StemSpan™ Medium and Supplements for the Generation of T Cells from Cord Blood-Derived CD34+ Cells
- Document # 27050
- Version 3.0.0
- Aug 2020
Background
Investigators studying hematopoiesis require standardized culture conditions to promote the proliferation and/or lineage-specific differentiation of hematopoietic stem and progenitor cells (HSPCs). Methods to differentiate and expand HSPCs into T cells offer a source of cells for research into this lineage. The in vitro differentiation of HSPCs to T cells has previously only been possible by co-culture of HSPCs with a stromal or “feeder” cell line that has been engineered to express Notch ligands. In such cultures, CD34+CD38-/lo HSPCs develop into CD7+CD5+ progenitor T (pro-T) cells that further differentiate to CD4 immature single-positive (CD4 ISP) cells. These cells give rise to CD4+CD8+ double-positive (DP) cells which finally mature into CD3+TCRαβ+ single-positive (SP) T cells that express either CD4 or CD8.
The StemSpan™ T Cell Generation Kit (Catalog #09940) facilitates the differentiation of cord blood (CB)-derived CD34+ cells into T cells without the use of a stromal cell line, and in serum-free culture conditions.
Media and Supplements for the Expansion and T Lineage Differentiation of Human HSPCs
The StemSpan™ T Cell Generation Kit contains serum-free medium, coating material, expansion and maturation supplements (see table below) required for the generation of pro-T cells and DP cells in a two-step protocol. In the first step, CD34+ HSPCs are cultured for 14 days in medium containing expansion supplement to promote their proliferation and differentiation into CD7+CD5+ pro-T cells. In the second step, pro-T cells generated during the first 14 days are cultured for another 4 weeks in medium containing the maturation supplement to promote their maturation into DP cells. In this system, more than 20,000 DP cells can be generated per input CD34+ cell (See Figures 3 and 4). Both fresh and cryopreserved CB-derived CD34+ cells may be used with the StemSpan™ T Cell Generation Kit. If pro-T cells are the desired cell type, only materials for the first step of the protocol are necessary and can be purchased individually.
Why Use StemSpan™ for T Cell Generation?
- Serum- and feeder-free conditions eliminate variation introduced by serum and stromal cell lines.
- Produce more than 20,000 DP cells per input CB-derived CD34+ cell.
- Eliminate extra passaging steps required for stromal cell-based cultures.
StemSpan™ T Cell Generation Kit
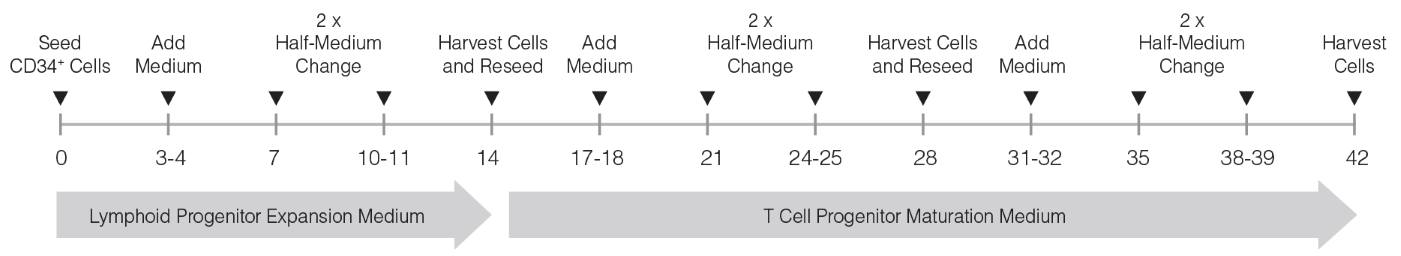
Figure 1. General StemSpan™ T Cell Generation Protocol
CB-derived CD34+ cells are seeded on day 0. Medium should be topped up after 3 - 4 days of culture followed by two half-medium changes every 3 - 4 days. On day 14, the CD7+CD5+ pro-T cells are harvested and can be reseeded if further maturation into DP cells is desired. Top-up and half-medium changes should be performed every 3 - 4 days after each harvest and reseed, as illustrated in the figure. For more information see protocol below.
Protocol
This protocol is designed to promote the proliferation and differentiation of CB-derived CD34+ cells into CD7+CD5+ pro-T cells, followed by their subsequent differentiation into CD4+CD8+ DP cells after 42 days of culture. See Figure 1 for a general representation of this protocol. An optional protocol extension for further maturation of DP cells into CD8 single-positive (SP) T cells is also illustrated in Figure 2.
Optimal cell yields depend on maintenance of proper cell health, which largely depends on following the recommended schedule of feeding and medium changes.
- Non-tissue culture treated plates must be coated with StemSpan™ Lymphoid Differentiation Coating Material prior to culture. Prepare coating material following instructions in the Product Information Sheet (PIS; Document #DX22213).
- Thaw frozen CB CD34+ cells (Catalog #70008), or isolate CD34+ cells separately from fresh CB using the EasySep™ Human Cord Blood CD34 Positive Selection Kit II (Catalog #17896).
- Prepare complete lymphoid progenitor expansion medium (StemSpan™ SFEM II + Lymphoid Progenitor Expansion Supplement). Refer to PIS for details.
- Dilute CD34+ cells to 1 x 104 cells/mL in complete lymphoid progenitor expansion medium and seed into the pre-coated plate.
- Incubate at 37°C for 14 days, following instructions in the PIS for necessary half-medium changes. Cells are harvested on day 14 and can be used for downstream applications if pro-T cells are desired (See Figure 3), or reseeded to further mature into DP cells.
- Before reseeding cells, prepare a freshly coated plate (See step 1).
- Prepare complete T cell progenitor maturation medium (StemSpan™ SFEM II + T Cell Progenitor Maturation Supplement).
- Dilute pro-T cells to 1 x 105 cells/mL in complete T cell progenitor maturation medium, seed into the coated plate and incubate at 37°C following instructions in the PIS for necessary half-medium changes and replating.
- On day 28, harvest and dilute cells to 5 x 105 cells/mL in complete T cell progenitor maturation medium.
- Reseed on a freshly coated plate and incubate at 37°C.
- Harvest cells containing CD4+CD8+ DP cells on day 42 (See Figure 4) for use in downstream assays, or follow the optional protocol extension for further maturation to CD8 SP T cells.
Optional Protocol Extension
An optional protocol is available to mature DP cells to CD8 SP T cells. This extended protocol uses complete T cell progenitor maturation medium made with reagents included in the StemSpan™ T Cell Generation Kit and must be combined with additional components including ImmunoCult™ CD3/CD28/CD2 T Cell Activator (Catalog #10970) and IL-15 (Catalog #78031).
- Prepare a freshly coated plate (See step 1 of Protocol).
- Prepare complete CD8 SP T cell maturation medium (StemSpan™ SFEM II + T Cell Progenitor Maturation Supplement + ImmunoCult™ T Cell Activator + IL-15). Refer to PIS for details.
- Dilute cells to 1 x 106 cells/mL in complete CD8 SP T cell maturation medium, seed into the coated plate and incubate at 37°C.
- After 3 - 4 days of culture, top up with CD8 SP T cell maturation medium, without ImmunoCult™ T Cell Activator.
- Incubate at 37°C and harvest cells on day 49 (See Figure 5).
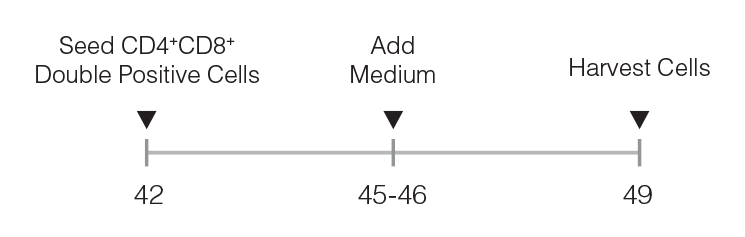
Figure 2. Optional CD8 SP T Cell Maturation Protocol
DP cells are seeded and medium is added after 3 - 4 days. CD8 SP T cells can be harvested on day 49.
Applications
- Research into the differentiation of T lineage cells from HSPCs
- Development of procedures to expand T cells from CD34+ cells in culture
- Assessment of efficacy and toxicity of candidate therapeutics on T cell differentiation during drug development
- Development of in vitro models to study diseases which involve T cell development
Cell Analysis
CD7+CD5+ pro-T cells are produced after 14 days of culture of CB-derived CD34+ cells (Figure 3). These cells differentiate further into CD4 immature single-positive (CD4 ISP) cells and CD4+CD8+ DP cells during a second 28-day culture step (Figure 4). By following the optional protocol extension, DP cells can finally mature into CD8 SP T cells (Figure 5). Cells may be stained with antibodies directed against cell surface markers CD3, CD4, CD5, CD7, CD8 and TCRαβ for analysis by flow cytometry. In the representative flow cytometry plots shown, dead cells were excluded by light scatter profile and viability staining.
