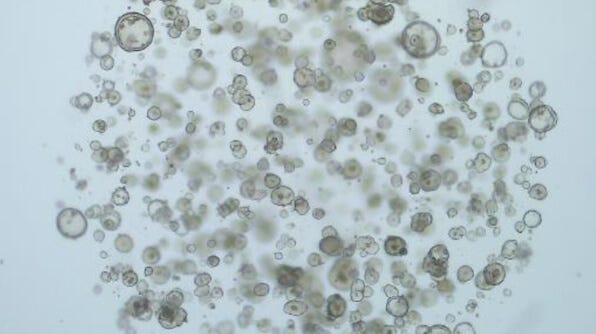
Culturing Intestinal Cancer-Derived Organoids
Cancer is one of the leading causes of death worldwide, with colorectal cancer representing one of the most common cancer types. Although cancer cell lines and animal models have revealed much information about intestinal cancers, cancer-derived intestinal organoids have been demonstrated to more faithfully recapitulate the tissue architecture, cellular heterogeneity, and morphology of the originating tumor.1 Cancer-derived organoids are consequently proving to be useful experimental models for investigating cancer biology, including disease progression and the affected signaling pathways and niche requirements of the tumor.2 Cancer-derived organoids have also enabled more translational applications, such as activating and expanding tumor-reactive T cell populations, predicting patient-specific treatment outcomes, and screening potential therapeutics.3,4
In this protocol, developed in collaboration with Hubrecht Organoid Technology, we describe the culture of Wnt-independent cancer-derived organoids using IntestiCult™ OGM Human Basal Medium (Catalog #100-0190). For complete instructions, use this document in coordination with the Product Information Sheet.
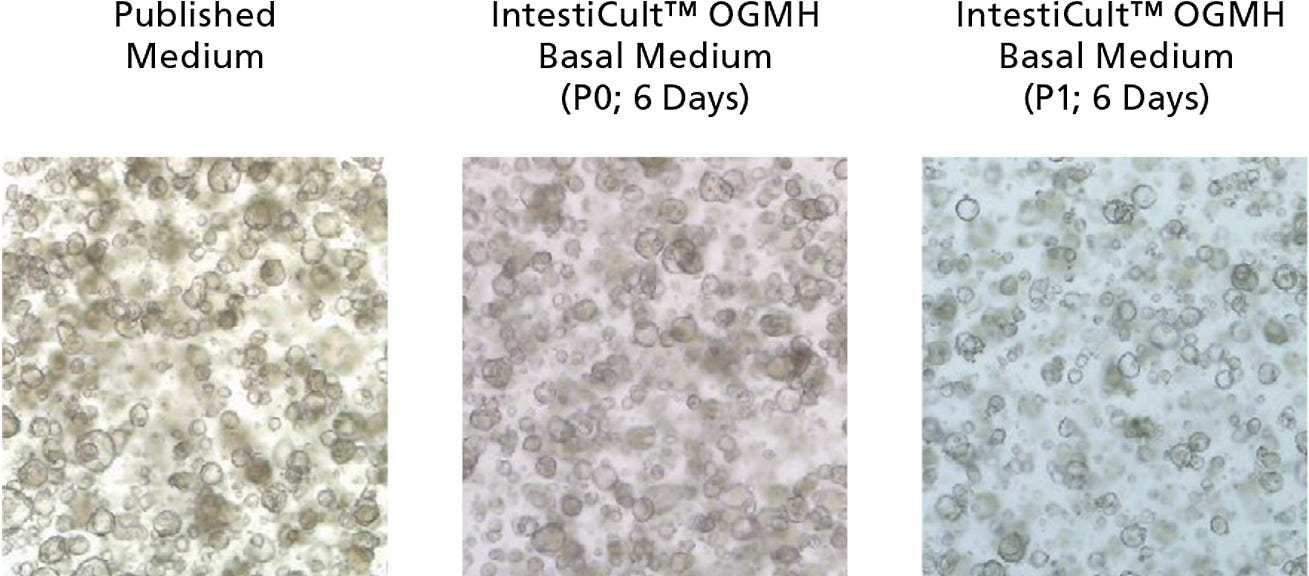
Figure 1. Growth of Cancer-Derived Organoids in IntestiCult™ OGMH
Organoids were established from colorectal cancer biopsies in published medium,1 then switched to IntestiCult™ OGMH (P0), as described below. Cancer-derived organoids demonstrated efficient growth both after establishment in IntestiCult™ OGMH, as well as after passaging. Data used with permission from Hubrecht Organoid Technology.
