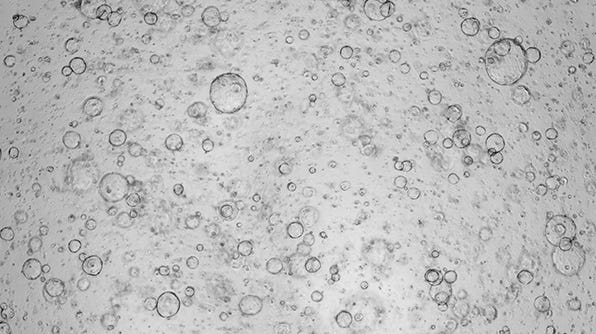
Expansion of Hepatic Organoids via Single Cells Using HepatiCult™ Organoid Growth Medium
In order to standardize seeding input for liver organoid expansion and differentiation, established organoids may be passaged using single cells rather than fragments. The following is a protocol for the expansion of human liver organoid cultures from single cells using HepatiCult™ Organoid Growth Medium.
Materials
- Healthy hepatic organoid cultures expanded in HepatiCult™ Organoid Growth Medium (Human)
- HepatiCult™ Organoid Growth Medium (Human) (Catalog #100-0385)
- DMEM/F-12 + 15 mM HEPES (Catalog #36254)
- 25% Bovine serum albumin (BSA) solution in water
- Corning® Matrigel® (Corning 356231, ≥ 8mg/mL protein)
- Sterile phosphate-buffered saline (PBS)
- Antibiotics (e.g. gentamicin)
- TrypLE™ Express Enzyme (1x), no phenol red (Thermo Fisher 12605010)
- 5 mM Y-27632 (ROCKi; Catalog #72302) solution in water
- 15 mL conical tubes (Catalog #38009)
- 24-well tissue culture-treated plate (Catalog #38017)
- 6-well tissue culture-treated plate (Catalog #38015)
- Sterile 200 µL and 1000 µL tips
- 10 mL and 25 mL serological pipettes (Catalog #38004 and Catalog #38005)
- Styrofoam box with ice
- Styrofoam box with dry ice
- Incubator with atmospheric O2 and 5% CO2 at 37°C
Preparation
- Thaw HepatiCult™ Organoid Growth Supplement overnight at 2 - 8°C. Prepare 100 mL complete HepatiCult™ Organoid Growth Medium (OGM) + Y-27632 using sterile technique:
- 5 mL HepatiCult™ Organoid Growth Supplement
- 94.8 mL HepatiCult™ Organoid Basal Medium
- 200 µL of 5 mM Y-27632 (final concentration 10 µM)
- Add antibiotics (e.g. 50 µg/mL gentamicin)
- Warm OGM + Y-27632 to room temperature (15 - 25°C) before use.
Note: Once thawed, use HepatiCult™ Organoid Growth Supplement immediately or aliquot and store at -20°C. After thawing aliquots, use immediately. Do not re-freeze.
Note: If not used immediately, store complete HepatiCult™ OGM + Y-27632 at 2 - 8°C for up to 2 weeks.
- Prepare 50 mL of serum-supplemented DMEM/F-12 + 15 mM HEPES (DMEM + BSA):
- 2 mL 25% BSA solution in water (1% final concentration)
- 100 µL 5 mM Y-27632 (final concentration 10 µM)
- 47.9 mL DMEM/F-12 + 15 mM HEPES
- Mix thoroughly. Store on ice.
- Prepare 30 mL of TrypLE™ + BSA:
- 28.8 mL TrypLE™ Express
- 1200 µL 25% BSA solution in water (1% final concentration)
- Mix thoroughly. Store on ice.
- Place a 24-well tissue culture-treated plate in a 37°C incubator for at least 1 hour.
- On ice, thaw approximately 40 µL Matrigel® per well to be seeded in the subsequent passage. Keep Matrigel® on ice when handling to prevent it from solidifying.
Protocol
Note: Cell strainers can be used to collect a suspension of more uniformly sized fragments if desired.
- Process organoid domes as follows:
- Check that the Matrigel® domes to be passaged are intact (i.e. the whole dome remains attached to the plate and no loose Matrigel® pieces or organoids are observed in the well). If the dome is intact, proceed to step 2. If the dome is loose, carefully remove ~500 µL of the medium in the well then add cold TrypLE™ + BSA to top-up the total volume in the well to 1 mL. Let sit for 1 minute then proceed to step 4.
- Without touching the dome, aspirate and discard the medium in each well to be passaged.
- Using a 1 mL pipettor, forcefully add 1 mL TrypLE™ + BSA to the center of each dome, then let sit for 1 minute.
- Using a 1 mL pipette tip on the pipettor, vigorously pipette the total volume in the well up and down 45 times, taking care not to generate bubbles.
Note: This results in mechanical breakdown of organoids and Matrigel® into a single-cell suspension. Some smaller fragments may remain – these can be further dissociated by incubating the suspensions at 37°C for 5 - 10 minutes with periodic trituration/agitation using a pipettor or vortex.
- Add 1 mL cold DMEM + BSA to each well. Transfer the entire volume of single-cell suspensions from each well to a 15 mL conical tube on ice.
Note: If required, pool the contents of up to 4 wells in one 15 mL tube.
- Wash the wells that the organoids were harvested from with 1 mL cold DMEM + BSA. Add this volume to the 15 mL tube prepared in step 5.
- Add ice-cold DMEM + BSA to each tube so that the total volume of each tube is 12 mL. Centrifuge the tube(s) at 290 x g for 5 minutes. Aspirate as much of the supernatant as possible without disturbing the pellet.
- Resuspend cells in 2 mL of DMEM + BSA.
Note: If more than 4 wells from the same treatment condition/donor were dissociated, cells from all processed wells can be pooled into the same 15 mL tube at this stage.
- Perform a live cell count.
- Determine the volume required to transfer the desired number of single cells to domes in the subsequent passage. Transfer this volume to a 15 mL conical tube containing 1 mL of DMEM + BSA. Prepare one tube for every dome to be seeded in the subsequent passage.
Note: Domes should be seeded with 5,000 - 15,000 live cells. Seeding density should be optimized for each donor.
- Centrifuge tubes containing single cells at 290 x g for 10 minutes. Aspirate as much of the supernatant as possible without disturbing the pellets (the pellet is often not visible). Place tubes on ice.
- Remove the 24-well plate from the incubator and place in a biosafety cabinet.
- Process one tube/pellet at a time, as described below. Work quickly to ensure the Matrigel® does not solidify. Pipette tips can be cooled when working with Matrigel® to help minimize premature solidifying.
- Using a pipettor with a 200 µL pipette tip, add 30 µL of thawed Matrigel® on top of the pellet. Without generating bubbles, gently mix the duct-Matrigel® suspension by pipetting up and down 5 - 8 times, going to only the first stop of the pipettor.
- Set the pipettor volume to 40 µL. Add the entire suspension to the center of one well of the 24-well plate to form a dome. While dispensing, gradually move the pipette tip upwards so that the fragments are evenly distributed throughout the dome. Dispense only to the first stop of the pipettor to avoid generating bubbles on top of the dome.
Note: The 8 wells in the center of a 24-well plate are the most suitable for domes since their surfaces are the most even. Wells at the edges of the plate are often slightly slanted, resulting in domes touching the wall of the well and flattening out.
- Repeat step 10 for the remaining pellets.
- Place the lid on the culture plate. Carefully place the plate in an incubator at 37°C and 5% CO2 for 10 minutes to let domes solidify.
- Remove the plate from the incubator and place in the biosafety cabinet.
- Without disturbing the domes, carefully add 750 µL of room temperature OGM + Y-27632 against the side of wells containing domes. Do not pipette directly onto the domes.
- Add sterile PBS to any unused wells. Place the lid on the culture plate.
- Incubate the plate at 37°C and 5% CO2 until ready to passage.
Note: Monitor hepatic organoids daily. They should be passaged before the lumen turns dark and collapses. Cultures require passaging every 10 - 12 days.
- Perform a full-medium change every 2 - 3 days by carefully aspirating the medium and adding 750 µL of fresh OGM + Y‑27632 at room temperature.
Note: If Matrigel® domes are loose, remove 500 µL of medium from the well, then add 500 µL of fresh medium. To avoid weekend medium changes, perform medium changes on Mondays, Wednesdays, and Fridays.
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration