Neural Stem Cells
Neural stem cells have been identified in nearly all regions of the developing mouse, rat and human central nervous system (CNS) and in specialized neurogenic regions of the mature CNS.The identification of CNS-resident NSCs and the discovery that adult somatic cells from mouse and human can be reprogrammed to a pluripotent state, and then directed to differentiate into neural cell types, has opened the door to new therapeutic avenues aimed at replacing lost or damaged CNS cells.
We've created these resources to support your neural stem cell research.
Neural Stem Cell Culture Systems - Neurosphere vs. Adherent Monolayer Culture
This webinar provides an overview of the neurosphere and adherent monolayer methods to culture neural stem and progenitor cells. Technical aspects and details associated with performing each of the culture methods are discussed. In addition, data comparing the two methods is presented.
View Now >The Central Nervous System
The mature mammalian central nervous system (CNS) is composed of three major differentiated cell types: neurons, astrocytes and oligodendrocytes. Neurons transmit information through action potentials and neurotransmitters to other neurons, muscle cells or gland cells. Astrocytes and oligodendrocytes, collectively called glial cells, play important roles of their own, in addition to providing a critical support role for optimal neuronal functioning and survival. During mammalian embryogenesis, CNS development begins with the induction of the neuroectoderm, which forms the neural plate and then folds to give rise to the neural tube. Within these neural structures there exists a complex and heterogeneous population of neuroepithelial progenitor cells (NEPs), the earliest neural stem cell type to form.1,2 As CNS development proceeds, NEPs give rise to temporally and spatially distinct neural stem/progenitor populations. During the early stage of neural development, NEPs undergo symmetric divisions to expand neural stem cell (NSC) pools. In the later stage of neural development, NSCs switch to asymmetric division cycles and give rise to lineage-restricted progenitors. Intermediate neuronal progenitor cells are formed first, and these subsequently differentiate to generate to neurons. Following this neurogenic phase, NSCs undergo asymmetric divisions to produce glial-restricted progenitors, which generate astrocytes and oligodendrocytes. The later stage of CNS development involves a period of axonal pruning and neuronal apoptosis, which fine tunes the circuitry of the CNS. A previously long-held dogma maintained that neurogenesis in the adult mammalian CNS was complete, rendering it incapable of mitotic divisions to generate new neurons, and therefore lacking in the ability to repair damaged tissue caused by diseases (e.g. Parkinson’s disease, multiple sclerosis) or injuries (e.g. spinal cord and brain ischemic injuries). However, there is now strong evidence that multipotent NSCs do exist, albeit only in specialized microenvironments, in the mature mammalian CNS. This discovery has fuelled a new era of research into understanding the tremendous potential that these cells hold for treatment of CNS diseases and injuries.
Identification of Neural Stem Cells
Neurobiologists routinely use various terms interchangeably to describe undifferentiated cells of the CNS. The most commonly used terms are “stem cell”, “precursor cell” and “progenitor cell”. The inappropriate use of these terms to identify undifferentiated cells in the CNS has led to confusion and misunderstandings in the field of NSC and neural progenitor cell research. However, these different types of undifferentiated cells in the CNS technically possess different characteristics and fates. For clarity, the terminology used here is:
Neural Stem Cell (NSCs): Multipotent cells which are able to self-renew and proliferate without limit, to produce progeny cells which terminally differentiate into neurons, astrocytes and oligodendrocytes. The non-stem cell progeny of NSCs are referred to as neural progenitor cells.
Neural Progenitor Cell: Neural progenitor cells have the capacity to proliferate and differentiate into more than one cell type. Neural progenitor cells can therefore be unipotent, bipotent or multipotent. A distinguishing feature of a neural progenitor cell is that, unlike a stem cell, it has a limited proliferative ability and does not exhibit self-renewal.
Neural Precursor Cells (NPCs): As used here, this refers to a mixed population of cells consisting of all undifferentiated progeny of neural stem cells, therefore including both neural progenitor cells and neural stem cells. The term neural precursor cells is commonly used to collectively describe the mixed population of NSCs and neural progenitor cells derived from embryonic stem cells and induced pluripotent stem cells.