Materials
- IntestiCult™ OGM Human Basal Medium (Catalog #100-0190)
- DMEM/F-12 with 15 mM HEPES (Catalog #36254)
- 25% Bovine serum albumin (BSA) solution in water
- Corning® Matrigel® Matrix, Growth Factor Reduced (GFR), Phenol Red-Free (Corning® 356231)
- Gentle Cell Dissociation Reagent (Catalog #100-0485)
- D-PBS (Without Ca++ and Mg++) (Catalog #37350)
- Antibiotics (e.g. Gentamicin)
Preparation
- Thaw IntestiCult™ OGM Human Basal Medium at room temperature (15 - 25°C) or at 2 - 8°C overnight. Prepare 100 mL IntestiCult™ OGM Human Basal Medium + DMEM/F12 with 15 mM HEPES:
- 50 mL IntestiCult™ OGM Human Basal Medium
- 50 mL DMEM/F-12 with 15 mM HEPES
- Mix thoroughly.
- Add desired antibiotics immediately before use. (e.g. 50 μg/mL gentamicin)
Note: Use IntestiCult™ OGM Human Basal Medium immediately or aliquot and store at -20°C for up to 3 months. After thawing aliquots, use immediately. Do not re-freeze.
Note: If IntestiCult™ OGM Human Basal Medium + DMEM/F12 with 15 mM HEPES is not used immediately, store at 2 - 8°C for up to 1 week.
- Prepare 50 mL of serum-supplemented DMEM/F-12 + 15 mM HEPES (DMEM + BSA) by combining the following:
- 2 mL 25% BSA solution in water (1% final concentration)
- 48.0 mL DMEM/F-12 + 15 mM HEPES
- Mix thoroughly. Store on ice.
- Place D-PBS (without Ca++ and Mg++) on ice.
- Warm a tissue culture-treated 24-well plate in a 37°C incubator for at least 2 hours.
- Thaw 100 μL of Matrigel® on ice.
Note: This is sufficient for plating up to 4 culture domes.
Protocol
Note: This protocol is meant for use in coordination with the IntestiCult™ Product Information Sheet (PIS; Document #10000003510). For complete culture instructions, refer to Section B of the PIS for plating instructions, and Section C for passaging, as indicated below.
- In a 15 mL conical tube, wash the tissue sample with 10 mL of ice cold PBS. Allow the tissue to settle by gravity (~5 seconds) then aspirate the supernatant.
- Repeat step 1, leaving 1 mL of supernatant in the tube.
- Transfer the tissue and remaining supernatant to a 1.5 mL microcentrifuge tube using a 1 mL pipettor.
- Using sterile scissors, thoroughly mince the tissue into ~5 mm pieces. Transfer the tissue fragments to a new 15 mL conical tube using a 1 mL pipettor. Rinse the microcentrifuge tube with PBS and add the rinse to the tissue fragments.
- Allow the tissue fragments to settle by gravity (~5 seconds) then aspirate the supernatant.
- Add 10 mL of Gentle Cell Dissociation Reagent. Incubate at 37°C on a rocking platform set at medium speed (~40 rpm) for 60 minutes.
- Centrifuge at 290 x g for 5 minutes. Aspirate the supernatant.
Note: For the remainder of the protocol, pre-wet pipette tips with DMEM + 1% BSA before manipulating the tissue sample. This prevents crypts from sticking to the wall of the pipette tip.
- Add 1 mL of ice-cold DMEM + 1% BSA. Vigorously pipette up and down 20 times with a 1 mL pipettor.
Note: Avoid touching the tube with the pipette tip.
- Using a 1 mL pipettor, pass the contents of the tube through a 70 μm cell strainer tilted on its side into a new 15 mL conical tube. Rinse the original tube with 1 mL of DMEM + 1% BSA and pass through the strainer into the tube.
Note: For complete instructions on plating isolated tissue as organoid cultures, proceed to section B, step 2 in the PIS (Document #10000003510). When plating organoids derived from cancerous tissue, use the medium prepared above instead of complete IntestiCult™ OGMH.
Note: Growth medium should be replaced every 2 days, and cultures can be passaged every 6 - 12 days. For complete passaging instructions, refer to section C in the PIS (Document #10000003510). When passaging organoids derived from cancerous tissue, use the medium prepared above instead of complete IntestiCult™ OGMH.
