ImmunoCult™ NK Cell Expansion Kit
Kit for the culture and expansion of human NK cells
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration
-
 Trypan Blue
Trypan BlueReagent for counting viable mammalian cells
-
 D-PBS (Without Ca++ and Mg++)
D-PBS (Without Ca++ and Mg++)Dulbecco’s phosphate-buffered saline without calcium and magnesium
-
 Flat-Bottom Plates, Non-Treated
Flat-Bottom Plates, Non-TreatedSterile flat-bottom, non-treated multiwell plate with lid; 6-, 12-, 24-, 48-, or 96-well formats
-
Labeling Antibodies
Compatible antibodies for purity assessment of isolated cells
Overview
Use ImmunoCult™ NK Cell Expansion Kit to provide optimized culture conditions for expanding NK cells with high yields. This kit comprises ImmunoCult™ NK Cell Base Medium, ImmunoCult™ NK Cell Expansion Supplement, and ImmunoCult™ NK Cell Expansion Coating Material, providing you with a complete, easy-to-use culture system. After just 14 days in culture, cells can be harvested and used directly in your downstream applications.
This kit is compatible with many of our other upstream and downstream products. For example, you can use EasySep™ cell separation kits to isolate NK cells that can then be immediately expanded using ImmunoCult™ NK Cell Expansion Kit.
Data Figures
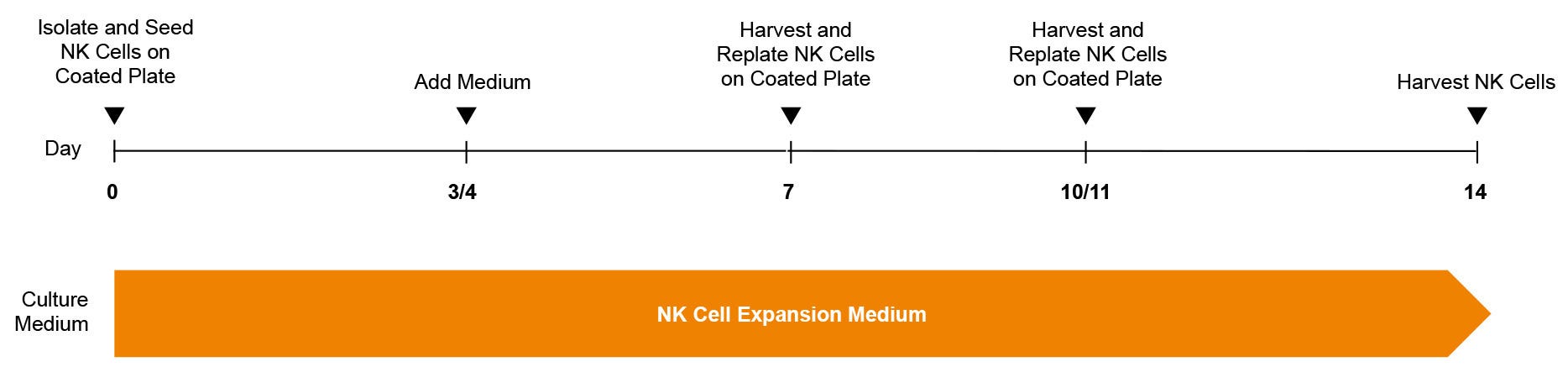
Figure 1. ImmunoCult™ NK Cell Expansion Protocol
Human natural killer (NK) cells are isolated from blood or leukapheresis using EasySep™ selection. The NK cells are cultured in ImmunoCult™ NK Cell Expansion Medium, on plates coated with ImmunoCult™ NK Cell Expansion Coating Material. After 3 days, fresh medium is added to the culture. On day 7, and again on day 10 or 11, expanding NK cells are harvested and replated on freshly coated plates. Expanded NK cells were harvested on day 14 for use in downstream assays.