Prior to 1992, numerous reports demonstrated evidence of neurogenesis and limited in vitro proliferation of neural progenitor cells isolated from embryonic tissue in the presence of growth factors.3-5 While several sub-populations of neural progenitor cells had been identified in the adult CNS, researchers were unable to demonstrate convincingly the characteristic features of a stem cell, namely self-renewal, extended proliferative capacity and retention of multi-lineage potential. In vivo studies supported the notion that proliferation occurred early in life, whereas the adult CNS was mitotically inactive, and unable to generate new cells following injury. Notable exceptions included several studies in the 1960s that clearly identified a region of the adult brain that exhibited proliferation (the forebrain subependyma)6 but this was believed to be species-specific and was not thought to exist in all mammals. In the early 1990s, cells that responded to specific growth factors and exhibited stem cell features in vitro were isolated from the embryonic and adult CNS.7-8 With these studies, Reynolds and Weiss demonstrated that a rare population of cells in the adult CNS exhibited the defining characteristics of a stem cell: self-renewal, capacity to produce a large number of progeny and multilineage potential. The location of stem cells in the adult brain was later identified to be within the striatum,9 and researchers began to show that cells isolated from this region, and the dorsolateral region of the lateral ventricle of the adult brain, were capable of differentiating into both neurons and glia.10
The Function of Neural Stem Cells In Vivo
During mammalian CNS development, neural precursor cells arising from the neural tube produce pools of multipotent and more restricted neural progenitor cells, which then proliferate, migrate and further differentiate into neurons and glial cells. During embryogenesis, neural precursor cells are derived from the neuroectoderm and can first be detected during neural plate and neural tube formation. As the embryo develops, neural stem cells can be identified in nearly all regions of the embryonic mouse, rat and human CNS, including the septum, cortex, thalamus, ventral mesencephalon and spinal cord. NSCs isolated from these regions have a distinct spatial identity and differentiation potential. In contrast to the developing nervous system, where NSCs are fairly ubiquitous, cells with “neural stem cell” characteristics are localized primarily to two key regions of the mature CNS: the subventricular zone (SVZ), lining the lateral ventricles of the forebrain, and the subgranular layer of the dentate gyrus of the hippocampal formation (described later).11 In the adult mouse brain, the SVZ contains a heterogeneous population of proliferating cells. However, it is believed that the type B cells (activated GFAP+/PAX6+ astrocytes or astrogliallike NSCs) are the cells that exhibit stem cell properties, and these cells may be derived directly from radial glial cells, the predominant neural precursor population in the early developing brain. NPCs in this niche are relatively quiescent under normal physiological conditions, but can be induced to proliferate and to repopulate the SVZ following irradiation.10 SVZ NSCs maintain neurogenesis throughout adult life through the production of fast-dividing transit amplifying progenitors (TAPs or C cells), which then differentiate and give rise to neuroblasts. TAPs and neuroblasts migrate through the rostral migratory stream (RMS) and further differentiate into new interneurons in the olfactory bulb. This ongoing neurogenesis, which is supported by the NSCs in the SVZ, is essential for maintenance of the olfactory system, providing a source of new neurons for the olfactory bulb of rodents and the association cortex of non-human primates.12 Although the RMS in the adult human brain has been elusive, a similar migration of neuroblasts through the RMS has also been observed.13 Neurogenesis also persists in the subgranular zone of the hippocampus, a region important for learning and memory, where it leads to the production of new granule cells. Lineage tracing studies have mapped the neural progenitor cells to the dorsal region of the hippocampus, in a collapsed ventricle within the dentate gyrus.10 Studies have demonstrated that neurogenic cells from the subgranular layer may have a more limited proliferative potential than the SVZ NSCs and are more likely to be progenitor cells than true stem cells.14 Recent evidence also suggests that neurogenesis plays a different role in the hippocampus than in the olfactory bulb. Whereas the SVZ NSCs play a maintenance role, it is thought that hippocampal neurogenesis serves to increase the number of new neurons and contributes to hippocampal growth throughout adult life.12 Neural progenitor cells have also been identified in the spinal cord central canal ventricular zone and pial boundary15-16, and it is possible that additional regional progenitor populations will be identified in the future.
Neural Stem Cell Culture Systems
In vitro methodologies designed to isolate, expand and functionally characterize NSC populations have revolutionized our understanding of neural stem cell biology, and increased our knowledge of the genetic and epigenetic regulation of NSCs.17 Over the past several decades, a number of culture systems have been developed that attempt to recapitulate the distinct in vivo developmental stages of the nervous system, enabling the isolation and expansion of different NPC populations at different stages of development. Here, we outline the commonly used culture systems for generating NPCs from pluripotent stem cells (PSCs), and for isolating and expanding NSCs from the early embryonic, postnatal and adult CNS.
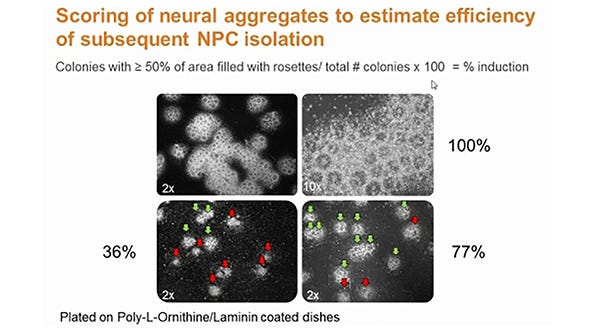
Neural induction and differentiation of pluripotent stem cells: Early NPCs can be derived from mouse and human PSCs, which include embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs), using appropriate neural induction conditions at the first stage of differentiation. While these neural differentiation protocols vary widely, a prominent feature in popular embryoid body-based protocols is the generation of neural “rosettes”, morphologically identifiable structures containing NPCs, which are believed to represent the neural tube. The NPCs present in the neural rosette structures are then isolated, and can be propagated to allow NPC expansion, while maintaining the potential to generate neurons and glial cells. More recently, studies have shown that neural induction of PSCs can also be achieved in a monolayer culture system, wherein human ESCs and iPSCs are plated onto a defined matrix, and exposed to inductive factors.18 A combination of specific cytokines or small molecules, believed to mimic the developmental cues for spatiotemporal patterning in the developing brain during embryogenesis, can be added to cultures at the neural induction stage to promote regionalization of NPCs. These “patterned” NPCs can then be differentiated into mature cell types with phenotypes representative of different regions of the brain.19-24 New protocols have been developed to generate cerebral organoids from PSC-derived neural progenitor cells. Cerebral organoids recapitulate features of human brain development, including the formation of discrete brain regions featuring characteristic laminar cellular organization.25
Neurosphere culture: The neurosphere culture system has been widely used since its development as a method to identify NSCs.26-29 A specific region of the CNS is microdissected, mechanically or enzymatically dissociated, and plated in a defined serum-free medium in the presence of a mitogenic factor, such as epidermal growth factor (EGF) and/or basic fibroblast growth factor (bFGF). In the neurosphere culture system, NSCs, as well as neural progenitor cells, begin to proliferate in response to these mitogens, forming small clusters of cells after 2 - 3 days. The clusters continue to grow in size, and by day 3 - 5, the majority of clusters detach from the culture surface and begin to grow in suspension. By approximately day seven, depending on the cell source, the cell clusters, called neurospheres, typically measure 100 - 200 μm in diameter and are composed of approximately 10,000 - 100,000 cells. At this point, the neurospheres should be passaged to prevent the cell clusters from growing too large, which can lead to necrosis as a result of a lack of oxygen and nutrient exchange at the neurosphere center. To passage the cultures, neurospheres are individually, or as a population, mechanically or enzymatically dissociated into a single cell suspension and replated under the same conditions as the primary culture. NSCs and neural progenitor cells again begin to proliferate to form new cell clusters that are ready to be passaged approximately 5 - 7 days later. By repeating the above procedures for multiple passages, NSCs present in the culture will self-renew and produce a large number of progeny, resulting in a relatively consistent increase in total cell number over time. Neurospheres derived from embryonic mouse CNS tissue treated in this manner can be passaged for up to 10 weeks with no loss in their proliferative ability, resulting in a greater than 100- fold increase in total cell number. NSCs and neural progenitors can be induced to differentiate by removing the mitogens and plating either intact neurospheres or dissociated cells on an adhesive substrate, in the presence of a low serum-containing medium. After several days, virtually all of the NSCs and progeny will differentiate into the three main neural cell types found in the CNS: neurons, astrocytes and oligodendrocytes. While the culture medium, growth factor requirements and culture protocols may vary, the neurosphere culture system has been successfully used to isolate NSCs and progenitors from different regions of the embryonic and adult CNS of many species including mouse, rat and human.
