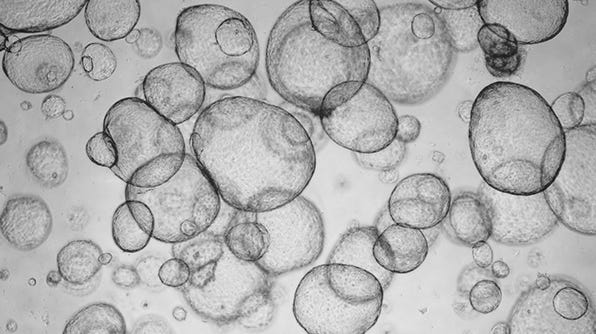
Recovery and Expansion of Hepatic Organoids Using HepatiCult™ Organoid Growth Medium
Properly thawing cryopreserved organoids can greatly impact the efficiency with which your cultures can be recovered before further use. Below is a protocol for recovery and expansion of cryopreserved hepatic organoid fragments using HepatiCult™ Organoid Growth Medium (Human).
Materials
- Cryopreserved human hepatic organoid fragments
- HepatiCult™ Organoid Growth Medium (Human) (Catalog #100-0385)
- DMEM/F-12 + 15 mM HEPES (Catalog #36254)
- 25% Bovine serum albumin (BSA) solution in water
- Corning® Matrigel® (Corning 356231, ≥ 8mg/mL protein)
- Sterile phosphate-buffered saline (PBS)
- 5 mg/mL Gentamicin
- 15 mL conical tubes (Catalog #38009)
- 24-well tissue culture-treated plate (Catalog #38017)
- 6-well tissue culture-treated plate (Catalog #38015)
- Sterile 200 µL and 1000 µL tips (Catalog #38032 and Catalog #38031)
- 10 mL and 25 mL serological pipettes (Catalog #38004 and Catalog #38005)
- Styrofoam box with ice
- Styrofoam box with dry ice
- Incubator with atmospheric O2 and 5% CO2 at 37°C
Preparation
- Thaw HepatiCult™ Organoid Growth Supplement overnight at 2 - 8°C. Prepare complete HepatiCult™ Organoid Growth Medium (OGM):
- 5 mL HepatiCult™ Organoid Growth Supplement
- 94 mL HepatiCult™ Organoid Basal Medium
- 1 mL of 5 mg/mL gentamicin (final concentration = 50 µg/mL)
- Mix thoroughly
Note: Once thawed, use HepatiCult™ Organoid Growth Supplement immediately or aliquot and store at -20°C. After thawing aliquots, use immediately. Do not re-freeze.
Note: Complete HepatiCult™ OGM can be stored at 2 - 8°C for up to 2 weeks. Warm complete medium to room temperature (15 - 25°C) before use.
- Prepare 15 mL of DMEM + BSA Thaw/Wash Medium (sufficient to thaw 3 cryovials of human hepatic organoid fragments):
- 600 µL of 25% BSA in water
- 14.4 mL DMEM/F-12 + 15 mM HEPES
- Mix thoroughly
Note: If not used immediately, store complete Thaw/Wash Medium at 2 - 8°C for up to 4 weeks. Warm Thaw/Wash Medium to room temperature before use.
- Place a 24-well tissue culture-treated plate in a 37°C incubator for at least 1 hour.
- Thaw Matrigel® on ice (approximately 40 µL per dome to be seeded). Keep Matrigel® on ice when handling to prevent it from solidifying.
Note: Cryopreserved organoids can also be recovered in dilute Matrigel® suspension cultures if desired. See the protocol for Expansion of Hepatic Organoids in Dilute Matrigel® Suspension Cultures Using HepatiCult™ Organoid Growth Medium for further details.
- Add 2 mL Thaw/Wash Medium to a 15 mL conical tube (1 per cryovial to be thawed).
- Retrieve cryovials from liquid nitrogen and store on dry ice until ready to thaw.
Protocol
- Working with one cryovial at a time, place the cryovial in a 37°C water bath to thaw for 2 - 2.5 minutes. Thawing is complete when the freezing medium becomes liquid. Proceed to the next steps immediately after the organoids are thawed.
- Wipe the outside of the cryovial with 70% isopropanol and transfer to a biosafety cabinet.
- Carefully uncap the cryovial and add 1 mL Thaw/Wash Medium to the cryovial. Using the same pipette tip, mix contents by pipetting up and down 4 times. Immediately transfer the contents to one of the 15 mL tubes containing 2 mL of Thaw/Wash Medium.
- Wash the inside of the cryovial and lid with 1 mL Thaw/Wash Medium and add this volume to the 15 mL tube. Repeat this process with another 1 mL Thaw/Wash Medium.
Note: The 15 mL tubes containing thawed fragments can be stored on ice while thawing additional cryovials.
- Centrifuge the 15 mL tubes at 290 x g for 5 minutes. Aspirate as much of the supernatant as possible without disturbing the pellet. Place the tubes on ice.
- Remove the 24-well plate from the incubator and place in the biosafety cabinet.
- Process one tube/pellet at a time, as described below. Work quickly to ensure the Matrigel® does not solidify. Pipette tips can be cooled when working with Matrigel® to help minimize premature solidifying.
Note: The 8 wells in the center of a 24-well plate are the most suitable for domes since their surfaces are the most even. Wells at the edges of the plate are often slightly slanted resulting in domes touching the wall of the well and flattening out.
- Using a pipettor with a 200 µL pipette tip, add 60 µL of thawed Matrigel® on top of the pellet. Without generating bubbles, gently mix the fragment-Matrigel® suspension by pipetting up and down 5 - 8 times, going only to the first stop of the pipettor.
- Set the pipettor volume to 35 µL. Mix the suspension and add 35 µL to the center of each of 2 wells of the 24-well plate to form domes. While dispensing, gradually move the pipette tip upwards so that the fragments are evenly distributed throughout the dome. Dispense only to the first stop of the pipettor to avoid generating bubbles on top of the dome.
- Repeat step 7 for the remaining pellets.
- Place the lid on the culture plate. Carefully place the plate in an incubator at 37°C and 5% CO2 for 10 minutes to let domes solidify.
- Remove the plate from the incubator and place in the biosafety cabinet.
- Without disturbing the domes, carefully add 750 µL of room temperature complete HepatiCult™ OGM against the side of wells containing a dome. Do not pipette directly onto the domes.
- Add sterile PBS to any unused wells. Place the lid on the culture plate.
- Incubate the plate at 37°C and 5% CO2 until ready to passage (up to 1 week).
Note: Monitor hepatic organoids daily. Organoids should be passaged before the lumen turns dark and organoids collapse. Cultures require passaging every 7 - 10 days.
- Perform a full-medium change every 2 - 3 days by carefully aspirating the medium and adding 750 µL of fresh complete HepatiCult™ OGM at room temperature (15 - 25°C).
Note: If Matrigel® domes are loose, remove 500 µL of medium from the well, then add 500 µL of fresh medium. To avoid weekend medium changes, perform medium changes on Mondays, Wednesdays, and Fridays.
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration