"COVID-19 Research Updates" Featuring Dr. Josef Penninger
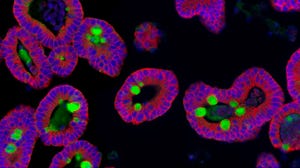
Dr. Josef Penninger's team has identified a trial drug that can significantly block early stages of COVID-19 infection in human blood vessel and kidney organoids
Dr. Josef Penninger is the Director of both the Life Sciences Institute at the University of British Columbia and the Institute for Molecular Biotechnology of the Austrian Academy of Sciences in Vienna, Austria. He was previously on The Stem Cell Podcast to talk about his research developing human blood vessel organoids. In 2020, Dr. Penninger’s team identified a trial drug that can significantly block early stages of COVID-19 infection in human blood vessel and kidney organoids.
Find more episodes at stemcellpodcast.com or wherever you get your podcasts.
In Vitro Modeling for Developing COVID-19 Therapeutics
Learn more about Dr. Penninger’s COVID-19 research and his experience with organoids.
Read Dr. Penninger’s Profile >This Episode's Science Round Up:
- COVID Drugs Cause Heart Abnormalities – Hydroxychloroquine and azithromycin have been considered possible drugs for COVID-19, but patients treated with the drugs had prolonged QT intervals that can lead to arrhythmias and sudden cardiac death.
- Monkeys Protected by COVID-19 Vaccine – For the first time, a COVID-19 vaccine candidate has protected rhesus macaques from being infected by the SARS-CoV-2 virus.
- New, Rapid COVID-19 Test – A new diagnostic test for COVID-19 has been established that uses CRISPR-Cas12 to identify the SARS-CoV-2 virus in samples in less than 40 minutes.
- SARS-CoV-2 Mapped Out – Investigators have generated a high-resolution map of the SARS-CoV-2 transcriptome and epitranscriptome, gaining insights into unknown RNA transcripts and RNA modification sites.
- Cell Likely Targeted by the COVID-19 Virus – Researchers have uncovered subsets of cells in the lung, the nasal passages, and the intestine that co-express ACE2 and TMPRSS2, which promote cellular entry of SARS-CoV-2.
- Nose Cells Identified as Likely COVID-19 Virus Entry Point – High expression levels of two viral entry-associated genes have been found in nasal epithelial cells, highlighting the cells’ potential role in initial viral infection.
- Heart Model for COVID-19 Infection – To study cardiomyocyte-specific infection by SARS-CoV-2, scientists have developed a model system using iPSC-derived cardiomyocytes that is susceptible to infection and expresses low levels of ACE2.
Related Resources
Nature Research Roundtable: Organoids
Learn key insights from the discussions and watch the presentations from our Nature Research Roundtable on organoids.
Organoid E-Book
Read the Wiley "Essential Knowledge Briefing" on the evolution and applications of organoid research.
Patient-Derived Organoids for Drug Screening and Development
Dr. Sylvia Boj describes how patient-derived intestinal organoids are being used to screen cystic fibrosis patients for treatment response.
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration