Monolayer Culture of Human Mammary Epithelial Cells
.
- Document # 29178
- Version 2.1.0
- Nov 2012
Mammary Epithelial Cell Hierarchy
The mammary gland is a compound, tubulo-alveolar gland composed of a series of branched ducts that drain sac-like alveoli (lobules) during lactation, surrounded by a fat pad in mice and by fibrous and fatty connective tissues in humans. Emerging evidence indicates that the mammary epithelium in both humans and mice may comprise a cellular hierarchy: from the mammary stem cell to various progenitors, and finally to differentiated luminal and myoepithelial cells. Two types of luminal cells line the mammary gland ducts and alveoli: those that express the estrogen receptor (ER), and those that do not (ER-). Basal to the luminal cells and adjacent to the basement membrane are the myoepithelial cells, specialized contractile epithelial cells that express smooth muscle actin and smooth muscle myosin.
Electron microscopic studies demonstrate that approximately 3 - 8% of all mammary epithelial cells do not have a terminally differentiated phenotype, and are considered to reside higher within the mammary epithelial cell hierarchy.1,2 For instance, it is thought that ER- luminal cells in the virgin mammary gland are progenitor cells that proliferate during pregnancy to generate alveoli; those cells in the alveoli are then capable of producing milk during lactation.3 The most primitive cell in the hierarchy would be the mammary stem cell (MaSC), traditionally defined as a cell that can generate both the ductal and lobular structures of the mammary gland (complete with all the cell lineages), and can self-renew. However, as will be described below, evidence suggests that a new definition for the MaSC may be needed.
Human Mammary Stem Cells
Early experiments used in vitro CFU assays to detect primitive cells in the human mammary epithelium, and demonstrated the existence of three distinct progenitors: the luminal-restricted progenitor (EpCAMhiCD49f+MUC1+), the myoepithelial-restricted progenitor, and the bipotent progenitor (EpCAMlowCD49fhiMUC1-), which can generate luminal and myoepithelial cells.4-7 Serial passaging of the colonies generated by bipotent progenitors has shown that the myoepithelial-restricted progenitor is descended from the bipotent progenitor.5
The detection of human MRUs has been much more difficult than that of mouse MRUs. Human mammary cells do not grow well when transplanted into cleared fat pads, presumably due to inappropriate epithelial-stromal interactions.8 To circumvent this, investigators have transplanted single cell suspensions of human mammary epithelial cells (HMECs) into humanized fat pads (i.e. fat pads pre-inoculated with human mammary fibroblasts) and have embedded HMECs within collagen gels and/or Matrigel™, placed subcutaneously or under the renal capsule of immune-deficient mice.9-11 HMECs transplanted under the renal capsule recapitulate histologically normal-looking human mammary epithelium, complete with polarized ß-casein+ luminal cells (when the host is made pregnant), ER+ luminal cells, and smooth muscle actin-expressing myoepithelial cells.11 Unfortunately, the generation of an epithelial outgrowth in vivo does not imply selfrenewal capacity in the progenitor cell. To add additional rigor to the assay, gels can be removed from the recipient mouse 4 weeks after implantation, dissociated, and the cells seeded into a CFU assay. In addition to quantifying MRUs, the CFU assay indirectly detects cells that can generate epithelial outgrowths in vivo as well as primitive daughter progenitor cells, including bipotent progenitors. Flow sorting of freshly dissociated human breast tissue shows that human MRUs have an EpCAMlowCD49fhi phenotype, implying that MRUs are a type of basal cell.11 This phenotype is identical to that of the bipotent progenitors, but it is not known whether MRUs and bipotent progenitors otherwise overlap.
Aldehyde dehydrogenase (ALDH), an enzyme whose activity can be detected by flow cytometry using the ALDEFLUOR™ kit (Catalog #01700), was originally reported to be a robust marker for identifying mammary stem cells in normal human breast tissue.12 However, a recent report indicates that ALDH expression may be restricted to a subset of luminal progenitor cells.13 It is not yet known if these ALDH+ luminal cells are equivalent to the luminal-restricted stem cells identified in the mouse mammary gland. Another recent surrogate assay for detecting primitive cells is the sphere assay. Most cells seeded into non-adherent cell culture systems in serum-free media will undergo anoikis; however, it is thought that primitive cells can survive and proliferate to generate clones of cells that cluster together. Mammary cell-derived clusters are termed ‘mammospheres’, in parallel to the neurospheres described for the neural system.14-16 Mammospheres display some self-renewal ability upon disaggregation and are enriched for multipotent epithelial progenitors,14 but it is not yet known which type of mammary stem cell - luminal, basal or both - read out in the mammosphere assay.