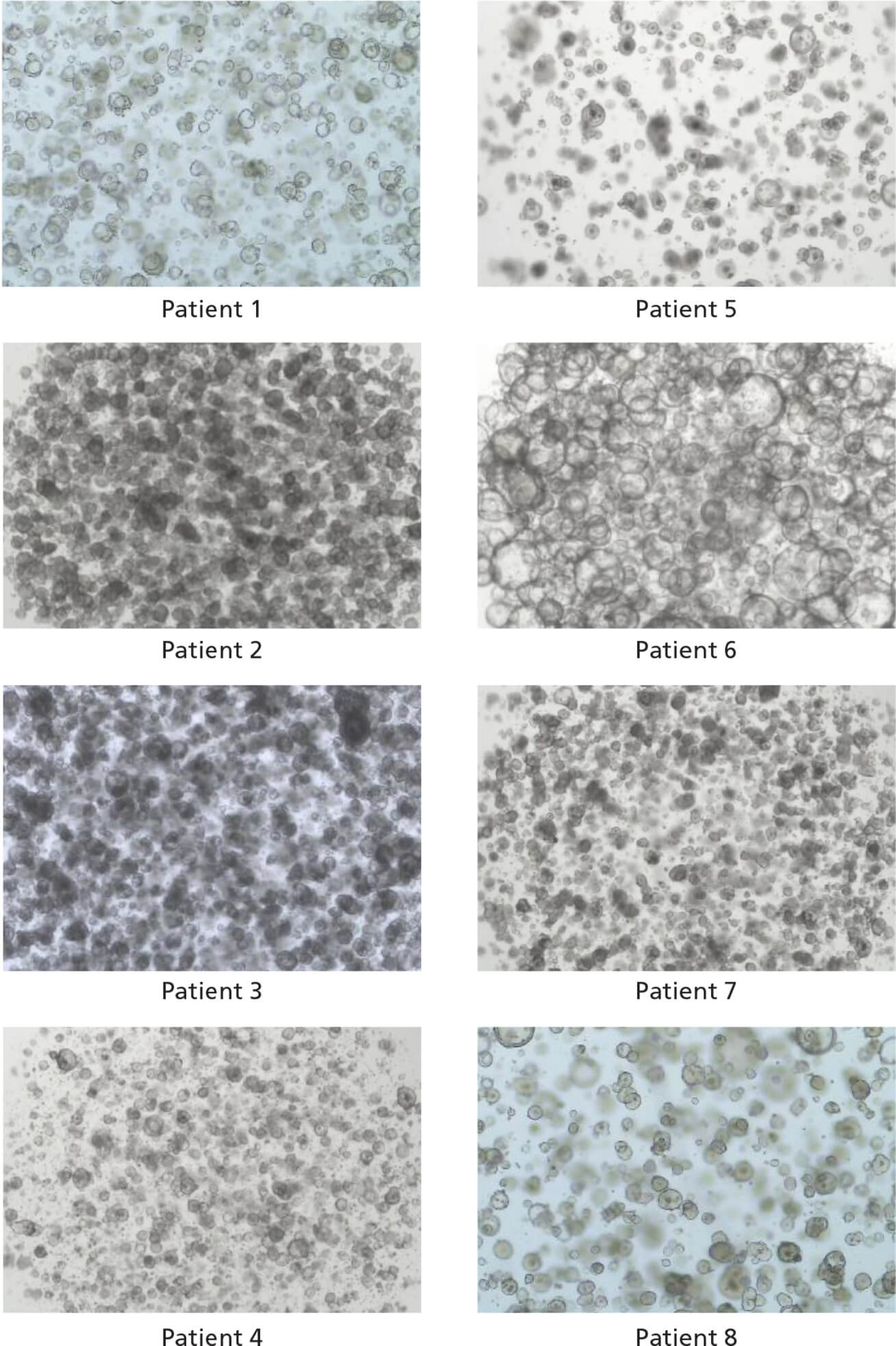
Figure 2. IntestiCult™ OGM Human Basal Medium Enables Organoid Growth Across Different Patients
Organoids were established in published medium1 from colorectal tumor biopsy samples, then switched to IntestiCult™ OGM Human Basal Medium after passaging. Organoids were passaged twice in IntestiCult™ OGMH Basal Medium and imaged at the end of the second passage (Day 6 - 12). Data used with permission from HUB Organoids.
References
- Sato T et al. (2011) Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterology. 141(5): 1762–72.
- Fujii M et al. (2016) A colorectal tumor organoid library demonstrates progressive loss of niche factor requirements during tumorigenesis. Cell Stem Cell. 18(6): 827–38.
- Dijkstra K et al. (2018) Generation of tumor-reactive T cells by co-culture of peripheral blood lymphocytes and tumor organoids. Cell. 174(6): 1586–98.
- Vlachogiannis G et al. (2018) Patient-derived organoids model treatment response of metastatic gastrointestinal cancers. Science. 359(6378): 920–26.
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration