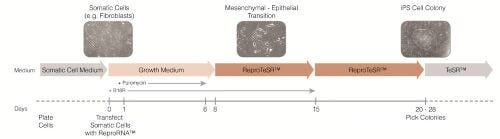
Figure 2. Schematic of Reprogramming Timeline with ReproRNA™-OKSGM
Somatic cells are transfected with ReproRNA™-OKSGM at day 0, and cultured in Advanced DMEM (AdvDMEM) with puromycin. After 6 days of puromycin selection post-transfection, cells are cultured in ReproTeSR™ for the remainder of the reprogramming induction phase until iPS cell colonies emerge. B18R recombinant protein is also added during the first 2 weeks after transfection to inhibit the interferon response and increase cell viability. Typically, by day 20, iPS cell colonies are large enough to be isolated and propagated in TeSR™ media. *TeSR™ = TeSR™ family media (mTeSR™1, TeSR™2, TeSR™-E8™).
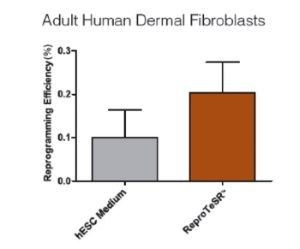
Figure 3. ReproRNA™-OKSGM Vector Efficiently Reprograms Fibroblasts
Dermal fibroblasts were transfected with the ReproRNA™-OKSGM vector and reprogrammed under feeder-dependent (standard KOSR-containing hES cell medium on irradiated mouse embryonic fibroblasts (iMEFs)) or feeder-independent conditions (ReproTeSR™ on Corning® Matrigel®). Fibroblasts (passage 4) were reprogrammed with average efficiencies of 0.10 ± 0.03% (hES cell medium) and 0.20 ± 0.01% (ReproTeSR™). Reprogramming efficiency of fibroblasts with ReproRNA™ and ReproTeSR™ is comparable to that reported with Sendai virus.¹ (n ≥ 6; Data shown are mean ± SEM).
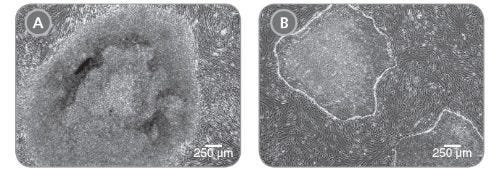
Figure 4. Feeder-Free Reprogramming with ReproRNA™-OKSGM Vector and ReproTeSR™ Generates iPS Cell Colonies with Superior Colony Morphology
Representative images of iPS cell colonies were generated using ReproRNA™‑OKSGM and cultured in (A) standard hES cell medium on irradiated mouse embryonic fibroblasts (iMEFs) or (B) ReproTeSR™ on Corning® Matrigel®. iPS cell colonies derived using ReproTeSR™ exhibit more defined borders, compact morphology, and reduced differentiation as compared to the ES cell medium.
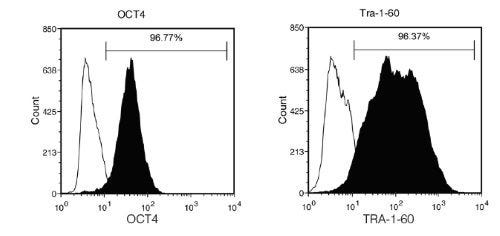
Figure 5. Human iPS Cells Generated with ReproRNA™-OKSGM Express Undifferentiated Cell Markers
Human iPS cells generated with ReproRNA™-OKSGM display high expression of undifferentiated cell markers (OCT4 and TRA-1-60) as shown by flow cytometry analysis after 12 passages in mTeSR™1. (Filled histogram = sample, hollow histogram = secondary antibody only).
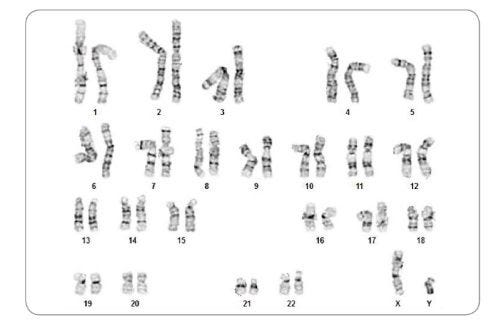
Figure 6. iPS Cells Derived Using ReproRNA™-OKSGM Display a Normal Karyotype
Karyogram of iPS cells derived with ReproRNA™-OKSGM and cultured in mTeSR™1 for 8 passages shows that a normal karyotype is retained.
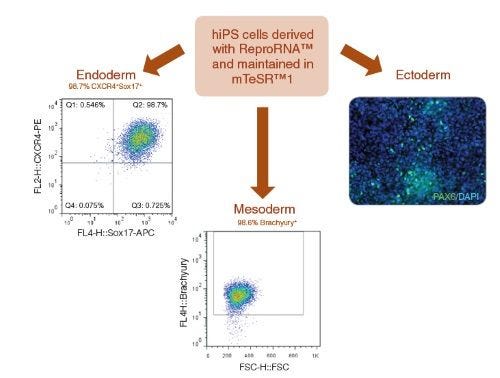
Figure 7. ReproRNA™-OKSGM Derived iPS Cells Have the Capacity to Differentiate to Cells of the Three Germ Layers
Human iPS cells derived with ReproRNA™-OKSGM and maintained in mTeSR™1 for 7 passages were differentiated into cells of the three germ layers. Endoderm specification was achieved using the STEMdiff™ Definitive Endoderm Kit and flow cytometry analysis shows a high percentage of cells (98.7%) positive for endoderm markers (CXCR4+SOX17+). Mesoderm induction was achieved with STEMdiff™ Mesoderm Induction Medium as shown by the high percentage of cells (98.6%) expressing Brachyury (T). Ectoderm specification was demonstrated using STEMdiff™ Neural Induction Medium. CNS-enriched NPC cultures expressing PAX6 (green) and stained with DAPI (blue) are shown.
Protocols and Documentation
Find supporting information and directions for use in the Product Information Sheet or explore additional protocols below.
Applications
This product is designed for use in the following research area(s) as part of the highlighted workflow stage(s). Explore these workflows to learn more about the other products we offer to support each research area.
Resources and Publications
Educational Materials (5)
Related Products
-
 Vitronectin XF™
Vitronectin XF™Defined, xeno-free matrix that supports the growth and differentiation of human pluripotent stem cells under serum-free, feeder-free conditions
-
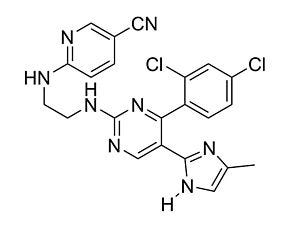 CHIR99021
CHIR99021WNT pathway activator; Inhibits GSK3
-
 Thiazovivin
ThiazovivinRHO/ROCK pathway inhibitor; Inhibits ROCK
Item added to your cart

ReproRNA™-OKSGM
PRODUCTS ARE FOR RESEARCH USE ONLY AND NOT INTENDED FOR HUMAN OR ANIMAL DIAGNOSTIC OR THERAPEUTIC USES UNLESS OTHERWISE STATED. FOR ADDITIONAL INFORMATION ON QUALITY AT STEMCELL, REFER TO WWW.STEMCELL.COM/COMPLIANCE.