Breast Cancer Stem Cells
Cancer theory suggests that the stem and progenitor cells of the mammary epithelium are the only cells long-lived enough to acquire the multiple genetic mutations required for malignant transformation.17 While human mammary tumors have stem cell and non-stem cell components, only the stem cell fraction can generate new tumors upon transplantation into immunodeficient hosts.18 These cancer stem cells (CSCs) have been described as having an EpCAM+CD44+CD24low phenotype, with low expression of luminal-specific CD24 suggesting a basal cell origin. However, some CSCs have also been described with an ALDH+ phenotype that has little overlap with the CD44+/ CD24- phenotype.12
Interestingly, women carrying mutations in the BRCA1 gene, a mutation normally associated with basal-like breast tumors, show an expanded luminal progenitor population.19 Gene expression profiling has revealed that breast tissue heterozygous for a BRCA1 mutation and basal breast tumors share similarities with normal luminal progenitor cells more so than with any other subset, including the stem cell-enriched population. These new findings suggest that the luminal progenitor population is a target for transformation in BRCA1- associated basal tumors, a finding supported by recent mouse models of basal-like breast cancer.20
1.0 Preparation of Complete EpiCult™ Media
1.1 Preparation of EpiCult™-B Medium (Human)
- Prepare Complete EpiCult™-B Medium by adding 1 mL of thawed EpiCult™-B Supplement (Catalog #05603) to 100 mL of EpiCult™-B Basal Medium (Catalog #05602).
- Add freshly prepared Hydrocortisone (HC; Catalog #07904) to a final concentration of 0.48 μg/mL. Dissolved HC is stable at 2 - 8°C for only 7 days (freezing is not recommended).
- Aliquot Complete EpiCult™-B into appropriate volumes for use and supplement with HC immediately before use.
1.2 Preparation of EpiCult™-C Medium
- Prepare Complete EpiCult™-C Medium by adding 5 mL of thawed EpiCult™-C Supplement (Catalog #05632) to 500 mL of EpiCult™-C Basal Medium (Catalog #05631).
- Prepare Hydrocortisone as in Section 1.1 and use to supplement Complete EpiCult™-C Medium immediately before use.
Note: EpiCult™-B Basal Medium and EpiCult™-C Basal Medium contain the labile amino acid L-glutamine which has a half-life of approximately 1 month when stored at 2 - 8°C. Medium stored for greater than 2 months following the date of manufacture should be supplemented with additional L-glutamine. Add 1 mL of 200 mM L-glutamine (Catalog #07100) to 100 mL of basal medium to achieve a final concentration of 2 mM.
2.0 Dissociation of Human Mammary Tissue Using EpiCult™-B Medium (Human)
Note: Avoid the use of glass pipettes and tubes when handling mammary epithelial cells as these cells will stick to the glass.
2.1 Dissociation of Human Mammary Tissue
- Transport human mammary tissue from the operating room on ice in sterile specimen cups in Complete EpiCult™-B Medium supplemented with 5% fetal bovine serum (FBS; Catalog #06100).
- Transfer the tissue to sterile glass petri dishes, mince with scalpels and then transfer to tissue dissociation flasks (Catalog #27300). Glass petri dishes can be used for this initial dissociation, as the concentration of epithelial cells is very low.
- Dilute 1 part 10X Collagenase/Hyaluronidase (Catalog #07912) with 9 parts Complete EpiCult™-B Medium and add to the minced tissue in the dissociation flasks. Ensure that the tissue is well suspended in the enzyme mixture and the final volume is level with the widest portion of the flask. Cover the opening of the flask with sterile aluminum foil.
