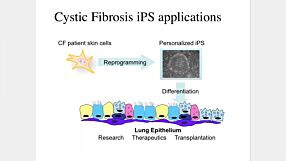
Organoid Mini-Symposium: Utilization of Next Generation CRISPR Tools to Model Health and Disease in Adult Stem Cell-Derived Organoids
Maarten Geurts, from Hans Clevers’ lab at the Hubrecht Institute, discusses the applications of adenine base editors in organoids and the potential clinical applications. Along with his colleagues, Maarten generated an intestinal organoid biobank from cystic fibrosis patients and demonstrates that adenine base editors enable efficient repair of nonsense mutations in CFTR and cause no detectable off-target effects during repair.
This webinar was part of our Organoid Mini-Symposium, held on April 22, 2020, which featured speakers using organoids in a variety of downstream applications.
Maarten Geurts, from Hans Clevers’ lab at the Hubrecht Institute, discusses the applications of adenine base editors in organoids and the potential clinical applications. Along with his colleagues, Maarten generated an intestinal organoid biobank from cystic fibrosis patients and demonstrates that adenine base editors enable efficient repair of nonsense mutations in CFTR and cause no detectable off-target effects during repair.
This webinar was part of our Organoid Mini-Symposium, held on April 22, 2020, which featured speakers using organoids in a variety of downstream applications.
Webinar Q&A
Below is a list of questions submitted by participants during the recording of the webinar. Answers are provided by Maarten Geurts (MG), with Ayse Guven (AG) as the moderator. 1. AG: How was the Cas-9 delivered into the organoid, and can you induce multiple mutations in a single gene or organoid? Also, how was the overall efficiency of this technique? MG: That’s a great question! For most of our experiments we transfect the organoids by electroporation, meaning we give them little pulses and the plasmid goes into the organoid. We can easily do multiple mutations at the same time and that’s what I showed when we did both the APC and P53 knockout at the same time. I’ve never tried two mutations in one single gene but I don’t see a good reason why that wouldn’t work. And lastly, regarding efficiency, this is very difficult for me to answer. In the field of Cas9 this really depends on the guide RNA and with our base editors we see varying efficiencies depending on the guide RNA we use. 2. AG: What is an efficient method of transfecting human tumor tissue-derived organoids, I assume using CRISPR-Cas9? MG: In our lab we mostly use electroporation. We have a NepaGene™ electroporator where you can very specifically set the electroporation conditions. Another option would be to go for lipofectamine, however, efficiencies are generally a little bit lower but if you want to start doing some experiments this is a good way to get started. In organoids that are very tricky to transfect you can also decide to go for a lentiviral transduction. Just make a lentivirus with Cas-9 and a guide RNA, incorporate it, and then with a very high efficiency you can bring Cas-9 to the organoids. 3. AG: Another question is on the technique you used to access the apical side of the organoids to introduce the drugs you tested. Which techniques did you use? MG: We culture the organoids as normal in Matrigel®, so we didn’t do any other techniques than that. The cells will just take up the drugs automatically and don’t require anything else to respond to the drugs. 4. AG: We will end the Q&A with a general question, Maarten in your opinion, what are the benefits and challenges of current organoid cultures and what solutions are available to address these challenges? MG: I think a benefit is that it’s relatively easy to establish a culture from a patient and of course you can do that with fibroblast, but with organoids you have more cell types, maybe not all, but we recapitulate the tissue of interest a lot more than through a cell line. One of the challenges that I am facing is transfection efficiency. With electroporation we tend to only, on a very good day, hit between 5 - 10% of cells in the organoids. However, if you did research on a HEK2 or a T cell line, your transfection efficiency would be 95 - 100%. So, it would be amazing to increase efficiency for organoids.
Publish Date:
May 05, 2020
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration
By submitting this form, you are providing your consent to STEMCELL Technologies Canada Inc. and its subsidiaries and affiliates (“STEMCELL”) to collect and use your information, and send you newsletters and emails in accordance with our privacy policy. Please contact us with any questions that you may have. You can unsubscribe or change your email preferences at any time.