Intestinal Organoid Assays
With Contract Assay Services
Organoids provide a convenient and highly predictive model system for studying the effects of novel therapeutic compounds on the intestinal epithelium in vitro. Because organoids contain the different cell types of the intestinal epithelium and can be maintained in various states of differentiation, they provide a better predictor of gastrointestinal toxicity than Caco-2 cells that are frequently used to model the intestine during drug development programs. Founded in our expertise from developing IntestiCult™ organoid growth and differentiation media, the intestinal organoid assays offered by Contract Assay Services (CAS) at STEMCELL Technologies are optimized to provide clear read-outs that better represent the effects observed in vivo.
Partner with CAS for a cost-effective and time-saving way of adding relevance of organoid-based assays to your research.
Why Use Our Intestinal Organoid Services?
- Get clear and relevant read-outs with intestinal organoid-based assays performed using SOPs optimized by our in-house experts.
- Obtain results in a model system that retains the key features and major cell types of the intestinal epithelium.
- Analyze compounds in a human-specific model system that is more predictive of in vivo toxicity than Caco-2 cells.
- Supplement your data, or try pilot studies, without committing to culturing your own organoids in-house.
Advantages of Intestinal Organoids
Traditional model systems, such as immortalized cell lines and animal models, have been useful for evaluating new therapeutic compounds by providing convenient tools that can be used during preclinical screening. However, these models may not contain the cell types necessary for your experiments, or may be of limited relevance to human physiology. In contrast, organoids offer the convenience of an in vitro cell culture model with the additional complexity of other cell types and spatial arrangements. As such, organoids have been used as a highly predictive model in new therapeutic development and toxicity testing.
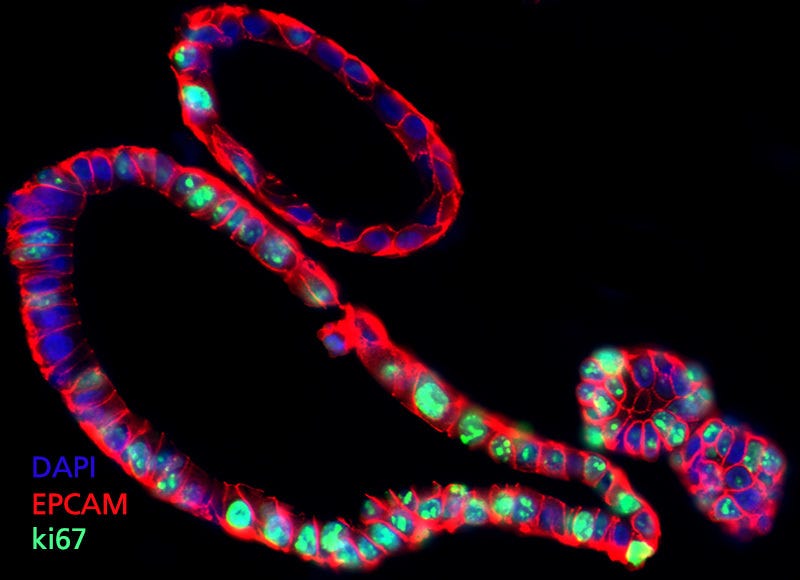
Figure 1. Intestinal Organoids Contain Actively Dividing Cells of the Intestinal Epithelium
Due to the presence of an actively dividing stem and progenitor cell population, human intestinal organoids provide a physiologically relevant model system for assessing intestinal effects of candidate therapeutic compounds. Organoids maintained in an undifferentiated state contain a high proportion of actively dividing cells as indicated by presence of the proliferation marker ki67 (green). This allows compounds to be tested for activity against genetically normal intestinal stem cells in a human-specific model system. Organoids shown are co-stained for EPCAM (red) and DAPI (blue).