Adherent monolayer culture: Alternatively, cells obtained from CNS tissues can be cultured as adherent cultures in a defined, serum-free medium supplemented with EGF and/or bFGF, in the presence of a substrate such as poly-L-ornithine, laminin, or fibronectin. When plated under these conditions, the neural stem and progenitor cells will attach to the substrate-coated cultureware, as opposed to each other, forming an adherent monolayer of cells, instead of neurospheres. The reported success of expanding NSCs in long-term adherent monolayer cultures is variable and may be due to differences in the substrates, serum-free media and growth factors used.17 Recently, protocols that have incorporated laminin as the substrate, along with an appropriate serum-free culture medium containing both EGF and bFGF have been able to support long-term cultures of neural precursors from mouse and human CNS tissues.30-32 These adherent cells proliferate and become confluent over the course of 5 - 10 days. To passage the cultures, cells are detached from the surface by enzymatic treatment and replated under the same conditions as the primary culture. It has been reported that NSCs cultured under adherent monolayer conditions undergo symmetric divisions in long-term culture.30,33 Similar to the neurosphere culture system, adherently cultured cells can be passaged multiple times and induced to differentiate into neurons, astrocytes and oligodendrocytes upon mitogen removal and exposure to a low serum-containing medium.
Several studies have suggested that culturing CNS cells in neurosphere cultures does not efficiently maintain NSCs and produces a heterogeneous cell population, whereas culturing cells under serum-free adherent culture conditions does maintain NSCs.17 While these reports did not directly compare neurosphere and adherent monolayer culture methods using the same medium, growth factors or extracellular matrix to evaluate NSC numbers, proliferation and differentiation potential, they emphasize that culture systems can influence the in vitro functional properties of NSCs and neural progenitors. It is important that in vitro methodologies for NSC research are designed with this caveat in mind, and with a clear understanding of what the methodologies are purported to measure.34-35
Isolation Strategies for Neural Stem Cells
Immunomagnetic or immunofluorescent cell isolation strategies using antibodies directed against cell surface markers present on stem cells, progenitors and mature CNS cells have been applied to the study of NSCs. Similar to stem cells in other systems, the phenotype of CNS stem cells has not been completely determined. Expression, or lack of expression, of CD34, CD133 and CD45 antigens has been used as a strategy for the preliminary characterization of potential CNS stem cell subsets. A distinct subset of human fetal CNS cells with the phenotype CD133+ 5E12+ CD34- CD45- CD24-/lo has the ability to form neurospheres in culture, initiate secondary neurosphere formation, and differentiate into neurons and astrocytes.36 Using a similar approach, fluorescence-activated cell sorting (FACS)- based isolation of nestin+ PNA- CD24- cells from the adult mouse periventricular region enabled significant enrichment of NSCs (80% frequency in sorted population, representing a 100-fold increase from the unsorted population).37 However, the purity of the enriched NSC population was found to be lower when this strategy was reevaluated using the more rigorous Neural Colony-Forming Cell (NCFC) assay.38-39 NSC subsets detected at different stages of CNS development have been shown to express markers such as nestin, GFAP, CD15, Sox2, Musashi, CD133, EGFR, Pax6, FABP7 (BLBP) and GLAST40-45. However, none of these markers are uniquely expressed by NSCs; many are also expressed by neural progenitor cells and other nonneural cell types. Studies have demonstrated that stem cells in a variety of tissues, including bone marrow, skeletal muscle and fetal liver can be identified by their ability to efflux fluorescent dyes such as Hoechst 33342. Such a population, called the “side population”, or SP (based on its profile on a flow cytometer), has also been identified in both mouse primary CNS cells and cultured neurospheres.46 Other non-immunological methods have been used to identify populations of cells from normal and tumorigenic CNS tissues, based on some of the in vitro properties of stem cells, including FABP7 expression and high aldehyde dehydrogenase (ALDH) enzyme activity. ALDH-bright cells from embryonic rat and mouse CNS have been isolated and shown to have the ability to generate neurospheres, neurons, astrocytes and oligodendrocytes in vitro, as well as neurons in vivo, when transplanted into the adult mouse cerebral cortex.47-50 NeuroFluor™ CDr3 is a membrane-permeable fluorescent probe that binds to FABP7 and can be used to detect and isolate viable neural progenitor cells from multiple species.42-43
