Maturation of Culture-Expanded Erythroid Progenitor Cells
Technical tip from our dedicated team of Product and Scientific Support specialists
- CD34+ cells (freshly isolated or cryopreserved from cord blood or bone marrow)
- StemSpan™ SFEM II Medium
- StemSpan™ Erythroid Expansion Supplement
- Erythropoietin (EPO)
- Human AB Serum
- Tissue culture plates
- General lab equipment (incubator, centrifuge, pipettes etc.)
- Anti-Human CD71 Antibody
- Anti-Human GlyA Antibody
Step 1. Expand Cells
Plate CD34+ cells at ~10,000 cells per mL in StemSpan™ SFEM II medium supplemented with Erythroid Expansion Supplement. Add fresh medium and supplement on day 4 and replate or dilute the cells in fresh medium and supplement every 3 - 4 days to ~1 x 105 cells per mL. If maintained properly, cells will continue to proliferate and generate CD71+GlyA+ erythroblasts for > 2 weeks.Step 2. Harvest and Wash Culture-Expanded Erythroblasts
Collect and count cells, while assessing viability, e.g., with trypan blue and a hemocytometer, or an automated cell counter. If desired, measure CD71 and GlyA expression using flow cytometry. Centrifuge for 5 - 10 minutes at 200 x g to pellet. Aspirate the supernatant. Wash once with either Iscove’s MDM or StemSpan™ medium (without added growth factors) to remove residual Erythroid Expansion Supplement or other additives. Cell yield can be variable, as it is highly dependent on the quality of the original CD34+ cell sample. A cell yield of at least 1000 CD71+GlyA+ cells per original CD34+ cell is typical after 2 weeks of culture, but it is possible to get as many as 100,000 or more erythroblasts per original CD34+ cell with high quality samples. If cells were maintained optimally during the expansion culture, viability should be high (~90%). Viability might be lower if cell growth was poor (< 1000 erythroblasts per original CD34+ cell) or if cultures were overgrown due to high plating density and/or suboptimal refeeding and replating.Step 3. Replate in Maturation Conditions
Resuspend the cell pellet at a concentration of 5 - 10 x 105 cells per mL in fresh SFEM II medium supplemented with 3% normal human AB serum and recombinant human EPO (at a concentration of 1 - 3 U/mL). The inclusion of human serum is important to maintain cell viability.Step 4. Analyze Cells
After 4 - 7 days of culture, examine the cells for CD71 and GlyA expression by flow cytometry. To measure hemoglobinization, cells may be prepared on slides using a cytocentrifuge and stained with benzidine or a similar reagent. During maturation cell numbers do not increase significantly, viability can be variable and is typically lower than before maturation, cell size decreases, CD71 expression decreases but remains detectable on most cells, GlyA expression remains high or increases further, and cell pellets become visibly red due to the accumulation of Hb. Enucleated cells may be observed when cultures are viewed under a microscope, indicated by the presence of reticulocytes and pyrenocytes (the extruded nucleus encased in a thin cytoplasmic layer) (Figure 1).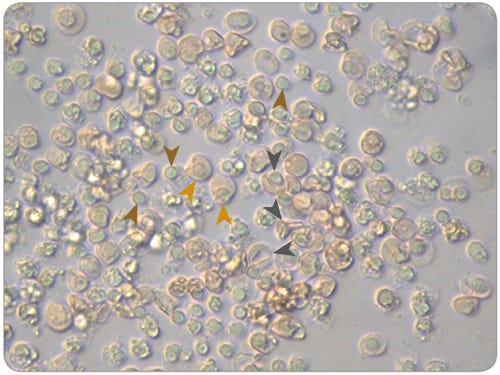
Figure 1. Morphology of Normoblasts, Reticulocytes and Pyrenocytes in Maturation Cultures of Erythroid Progenitor Cells Cultured in SFEM II
Erythroid cells, including pyrenocytes (brown arrows), normoblasts (gold arrows) and reticulocytes (dark grey arrows), viewed under a microscope at 40X magnification after 14 days of culture in expansion conditions and 7 days of culture in maturation conditions, using SFEM II and the protocol described above.
For more information about StemSpan™ SFEM II and StemSpan™ Erythroid Expansion Supplement please contact techsupport@stemcell.com.
This tech tip is published as part of our Hematopoiesis Hub. Visit to find more tips about classic and new products, see conversations with experts in the field about their science, request a wallchart and more!
References
- Giarratana MC et al. (2005) Nat Biotechnol 23: 69-74.
- Leberbauer C et al. (2005) Blood 105(1): 85-94.
- Miharada K et al. (2006) Nat Biotechnol 24(10): 1255-6.
- Migliaccio G et al. (2010) Cell Transplant 19(4): 453-69.
- van den Akker E et al. (2010) Haematologica 95(9): 1594-8.
- Timmins NE et al. (2011) Tissue Eng Part C Methods 17(11): 1131-7.
- Giarratana MC et al. (2011) Blood 118(19): 5071-9.
- Tirelli V et al. (2011) Stem Cells Int Epub, DOI: 10.4061/2011/602483.
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration


