MethoCult™ Formulation Optimized for Mouse BFU-E Detection
.
- Document # 29953
- Version 1.0.0
- Nov 2009
STEMCELL Technologies has developed MethoCult™ SF M3436 (Catalog #03436) for optimal detection of mouse burst-forming unit-erythroid (BFU-E) in colony-forming cell (CFC) assays.
MethoCult™ SF M3436 allows for easy and accurate quantitation of BFU-E in a variety of mouse cell sources by supporting erythroid colony formation and suppressing the growth of myeloid progenitors. This new medium also allows for effective evaluation of erythroid colony size, maturity and morphology.
DESCRIPTION
MethoCult™ SF M3436 is a methylcellulose-based semi-solid culture medium. Methylcellulose-based media are used to examine the frequency and growth properties of hematopoietic progenitors by their ability to form colonies of morphologically recognizable mature cells.
MethoCult™ SF M3436 is a complete medium so the addition of other components is not required.
COMPONENTS INCLUDE:
- Methylcellulose in Iscove’s MDM
- Bovine Serum Albumin
- rh Insulin
- Transferrin (iron-saturated)
- 2-Mercaptoethanol
- Cytokines (including erythropoietin)
- Supplements
APPLICATIONS
MethoCult™ SF M3436 is designed for detection, quantitation and evaluation of mouse erythroid progenitors in a CFC assay. The mouse CFC assay can be used to detect BFU-E for the following applications:
- Colony assays of mouse BFU-E from bone marrow (BM), peripheral blood (PB), spleen, fetal liver (FL) and other hematopoietic tissues
- Assessment of BFU-E frequencies in purified progenitor populations obtained by cell sorting or other isolation procedures
- Enumeration of erythroid progenitors in ex vivo expansion cultures of hematopoietic progenitors
- Detection of erythroid differentiation of mouse embryonic stem (ES) cells or induced pluripotent (iPS) cells
- Measurement of gene transfer efficiency in erythroid progenitors
- Evaluation of erythropoiesis in mutant or transgenic mouse models of hematopoietic disorders (e.g. sickle cell anemia, thalasemia, polycythemia, leukemia, myelodysplasia)
- Evaluation of compounds, such as cytokines, cytotoxic drugs, and RNAi, for their effects on erythroid progenitor numbers, proliferation, differentiation, morphology and viability
PROCEDURE
For detailed instructions please refer to the Technical Manual for Mouse Colony-Forming Cell Assays Using MethoCult™ (Catalog #28405) or the Product Information Sheet (PIS)
PLATING DENSITY
The recommended cell plating density in MethoCult™ SF M3436 depends on the frequency of erythroid progenitors in the sample. Plating density guidelines for various cell sources are presented in Table 1. Within these ranges, a linear relationship is expected between the number of plated cells and the number of colonies.
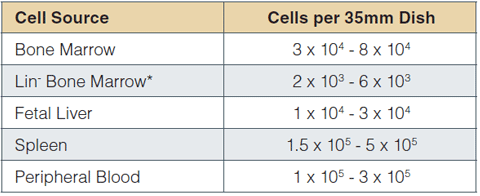
*Isolated using EasySep™ (Catalog #19756) or StemSep™ (Catalog #13036) Mouse Progenitor Cell Enrichment kits
Table 1: Recommended plating density ranges for different mouse cell populations. Plating concentrations were established using 6 - 12-week-old C57BL/6 mice. For other strains, transgenic or treated mice, we recommend plating cells at 2 - 3 densities to establish optimal plating concentrations to achieve 25 - 50 colonies per dish.
Cells should be plated at various densities, particularly if the BFU-E frequency in a cell preparation is unknown or expected to differ from normal tissue. Examples of circumstances where BFU-E frequency may vary:
- The sample is from a mutant mouse strain
- The sample is from an ex vivo expansion culture
- A cytokine, drug or other compound is being tested
COLONY NUMBERS
Typical progenitor frequencies (per 105 cells) obtained in CFC assays using MethoCult™ SF M3436 are presented in Table 2. Similar results can be expected for other mouse strains, e.g. BALB/c. BFU-E frequencies may differ in various mouse disease models or in mutant mouse strains where hematopoiesis is affected. Various plating densities should be used under these conditions to establish the optimal plating density.
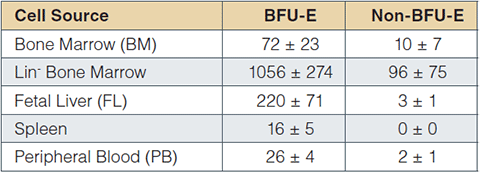
Table 2: BM, spleen and PB cells were obtained from 6 - 12-week-old C57BL/6 mice. FL cells were obtained from C57BL/6 embryos at day 14.5 of gestation. Lineage depleted (Lin-) BM cells were prepared using the EasySep™ Mouse Progenitor Enrichment Kit (Catalog #19756). Values are expressed as mean ± 1 standard deviation per 105 cells plate (BM, n=11; other tissues, n=3). Non-BFU-E colonies are typically colonies that do not meet the morphological criteria of BFU-E. The colonies are usually small and may contain undifferentiated myeloid and/or multilineage cells that do not develop optimally in MethoCult™ SF M3436.
COLONY MORPHOLOGY
MethoCult™ SF M3436 cultures should be scored after 14 to 16 days of incubation. Colony growth in MethoCult™ SF M3436 can be evaluated after 7 days of incubation but most colonies are undifferentiated and cannot be properly identified as erythroid by morphological criteria. Most colonies in MethoCult™ SF M3436 differentiate to morphologically recognizable erythroid colonies between 7 and 14 days. See ‘Kinetics of Colony Formation’ for more information.