Brain Tumor Stem Cells
Multipotent neural stem-like cells, known as brain tumor stem cells (BTSCs) or cancer stem cells (CSCs), have been identified and isolated from different grades (low and high) and types of brain cancers, including gliomas and medulloblastomas.51-52 Similar to NSCs, these BTSCs exhibit self-renewal, high proliferative capacity and multi-lineage differentiation potential in vitro. They also initiate tumors that phenocopy the parent tumor in immunocompromised mice.53 No unique marker of BTSCs has been identified but recent work suggests that tumors contain a heterogenous population of cells with a subset of cells expressing the putative NSC marker CD133.53 CD133+ cells purified from primary tumor samples formed primary tumors, when injected into primary immunocompromised mice, and secondary tumors upon serial transplantation into secondary recipient mice.53 However, CD133 is also expressed by differentiated cells in different tissues and CD133- BTSCs can also initiate tumors in immunocompromised mice.54-55 Therefore, it remains to be determined if CD133 alone, or in combination with other markers, can be used to discriminate between tumor initiating cells and non-tumor initiating cells in different grades and types of brain tumors. Recently, FABP7 has gained traction as a CNS-specific marker of NSCs and BTSCs.42-43, 57
Both the neurosphere and adherent monolayer culture methods have been applied to the study of BTSCs. When culturing normal NSCs, the mitogen(s) EGF (and/or bFGF) are required to maintain NSC proliferation. However, there is some indication that these mitogens are not required when culturing BTSCs.57 Interestingly, the neurosphere assay may be a clinically relevant functional readout for the study of BTSCs, with emerging evidence suggesting that renewable neurosphere formation is a significant predictor of increased risk of patient death and rapid tumor progression in cultured human glioma samples.58-60 Furthermore, the adherent monolayer culture has been shown to enable pure populations of glioma-derived BTSCs to be expanded in vitro.61
Summary
Research in the field of NSC biology has made a significant leap forward over the past ~30 years. Contrary to the beliefs of the past century, the adult mammalian brain retains a small number of true NSCs located in specific CNS regions. The identification of CNS-resident NSCs and the discovery that adult somatic cells from mouse and human can be reprogrammed to a pluripotent state,62-68 and then directed to differentiate into neural cell types, has opened the door to new therapeutic avenues aimed at replacing lost or damaged CNS cells. This may include transplantation of neural progenitors derived from fetal or adult CNS tissue, or pluripotent stem cells. Recent research has shown that adult somatic cells can be directly reprogrammed to specific cell fates, such as neurons, using appropriate transcriptional factors, bypassing the need for an induced pluripotent stem cell intermediate.69 Astroglia from the early postnatal cerebra cortex can be reprogrammed in vitro to neurons capable of action potential firing, by the forced expression of a single transcription factor, such as Pax6 or the pro-neural transcription factor neurogenin-2 (Neurog2).70 To develop cell therapies to treat CNS injuries and diseases, a greater understanding of the cellular and molecular properties of neural stem and progenitor cells is required. To facilitate this important research, STEMCELL Technologies has developed NeuroCult™ proliferation and differentiation kits for human, mouse and rat, including xenofree NeuroCult™-XF. The NeuroCult™ NCFC Assay provides a simple and more accurate assay to enumerate NSCs compared to the neurosphere assay. These tools for NSC research are complemented by the NeuroCult™ SM Neuronal Culture Kits, specialized serum-free medium formulations for culturing primary neurons. Together, these reagents help to advance neuroscience research and assist in its transition from the experimental to the therapeutic phase.