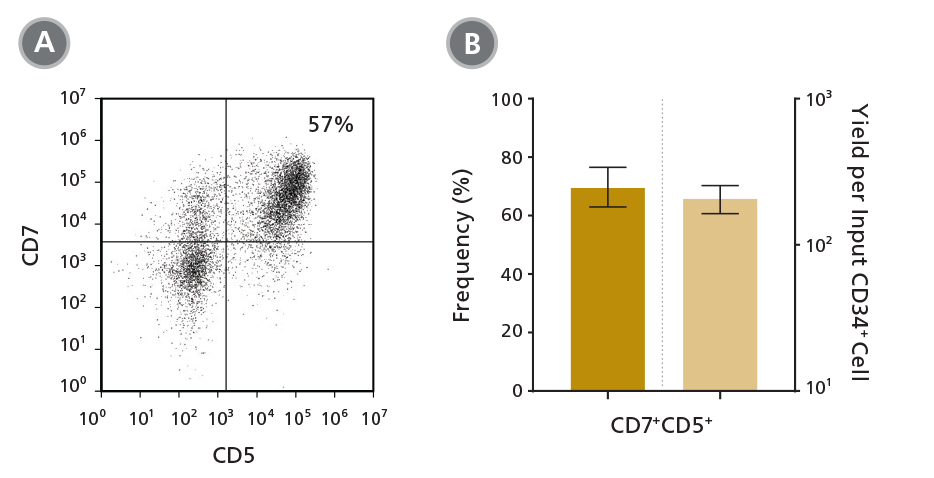
Figure 3. Frequency and Yield of CD7+CD5+ Pro-T Cells After 14 Days of Culture
CB-derived CD34+ cells (freshly isolated or frozen) were cultured for 14 days in StemSpan™ SFEM II containing Lymphoid Progenitor Expansion Supplement (Catalog #09915) on plates coated with Lymphoid Differentiation Coating Material (Catalog #09925). Cells were harvested and analyzed for CD7 and CD5 expression by (A) flow cytometry. The (B) average frequency of viable CD7+CD5+ pro-T cells on day 14 was 70%, with ~200 CD7+CD5+ cells produced per input CD34+ cell. Shown are means with 95% confidence intervals (n = 33).
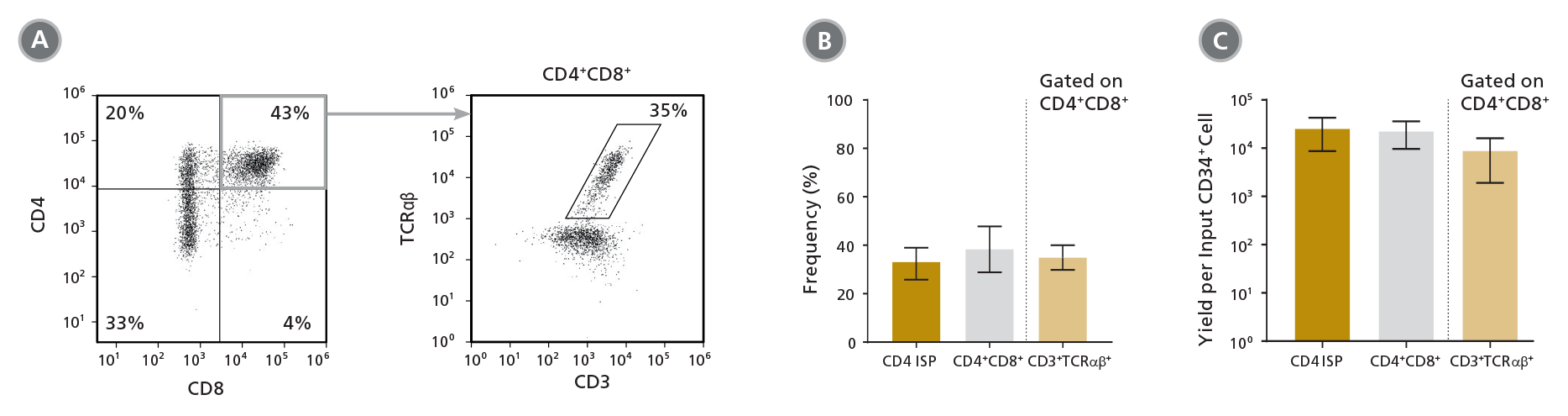
Figure 4. Frequency and Yield of CD4 ISP and CD4+CD8+ DP Cells After 42 Days of Culture
CB-derived CD34+ cells (freshly isolated or frozen) were cultured with the StemSpan™ T Cell Generation Kit (Catalog #09940) for 42 days and (A) analysed by flow cytometry for the expression of CD4, CD8, CD3 and TCRαβ. The (B) frequency and (C) yield of CD4 ISP, double-positive (CD4+CD8+) and CD3+TCRαβ+-expressing double-positive cells (CD4+CD8+CD3+TCRαβ+) are shown. On average, 38% of the total viable population were DP (CD4+CD8+), of which 35% co-expressed CD3 and TCRαβ. The yields of total DP cells and CD3+TCRαβ+ DP cells per input CD34+ cell were ~23,000 and ~9,000, respectively. Shown are means with 95% confidence intervals (n = 31).
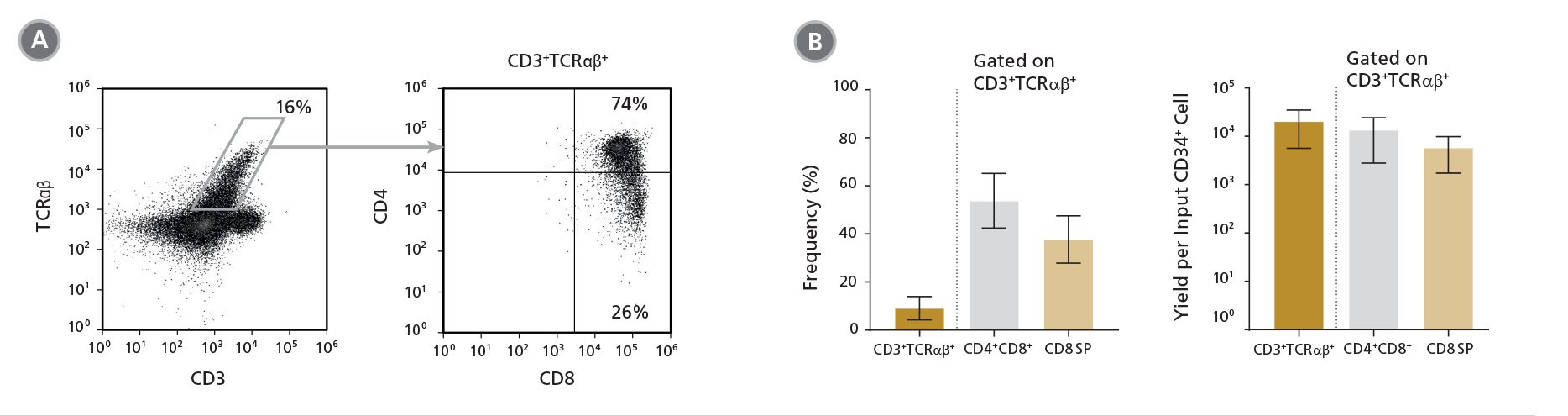
Figure 5. Frequency and Yield of CD8 SP T Cells After 49 Days of Culture
DP cells were further matured into CD8 SP T cells by culturing for an additional 7 days in StemSpan™ SFEM II with T Cell Progenitor Maturation Supplement (Catalog #09930), IL-15 (Catalog #78031) and ImmunoCult™ CD3/CD28/CD2 T Cell Activator (Catalog #10970) on coated plates. On day 49, cells were (A) analyzed by flow cytometry for the expression of CD3, TCRαβ, CD4 and CD8. The (B) frequency and yield of CD3+TCRαβ+-expressing cells and their subsets are shown. On average, 54% of the CD3+TCRαβ+ cells were DP (CD4+CD8+) and 38% were CD8 SP (CD4-CD8+). The average yield of CD8 SP T cells per input CD34+ cell was ~6,000. CD3+TCRαβ+ CD4 SP (CD4+CD8-) T cells were detected at very low frequencies (data not shown). Shown are means with 95% confidence intervals (n = 12).
Suggested Antibodies for Analysis*
*Not included in the kit
Accessory Products*
*Not included in the kit
Products Required for Optional Protocol Extension*
*Not included in the kit
** may also be used for activation
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration

