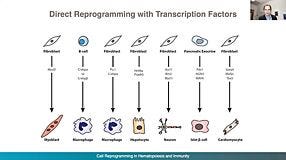
From hPSCs to Immune Cells: Key Considerations for Your Translational Journey
In this presentation, Lynn Csontos provides insight into the translational journey from a European perspective, and discusses the various demands that have to be considered during the development of clinical research projects. Lynn Csontos is Vice President for Quality Assurance and Regulatory Affairs at STEMCELL Technologies.
In this presentation, Lynn Csontos provides insight into the translational journey from a European perspective, and discusses the various demands that have to be considered during the development of clinical research projects. Lynn Csontos is Vice President for Quality Assurance and Regulatory Affairs at STEMCELL Technologies.
This presentation and the following Q&A were moderated by Dr. Glyn Stacey from EuroStemCell and Dr. Adam Hirst from STEMCELL Technologies. It was part of the workshop “From hPSCs to Immune Cells”, hosted together with the London Stem Cell Network. During the event, scientists and experts presented on advancements in hPSC culture, the generation of immune cells, and the demands of cell therapy research. Explore the full series here >
This presentation and the following Q&A were moderated by Dr. Glyn Stacey from EuroStemCell and Dr. Adam Hirst from STEMCELL Technologies. It was part of the workshop “From hPSCs to Immune Cells”, hosted together with the London Stem Cell Network. During the event, scientists and experts presented on advancements in hPSC culture, the generation of immune cells, and the demands of cell therapy research. Explore the full series here >
Publish Date:
February 11, 2022
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration
By submitting this form, you are providing your consent to STEMCELL Technologies Canada Inc. and its subsidiaries and affiliates (“STEMCELL”) to collect and use your information, and send you newsletters and emails in accordance with our privacy policy. Please contact us with any questions that you may have. You can unsubscribe or change your email preferences at any time.