- Gently dissociate the minced tissue on a rotary shaker at 37°C until all large tissue fragments are digested. Typical digestion time is 16 hours (overnight) for normal human mammary tissue. Longer digestion times may be required for tough fibrous tissue, shorter digestion times for softer tissue. The flasks should be sealed if the rotary shaker is not in a 5% CO2 incubator.
- After dissociation, transfer the dissociated tissue to 50 mL centrifuge tubes, and centrifuge for 30 seconds at 80 x g.
- Discard the overlying liquefied fat layer. The pellet (‘A’ pellet) is highly enriched for epithelial organoids. To generate a single cell suspension from the ‘A’ pellet, refer to Section 2.2.
- Transfer the supernatant to a new 50 mL centrifuge tube and centrifuge at 200 x g for 3 minutes. The pellet (‘B’ pellet) from this second centrifugation contains variable numbers of epithelial cells, stromal cells and red blood cells. To generate a single cell suspension from the ‘B’ pellet, refer to Section 2.2.
- The supernatant from the second centrifugation is enriched for human mammary fibroblasts. To collect, transfer the supernatant to a new 50 mL centrifuge tube and centrifuge at 350 x g for 5 minutes.
- The different cell fractions can now be cryopreserved. It is recommended that cells are cryopreserved in Complete EpiCult™-B Medium supplemented with 50% FBS (Catalog #06100) and 6% Dimethyl Sulfoxide.
2.2 Generation of Single Cell Suspensions from Dissociated Human Mammary Tissue
- Add 1 - 5 mL of pre-warmed Trypsin-EDTA (Catalog #07901) to the Collagenase/Hyaluronidase-dissociated mammary cells and resuspend cells. The best starting material is ‘A’ pellets, however, ‘B’ pellets may also be used. The success of the cultures derived from ‘B’ pellets is more variable due to the variable epithelial content.
- Gently pipette up and down with a P1000 micropipettor for 1 - 3 minutes. The sample should become very stringy due to lysis of dead cells and the release of DNA.
- Add 10 mL of cold Hanks’ Balanced Salt Solution Modified (Catalog #37150) supplemented with 2% FBS (Catalog #06100) and centrifuge at 350 x g for 5 minutes. The Hanks’ + FBS solution is now referred to as HF.
- Remove as much of the supernatant as possible. The cells may be a large ‘stringy mass’ floating in the HF.
- Add 2 mL of pre-warmed 5 mg/mL Dispase (Catalog #07913) and 200 μL of 1 mg/mL DNase I (Catalog #07900). Pipette the sample for one minute with a P1000 micropipettor to further dissociate cell clumps. The sample should now be cloudy, but not stringy. If still stringy, add more DNase I.
- Dilute the cell suspension with an additional 10 mL of cold HF and filter the cell suspension through a 40 μm cell strainer (Catalog #27305) into a new 50 mL centrifuge tube. Centrifuge at 350 x g for 5 minutes and discard the supernatant.
- If the cell pellet is heavily contaminated with red blood cells, resuspend the pellet in a 1:4 mixture of cold HF:ammonium chloride (NH4Cl; Catalog #07800), centrifuge at 350 x g for 5 minutes and discard the supernatant.
3.0 Culture of Human Mammary Epithelial Cells Using EpiCult™-B or EpiCult™-C Medium (Human)
Human mammary epithelial cell cultures should be initiated from single cell suspensions (refer to Section 2.2). Otherwise, cells will not adhere well to the tissue culture flask.
Enhanced growth of human mammary cells can be achieved by pre-coating the tissue culture dish with a thin film of bovine collagen (Catalog #04902).
- Seed human mammary cells into tissue culture flasks at a density of 1 - 5 x 104 cells/cm2 in either Complete EpiCult™-B Medium or Complete EpiCult™-C Medium, both supplemented with 5% FBS (Catalog #06100).
Failure to include serum during plating of mammary cells will result in poor adherence of the cells to the tissue culture plastic. - After 24 hours, change the culture medium to either Complete EpiCult™-B Medium or Complete EpiCult™-C Medium, without
Failure to change to serum-free medium could result in overgrowth of the culture by contaminating stromal cells. - Mammary epithelial cells can be sub-cultured by first washing the adherent cells with Hanks’ Balanced Salt Solution Modified (Catalog #37150) followed by incubation with pre-warmed Trypsin-EDTA (Catalog #07901). Once the cells have detached from the culture vessel, an equal volume of cold HF (refer to Section 2.2, Step 3) should be added and the cell suspension centrifuged at 350 x g. Collected cells can then be reseeded into tissue culture as described above in Steps 1 and 2.