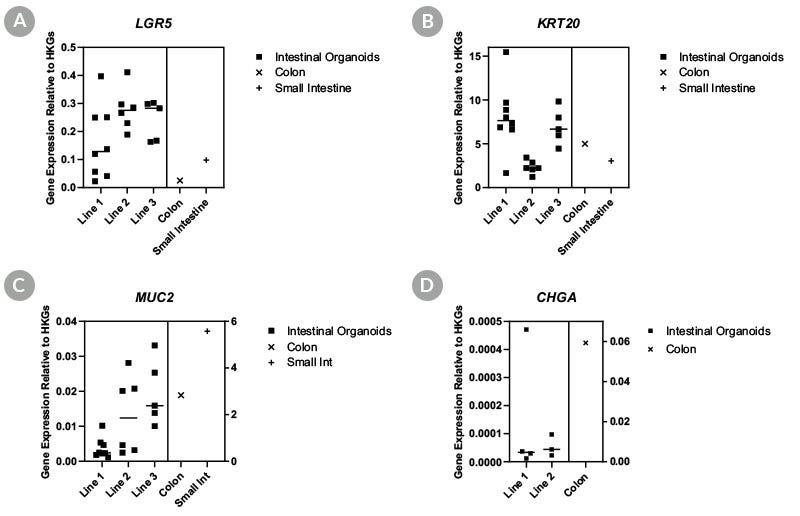
Figure 2. Intestinal Organoids Show Similar Genetic Expression Profiles to Primary Tissue for Key Intestinal Markers
Human intestinal organoids display similar genetic expression profiles to primary colonic and intestinal tissue. Organoids analyzed by qPCR for expression of (A) LGR5, (B) KRT20, (C) MUC2, and (D) CHGA show comparable expression to primary tissues demonstrating recapitulation of the intestinal epithelium.
Book a consultation for more information on assays using intestinal organoids >
Intestinal Cytotoxicity Testing
Gastrointestinal toxicity is often one of the dose-limiting factors for new therapeutics. However, compounds that interfere with normal cell growth may not show the same effects when examined in immortalized cell lines or animal model systems. By maintaining a population of actively dividing stem cells, as well as other differentiated cell types of the human intestine, intestinal organoids are an accurate predictor of intestinal toxicity and provide results that are more predictive of the in vivo response than Caco-2 cells, which are a popular model system for assessing gastrointestinal toxicity.
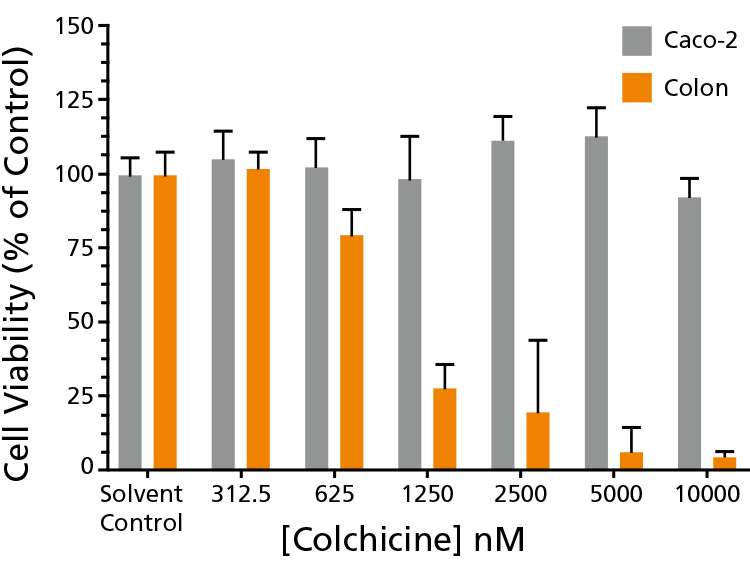
Figure 3. Intestinal Organoids Are a More Sensitive Indicator of Intestinal Toxicity Than Caco-2 Cells
Intestinal organoids provide a better predictor of intestinal cytotoxicity in actively dividing cells. Intestinal organoids were grown from different regions of the intestine, and assayed against compounds that primarily affect the growth and cellular division of the intestinal epithelium. Fully grown organoids treated with Colchicine demonstrate a higher toxicity than is observed in Caco-2 cells, which show no sensitivity to Colchicine.
Contact us for more information on intestinal cytotoxicity testing >
Toxicity Testing in Mouse Intestinal Organoids
Mouse intestinal organoids allow you to easily supplement your data without having to sacrifice additional animals. Because mouse intestinal organoids contain the different cell types of the intestinal epithelium, they provide a convenient model to add additional relevance to your research.
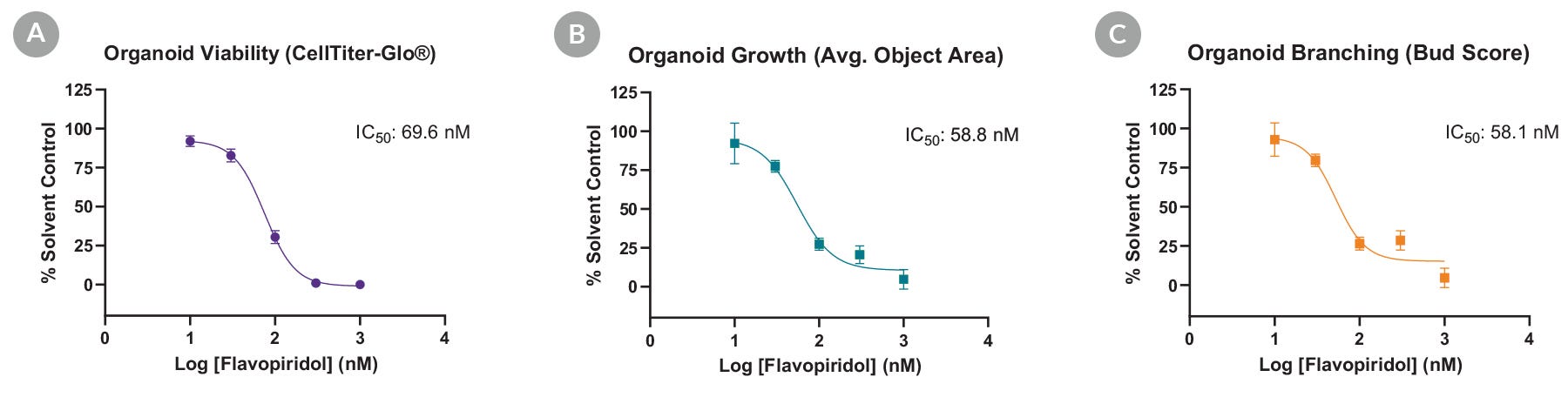
Figure 4. Flavopiridol Toxicity Assessment in Mouse Intestinal Organoids
Intestinal epithelial toxicity of flavopiridol is demonstrated by measuring the effects of the compound on (A) cell viability, (B) total organoid area, and (C) organoid morphology measured by the degree of organoid branching.
Request more information about assays using mouse intestinal organoids >
Intestinal Barrier Integrity Testing
In addition to interfering with normal turnover of the intestinal epithelium, therapeutic compounds can disrupt the barrier function of the intestinal epithelium. Because organoid-derived intestinal monolayers contain the different cell types of the intestinal epithelium, they provide a convenient system for assessing disruption of the intestinal barrier function in vitro.
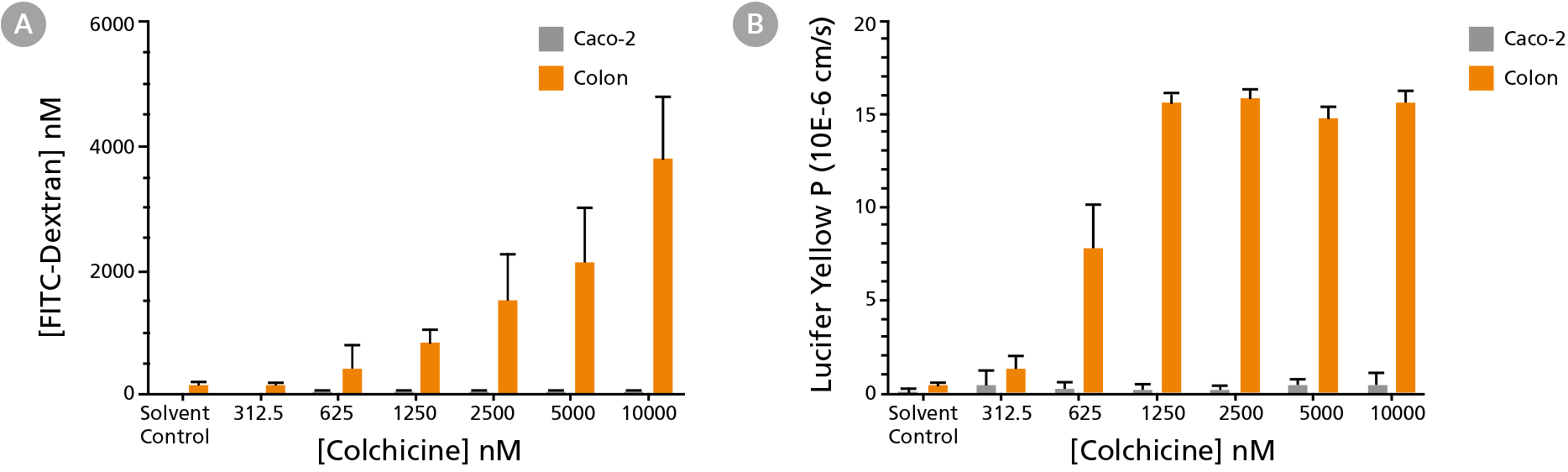
Figure 5. Intestinal Organoids Detect Barrier-Function Disruption Missed By Caco-2 Cells
Organoids are able to detect the barrier function disruption caused by Colchicine as measured by permeability to (A) FITC-Dextran and (B) Lucifer Yellow. This barrier disruption is not detected in Caco-2 cells.
Contact us for more information on intestinal barrier function testing >