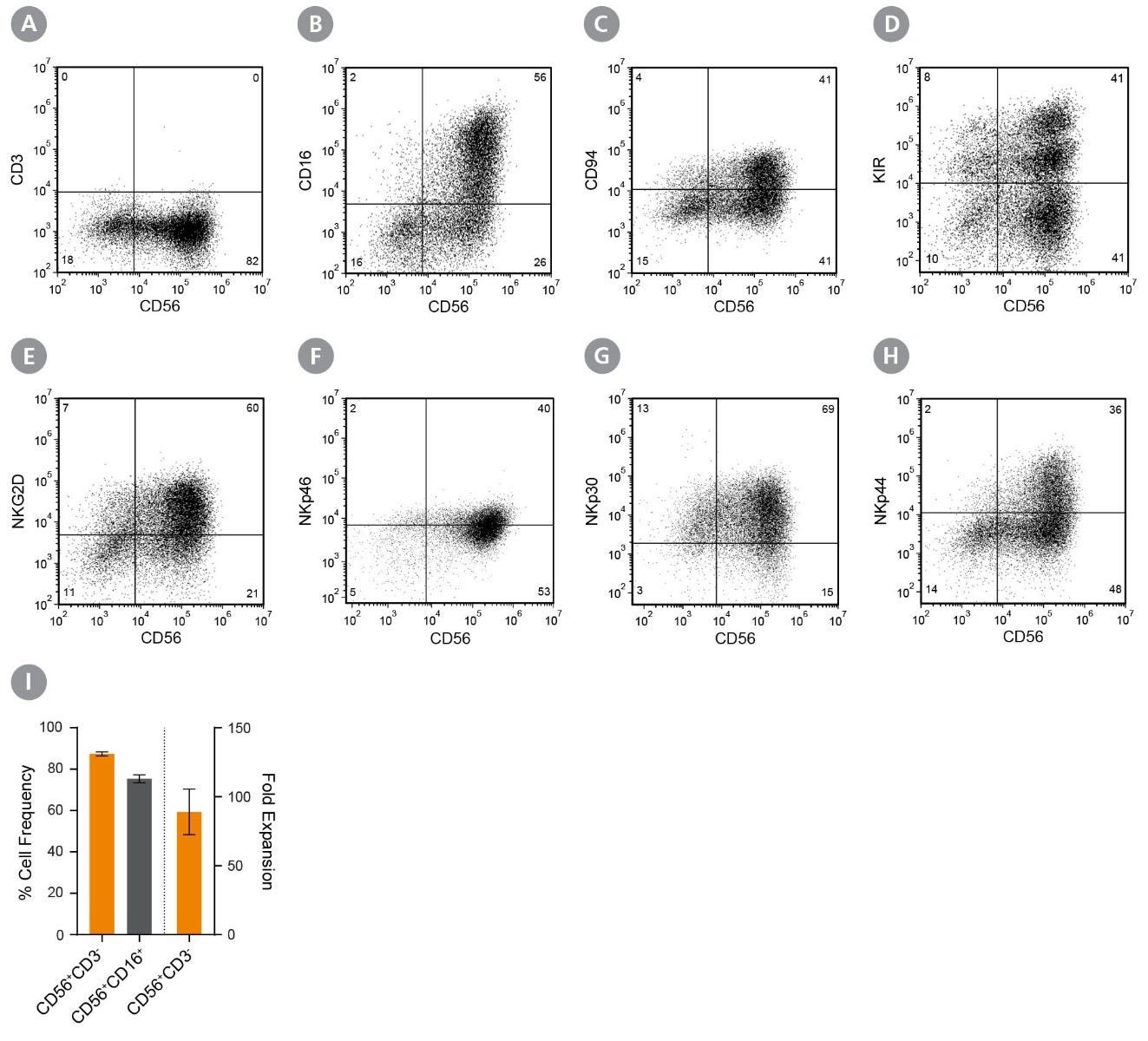
Figure 2. CD56+CD3− NK Cells Expand Over 14 Days in Feeder- and Serum-Free Culture Conditions
Isolated human CD56+CD3− NK cells were cultured using ImmunoCult™ NK Cell Expansion Kit for 14 days (Figure 1). Cells were harvested and analyzed for expression of characteristic NK cells markers, including CD56, CD3, CD16, CD94, KIR, NKG2D, NKp46, NKp30, and NKp44 by flow cytometry. Staining for killer cell immunoglobulin-like receptor (KIR) molecules was performed using two different antibody clones, HP-MA4 and 180704, which recognize distinct KIR molecules. Dead cells were excluded by light-scatter profile and DRAQ7™ staining. (A - H) Representative flow cytometry plots. (I) The average frequencies of viable CD56+CD3− and CD56+CD16+ NK cells on day 14 were 87 ± 1% and 75 ± 2%, respectively. The average fold expansion of CD56+CD3− cells was 89 ± 17. Results shown represent mean ± SEM (n = 34).