BFU-E
Erythroid colonies are readily detectable in MethoCult™ SF M3436 after approximately 14 days of culture. Non-erythroid colonies are notably absent. Typical colony morphology from various cell sources is described in Table 3.
Non-BFU-E
A small subset of colonies detected in MethoCult™ SF M3436 after 14 days do not meet the morphological criteria of BFU-E. These are classified as ‘non-BFU-E’ colonies.
- Intermediate in size
- Do not have a dense core
- Cells are larger than cells in a BFU-E colony and look less differentiated
- Clusters of small cells may be present but most cells are separate
- May contain large cells resembling megakaryocytes

Table 3: Description of colonies derived from BFU-E from various cell sources in MethoCult™ SF M3436.
OTHER CELLS
Small clusters of cells may be present in MethoCult™ SF M3436 cultures. These clusters do not usually contain enough cells to be counted as a colony (i.e. less than 30 cells). Similar clusters are identified in control cultures without erythropoietin (EPO). This suggests that they are derived from non-erythroid progenitors that do not develop optimally in MethoCult™ SF M3436.
KINETICS OF COLONY FORMATION
The changes in colony development between 7 and 21 days of incubation are illustrated by changes in expression of the erythroid differentiation antigens CD71 and TER-119 in colonies harvested from culture dishes and analyzed by flow cytometry (Figure 1). At Day 7, the majority of viable cells (propidium iodide negative) in plucked colonies express high levels of CD71. Very few viable cells in plucked colonies express TER-119, the late erythroid marker (Figure 1A). These CD71+TER119- erythroid cells represent pro-erythroblasts and early basophilic erythroblasts.1
At Day 14, there is an increase of viable cells in plucked colonies expressing TER-119. CD71+TER119+ cells reach 18% in the population of cells harvested from colonies on Day 14 (Figure 1B). These cells represent the more mature basophilic erythroblast stage.
Colonies that appear morphologically undifferentiated on Day 14 contain CD71+ cells, but fewer than 1% of the cells are TER119+ (Figure 1C). These colonies continue to differentiate and contain 27% TER119+ cells after 21 days (Figure 1D).
MethoCult™ SF M3436 should not be used to assay multilineage progenitors as it does not support proliferation and differentiation of non-erythroid progenitors. Neither Gr-1, the monocyte/granulocyte marker, nor CD11b/Mac1, the monocyte/macrophage marker, were expressed in colonies plucked from MethoCult™ SF M3436 at any time-point between 7 and 21 days.
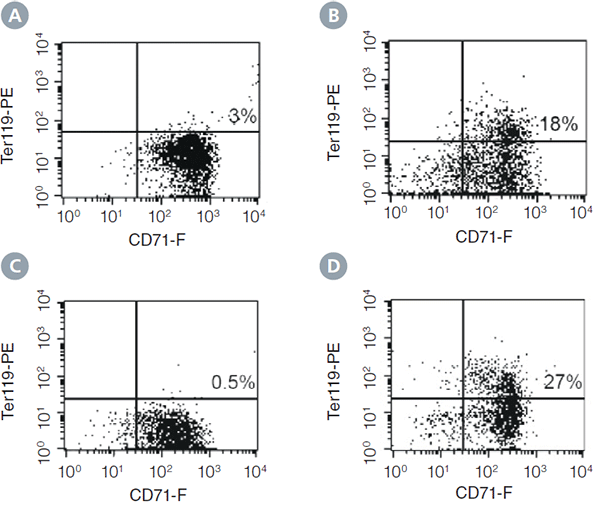
Figure 1: Expression of the erythroid differentiation antigens CD71 and TER-119 on cells from colonies harvested at different time points from cultures of mouse BM cells in MethoCult™ SF M3436 Medium. A) Colony harvested at day 7 when most colonies are still morphologically undifferentiated. B) BFU-E derived colony harvested on day 14 when most colonies are morphologically recognizable as erythroid. C) Undifferentiated colony harvested on day 14. D) Colony that was undifferentiated on day 14, and cultured on until harvest at day 21. Percentages indicate the frequency of CD71+TER119+ cells.
OTHER MOUSE METHOCULT™ MEDIA
STEMCELL provides several methylcellulose-based media formulations that can be used for colony assays of mouse erythroid progenitor cells (Table 4).
MethoCult™ GF M3434 supports colony formation by immature to mature BFU-E, as well as granulocyte/macrophage progenitors, which are much more abundant than erythroid progenitors in mouse BM. In addition to MethoCult™ SF M3436, STEMCELL Technologies offers MethoCult™ M3334 for studying erythroid progenitors. While this formulation also only supports erythroid progenitors, it is specifically formulated for the detection and quantification of late progenitors, especially CFU-E and late BFU-E.
STEMCELL Technologies provides incomplete media (MethoCult™ M3231, MethoCult™ M3234 and MethoCult™ SF M3236) which can be used to test the effects of cytokines, mimetics and other compounds added by the investigator on mouse progenitor cell growth.
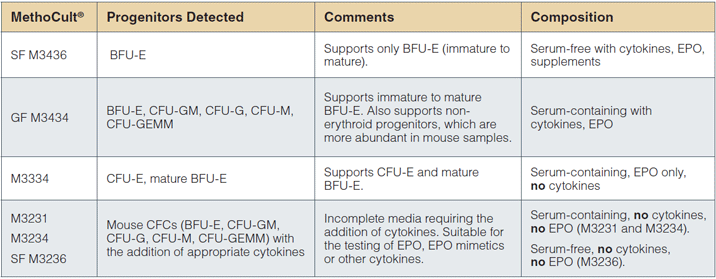
Table 4: Mouse MethoCult™ products and CFC types detected.
REFERENCES
- Zhang 1. et al., Blood 102: 3938 -3946, 2003
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration