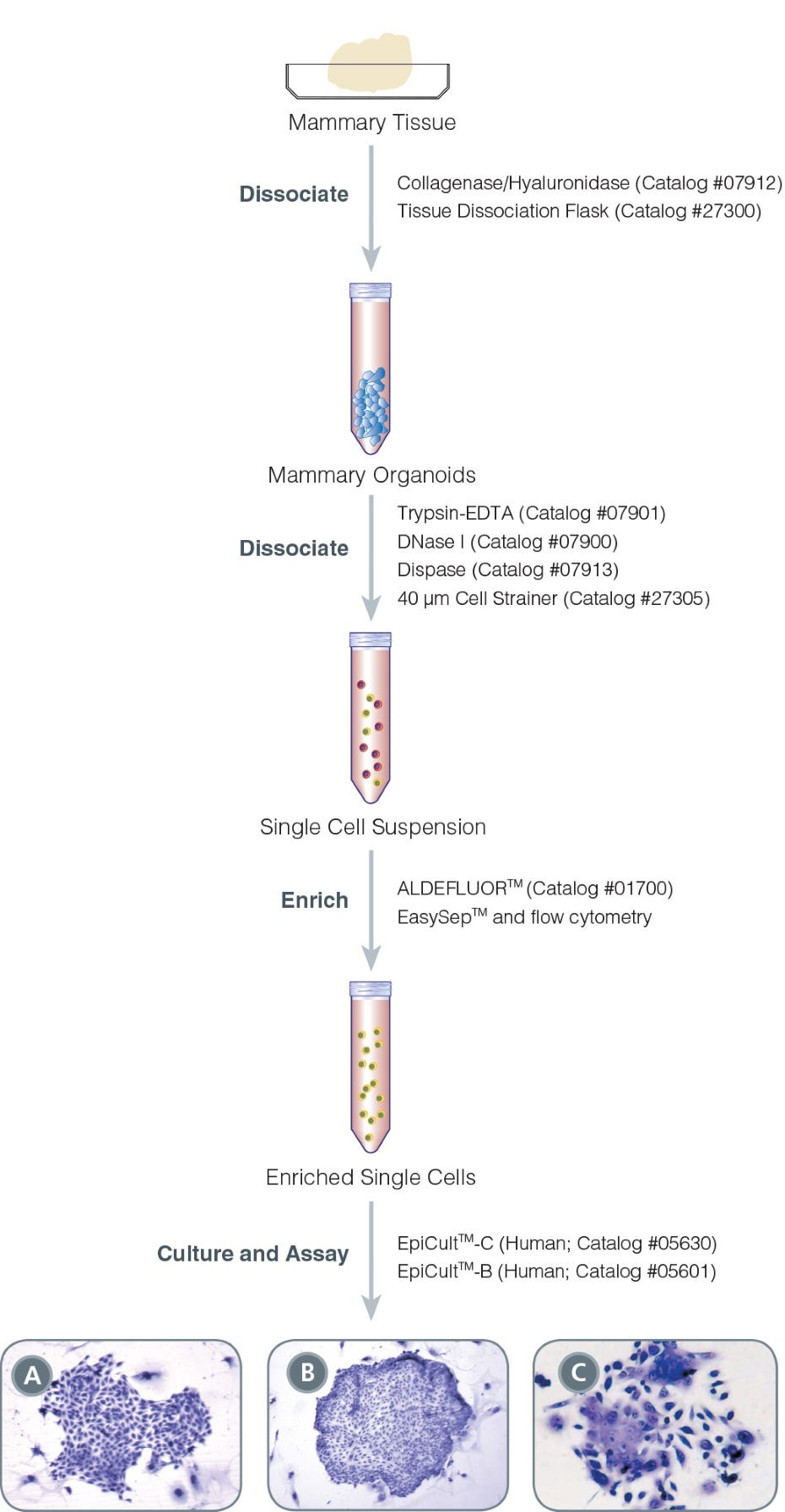
Figure 1. Protocol for Isolation and Identification of Human Mammary Progenitor Epithelial Cells
Phase contrast photographs of (A) a pure human myoepithelial cell colony, (B) a pure human luminal cell colony and (C) a mixed human colony. Colonies were obtained in EpiCult™-B (Human); all were cultured in the presence of an irradiated NIH 3T3 feeder layer.
Detect Human Mammary Colony-Forming Units

RECOMMENDED FOR:
Culture of human mammary luminal and myoepithelial cells. Promotes the clonogenic growth of bipotent, luminal-restricted and myoepithelial-restricted human mammary epithelial progenitors when used in conjunction with an irradiated feeder layer (NIH 3T3). Also used for the enzymatic dissociation of human mammary tissue when supplemented with Collagenase/Hyaluronidase (Catalog #07912).
CONTAINS:
- EpiCult™-B Basal Medium (Human)
- EpiCult™-B Supplement (Human)
REQUIRES:
Supplement with 0.48 μg/mL freshly dissolved Hydrocortisone (Catalog #07904) before use.
Culture Human Mammary Epithelial Cells

RECOMMENDED FOR:
Routine monolayer culture of primary human mammary epithelial cells and a variety of breast cancer cell lines.
CONTAINS:
- EpiCult™-C Basal Medium (Human)
- EpiCult™-C Proliferation Supplements (Human)
REQUIRES:
Supplement with 0.48 μg/mL freshly dissolved Hydrocortisone (Catalog #07904) before use.
Support Reagents
Collagenase/Hyaluronidase

RECOMMENDED FOR:
Enzymatic dissociation of human and mouse mammary tissue into mammary organoids. Enzymes are supplied at a 10X concentration.
CONTAINS:
- 10X stock solution containing 3000 U/mL collagenase and 1000 U/mL hyaluronidase in Dulbecco’s Modified Eagle’s Medium (1000 mg D-Glucose/L).
Other Dissociation Enzymes

RECOMMENDED FOR:
Prevention or reduction of clumping of human and mouse cells during dissociation.
CONTAINS:
- Dispase: 5 mg/mL in Hanks’ Balanced Salt Solution Modified
- DNAse I: 1 mg/mL in PBS
- Trypsin-EDTA: 2.5g/L porcine trypsin (tested for porcine parvo virus) and 1 mM EDTA•4Na in Hanks’ Balanced Salt Solution (Ca++ and Mg++ free).
References
- Chepko G, et al. Tissue Cell 29: 239-253, 1997
- Chepko G, et al. J Mammary Gland Biol Neoplasia 4: 35-52, 1999
- Sleeman KE, et al. J Cell Biol 176: 19-26, 2007
- Stingl J, et al. Differentiation 63: 201-213, 1998
- Stingl J, et al. Breast Cancer Res Treat 67: 93-109, 2001
- Pechoux C, et al. Dev Biol 206: 88-99, 1999
- Clayton H, et al. Exp Cell Res 297: 444-460, 2004
- Hovey RC, et al. J Mammary Gland Biol Neoplasia 4: 53-68, 1999
- Yang J, et al. Cancer Lett 81: 117-127, 1994
- Proia DA, et al. Nat Protoc 1: 206-214, 2006
- Eirew P, et al. Nat Med 14: 1384-1389, 2008
- Ginestier C, et al. Cell Stem Cell 1: 555-567, 2007
- Eirew P, et al. Stem Cells 30: 344-348, 2012
- Dontu G, et al. Genes Dev 17: 1253-1270, 2003
- Reynolds BA, et al. Science 255: 1707-1710, 1992
- Rietze RL, et al. Nature 412: 736-739, 2001
- Sell S, et al. Lab Invest 70: 6-22, 1994
- Al-Hajj M, et al. Proc Natl Acad Sci U S A 100: 3983-3988, 2003
- Lim E, et al. Nat Med 15: 907-913, 2009
- Molyneux G, et al. Cell Stem Cell 7: 403-417, 2010
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration