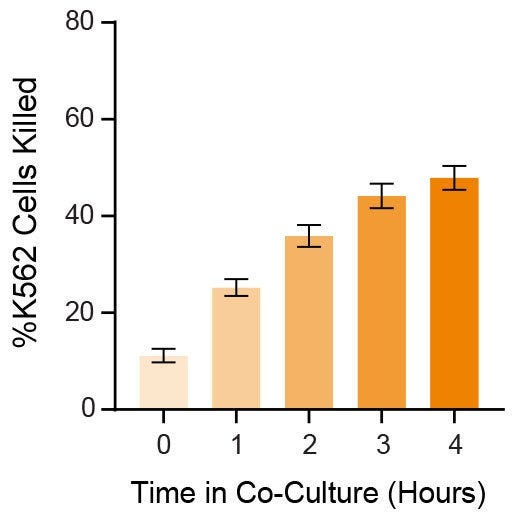
Figure 3. Expanded NK Cells Are Functional, Killing K562 Cells in Co-Culture
Isolated CD56+CD3− NK cells were expanded as described in Figure 1. Expanded NK cells were co-cultured with Incucyte® Cytolight Rapid Dye-labeled K562 cells at 1:1 ratio of NK:K562 cells at 37°C for 4 hours. Incucyte® Caspase-3/7 Dye, a caspase-inducible dye, was added to the co-culture to detect caspase-induced apoptosis of the K562 cells. Images were obtained every hour using the Incucyte® imaging system and then analyzed to determine % killing (# apoptotic K562 cells ÷ # total labeled K562 cells). After 4 hours, an average of 48 ± 2.4% K562 cells were killed (n = 9). Data represent mean ± SEM.
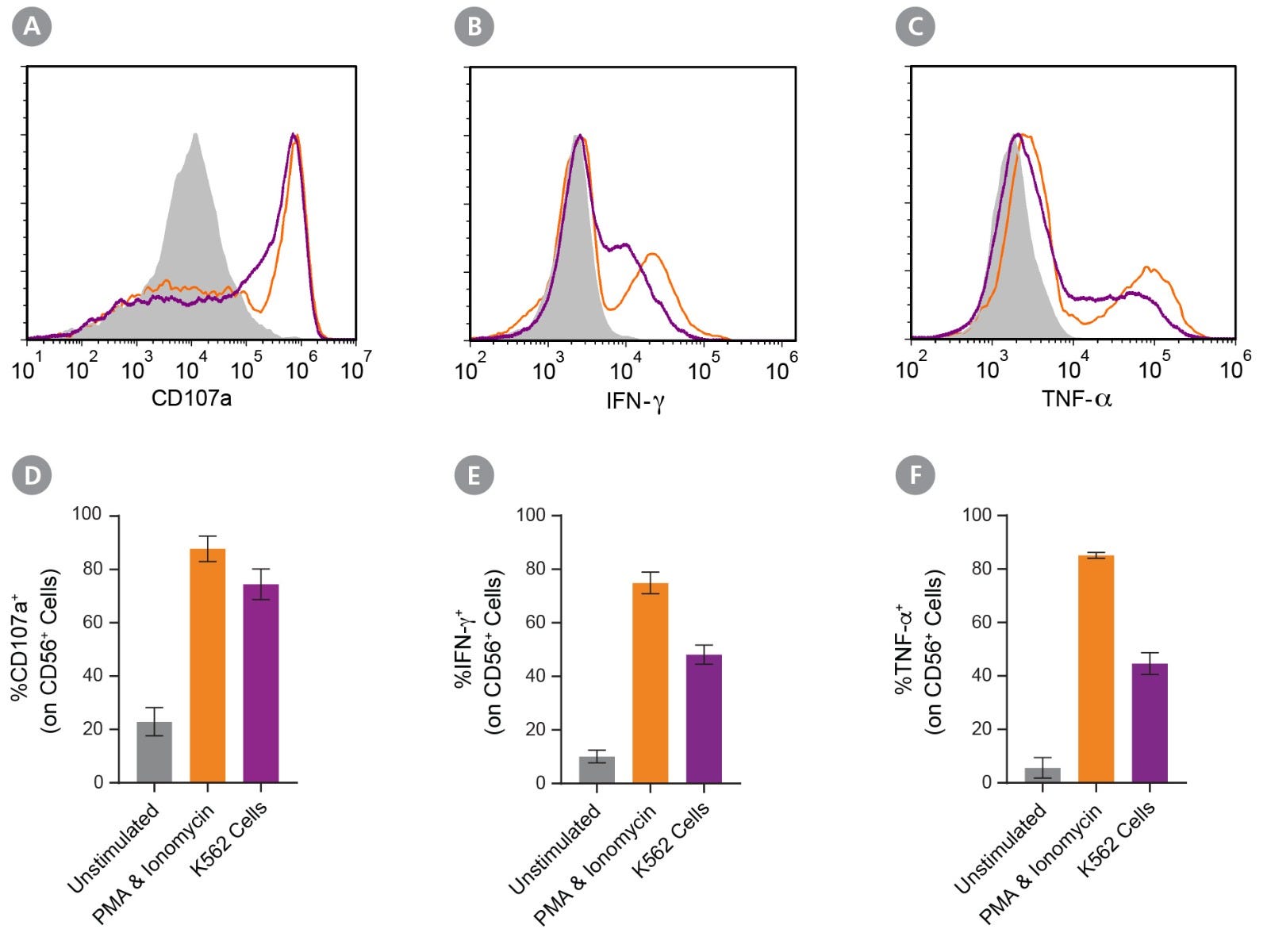
Figure 4. Expanded NK Cells Degranulate and Produce Cytokines After Stimulation
Isolated CD56+CD3− NK cells were expanded for 14 days (Figure 1). Expanded NK cells were left unstimulated (control) or were stimulated with either phorbol 12-myristate 13-acetate (PMA) and ionomycin or K562 cells at a ratio of 1:1 effector:target cells. CD107a antibody was added, and cultures were incubated at 37°C for 4 hours. After the first hour, Monensin and Brefeldin A were added. Cells were assessed for surface CD56, CD107a, and intracellular IFN-γ and TNF-α expression by flow cytometry. (A-C) Representative histograms of CD107a, IFN-γ, and TNF-α expression of unstimulated (grey filled), PMA and ionomycin-stimulated (orange), and K562-stimulated (purple) NK cell samples. (D) The average frequency of NK cells expressing surface CD107a, a marker of degranulation, was 23 ± 5% for the unstimulated control, 88 ± 5% after stimulation with PMA and ionomycin, and 74 ± 6% after stimulation with K562 cells. (E) The average frequency of NK cells expressing intracellular IFN-γ was 10 ± 2% for the unstimulated control, 75 ± 4% for cells stimulated with PMA and ionomycin, and 48 ± 4% for cells co-cultured with K562 cells. (F) The average frequency of NK cells expressing intracellular TNF-α was 6 ± 4% for the unstimulated control, 85 ± 1% cells stimulated with PMA and ionomycin, and 45 ± 4% for cells co-cultured with K562 cells. Data represent mean ± SEM (n = 6 - 13).
Protocols and Documentation
Find supporting information and directions for use in the Product Information Sheet or explore additional protocols below.
Applications
This product is designed for use in the following research area(s) as part of the highlighted workflow stage(s). Explore these workflows to learn more about the other products we offer to support each research area.
Resources and Publications
Educational Materials (7)
Related Products
-
 EasySep™ Human CD56 Positive Selection Kit ...
EasySep™ Human CD56 Positive Selection Kit ...Immunomagnetic positive selection of human CD56+ cells
-
 EasySep™ Human NK Cell Isolation Kit
EasySep™ Human NK Cell Isolation KitImmunomagnetic negative isolation of untouched human NK cells
-
 EasySep™ Human NK Cell Enrichment Kit
EasySep™ Human NK Cell Enrichment KitImmunomagnetic negative isolation of untouched human NK cells
-
 SepMate™-50 (IVD)
SepMate™-50 (IVD)Tube for density gradient centrifugation for in vitro diagnostic (IVD) applications
Item added to your cart

ImmunoCult™ NK Cell Expansion Kit
For related products, including specialized culture and storage media, supplements, antibodies, cytokines, and small molecules, visit www.stemcell.com, or contact us at techsupport@stemcell.com.