References
- Temple S. Nature 414: 112-117, 2001
- Kriegstein A and Alvarez-Buylla A. Annu Rev Neurosci 32: 149- 184, 2009
- Bögler O, et al. Proc Natl Acad Sci U S A 87: 6368-6372, 1990
- Ray J, Gage FH. J Neurosci 14: 3548-3564, 1994
- Vescovi AL, et al. Neuron 11: 951-966, 1993 6. Altman J, et al. In: Restorative Neurology, Vol
- Neuronal Cell Death and Repair (AC Cuello, ed), Elsevier, Amsterdam, pp 203-225, 1993
- Reynolds BA, et al. Science 255: 1707-1710, 1992
- Reynolds BA, et al. J Neurosci 12: 4565-4574, 1992
- Reynolds BA, et al. Dev Biol 175: 1-13, 1996
- Morshead CM, et al. Neuron 13: 1071-1082, 1994
- Lois C, et al. Proc Natl Acad Sci U S A 90: 2074-2077, 1993
- Imayoshi I, et al. Nat Neurosci 11: 1153-1161, 2008
- Curtis MA, et al. Science 315: 1243-1249, 2007
- Bull ND, Barlett PF. J Neurosci 25: 10815-10821, 2005
- Conti L, Cattaneo E. Nature Reviews 11: 176-187, 2010
- Li W, et al. Proc Natl Acad Sci U S A 108(20): 8299-304, 2011
- Yan Y, et al. Stem Cells 23: 781-790, 2005
- Liem KF Jr, et al. Cell 82: 969-979, 1995
- Li XJ, et al. Development 136: 4055-4063, 2009
- Ye W, et al. Cell 93: 755-766, 1998
- Kim JE, et al. Proc Natl Acad Sci U S A 108: 3005-3010, 2011
- Li XJ, et al. Nat Biotechnol 23: 215-221, 2005
- Fasano CA, et al. Cell Stem Cell 6: 336-347, 2010
- Seaberg RM, et al. J Neurosci 22: 1784-1793, 2002
- Nakatomi H, et al. Cell 110: 429-441, 2002
- Tropepe V, et al. Dev Biology 208: 166-188, 1999
- Tropepe V, et al. Neuron 30: 65-78, 2001
- Conti L, et al. PLoS Biol 3: e283, 2005
- Pollard S, et al. Mol Cell Neurosci 38: 393-403, 2008
- Sun Y, et al. Mol Cell Neurosci 38: 245-258, 2008
- Pollard SM, et al. Cereb Cortex 16 Suppl 1: i112-i120, 2006
- Pollard SM, et al. Cell Stem Cell 4: 568-580, 2009
- Reynolds BA, Vescovi AL. Cell Stem Cell 5: 466-467, 2009
- Uchida N, et al. Proc Natl Acad Sci U S A 97: 14720-14725, 2000
- Rietze RL, et al. Nature 412: 736-739, 2001
- Reynolds BA, et al. Nat Methods 2: 333-336, 2005
- Louis SA, et al. Stem Cells 26: 988-996, 2008
- Pastrana E, et al. Proc Natl Acad Sci U S A 106(15):6387-6392, 2009
- Sun Y, et al. PLoS One 4: e5498, 2009
- Götz M, et al. Neuron 21: 1031-1044, 1998
- Hartfuss E, et al. Dev Biol 229: 15-30, 2001
- Hulspas R, et al. Cytometry 40: 245-250, 2000
- Bar EE, et al. Stem Cells 25: 2524-2533, 2007
- Cai J, et al. J Neurochem 88: 212-226, 2004
- Corti S, et al. Hum Mol Genet 15: 167-187, 2006
- Corti S, et al. Stem Cells 24: 975-985, 2006
- Vescovi AL, et al. Nat Rev Cancer 6: 425-436, 2006
- Galderisi U, et al. Cell Death Differ 13: 5-11, 2006
- Singh SK, et al. Cancer Res 63: 5821-5828, 2003
- Wang J, et al. Int J Cancer 122: 761-768, 2008
- Ogden AT, et al. Neurosurgery 62: 505-514, 2008
- Kelly JJ, et al. Stem Cells 27: 1722-1733, 2009
- Laks DR, et al. Stem Cells 27: 980-987, 2009
- Panosyan EH, et al. Pediatr Blood Cancer 55: 644-651, 2010
- Thirant C, et al. PloS One 6(1): e16375, 2011
- Pollard SM, et al. Cell Stem Cell 4: 568-580, 2009
- Takahashi K, et al. Cell 126: 663-676, 2006
- Okita K, et al. Nature 448: 313-317, 2007
- Wernig M, et al. Nature 448: 318-24, 2007
- Maherali N, et al. Cell Stem Cell 1: 55-70, 2007
- Takahashi K, et al. Cell 131: 861-872, 2007
- Yu J, et al. Science 318: 1917-1920, 2007
- Park IH, et al. Nature 451: 141-146, 2008
- Vierbuchen T, et al. Nature 463: 1035-1042, 2010
- Heinrich C, et al. PLoS Biol 8: e1000373, 2010
- Gage FH. Science 287: 1433-1438, 2000
- Singec I, et al. Nat Methods 3: 801-806, 2006
- Jensen JB, et al. Mol Neurobiol 34: 153-161, 2006
- Chaichana K, et al. Stem Cells 24: 2851-2857, 2006
- Young KM, et al. J Neurosci 27: 8286-8296, 2007