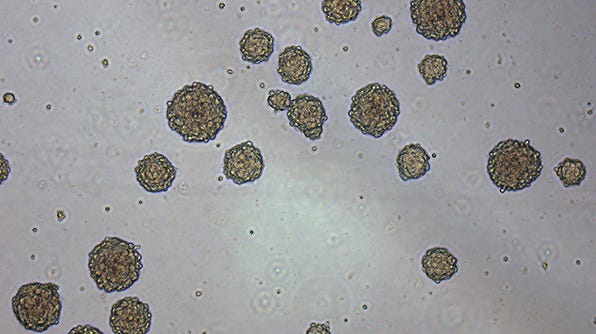
How to Cryopreserve Neurospheres
Bank neural stem and progenitor cells from patient samples or fresh animal tissue for downstream experiments on self-renewal, differentiation potential, or pathology
The option to cryopreserve neural stem and progenitor cells from central nervous system (CNS) tissue samples as neurospheres adds versatility to your experimental design and workflow. It introduces a pause point after obtaining rare patient samples or fresh animal tissue, so you can more conveniently schedule downstream assays of self-renewal, differentiation potential, or pathology. It also allows you to bank samples for future research and limit the number of passages the cells experience.
This protocol is for the cryopreservation of neurospheres using controlled rate freezing containers. The same freeze-thaw protocol can also be applied to finely chopped embryonic CNS tissue. Please refer to Gritti et al.1 for more information.
Materials
- Freezing container (e.g. 5100 Cryo 1˚C Freezing Container, “Mr. Frosty”; Nalgene Catalog #5100-0001)
- Isopropanol
- Dimethyl sulfoxide (DMSO) or CryoStor® CS10 (Catalog #07930)
- Complete culture medium (e.g. species-specific NeuroCult™ Proliferation Medium with appropriate supplements added)
- 2 mL cryogenic vials (e.g. Cryogenic Vials with Green Caps, Catalog #100-0093)
Protocol
- Neurospheres should be cryopreserved when they reach 100 - 150 µm in diameter. Do not allow the neurospheres to grow too large (> 200 µm in diameter) or they may develop necrotic cores.
- Do not mechanically dissociate neurospheres before freezing. This will increase the number of dead cells, significantly lowering the viability of the thawed culture.
- Neurosphere growth characteristics can vary between starting samples and species and the neurosphere density for cryopreservation may need to be optimized. In this protocol, we provide instructions for pooling neurospheres from 1 - 3 wells of a 6-well plate into one cryopreservation vial. For more wells or different cultureware, increase medium volumes accordingly.
Part I: Preparation
- Ensure that the freezing container is at room temperature (15 - 25˚C) and filled with isopropanol.
- Prepare the freezing medium:
- Option 1: Add DMSO to a final concentration of 10% (v/v) in the appropriate species-specific complete NeuroCult™ Proliferation Medium (i.e. containing rh EGF, rh bFGF, and Heparin as required).
- Option 2: CryoStor® CS10 may be used. Ensure that the CryoStor® CS10 is chilled at 4˚C until immediately before use.
- Label 2 mL cryogenic vials with name, date, passage number, and cell type.
Part II: Cryopreservation
- Collect the entire neurosphere suspension from 1-3 culture wells and transfer into a 15 mL tube. If neurospheres are attached to the bottom of the plate, tap the plate or shoot a stream of medium across the plate to detach them.
- Centrifuge at 110 x g for 10 minutes.
- Remove supernatant and wash the pellet once by gently pipetting in 1 mL of the appropriate species-specific complete NeuroCult™ Proliferation Medium.
- Centrifuge at 110 x g for 10 minutes.
- Remove supernatant and resuspend the neurosphere pellet gently in 1.5 mL freezing medium. Swirl gently to disperse the neurospheres.
- Transfer the entire neurosphere suspension into the pre-labeled cryovials.
- Transfer cryovials into the freezing container containing isopropanol.
- Leave the freezing container at -80˚C for a minimum of 4 hours (or overnight), to allow a slow and reproducible decrease in temperature (-1˚C/minute).
- Transfer cryovials into liquid nitrogen for long-term storage.
Part III: Thawing
- Warm the appropriate species-specific Complete NeuroCult™ Proliferation Medium in a water bath to 37˚C.
- Quickly transfer cryovial(s) from liquid nitrogen to 37˚C water bath and leave until thawed. Swirl the vial to promote thawing.
- Wipe the cryovial(s) thoroughly with 70% ethanol.
- Slowly transfer the neurosphere cell suspension from the cryovial to a 15 mL polypropylene tube containing 5 mL warm Complete NeuroCult™ Proliferation Medium.
- Centrifuge cell suspension at 110 x g for 8 minutes.
- Remove most of the supernatant, ensuring not to disturb the pellet.
- Resuspend the neurospheres by slowly pipetting up and down 2 - 3 times in warm complete NeuroCult™ Proliferation Medium and transfer the suspension to fresh tissue culture treated cultureware.
Notes:- Resuspend gently to ensure that the neurospheres remain intact.
- Neurosphere health and viability upon thaw may be variable and plating density may need to be optimized.
- Allow neurospheres to recover for 1-2 days before passaging.The neurospheres are ready to be passaged when the cells in the spheres are tightly packed together.
Reference
- Gritti A et al. (2001) Cultures of stem cells of the central nervous system, In: Federoff S. and Richardson A. (Eds.), Protocols for Neural Cell Culture (pp. 173–197). Totowa, NJ: Humana Press, Inc.
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration