STEMdiff™ Monocyte Kit
For differentiation of human pluripotent stem cells (hPSCs) to monocytes
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration
-
 mTeSR™1
mTeSR™1cGMP, feeder-free maintenance medium for human ES and iPS cells
-
Labeling Antibodies
Compatible antibodies for purity assessment of isolated cells
Overview
The simple protocol is performed in 2D adherent cultures. During the first 3 days, Medium A induces cells toward mesoderm. For the subsequent 4 days, mesodermal cells are further differentiated toward the hematopoietic lineage using Medium B. At Day 7, the medium is changed to Monocyte Differentiation Medium, which facilitates the differentiation to monocytes. CD14+ monocytes can be harvested directly from the culture supernatant starting as early as Day 14 and can be repeatedly harvested during the rest of the culture period. Peak CD14+ frequency is typically 60 - 80%.
The hPSC-derived monocytes can be further differentiated to dendritic cells or macrophages using the ImmunoCult™ Dendritic Cell Culture Kit or the ImmunoCult™-SF Macrophage Medium, respectively.
For your convenience, components required for the preparation of the Monocyte Differentiation Medium, StemSpan™ SFEM II and STEMdiff™ Monocyte Differentiation Supplement (100X), are also available for purchase individually.
Data Figures
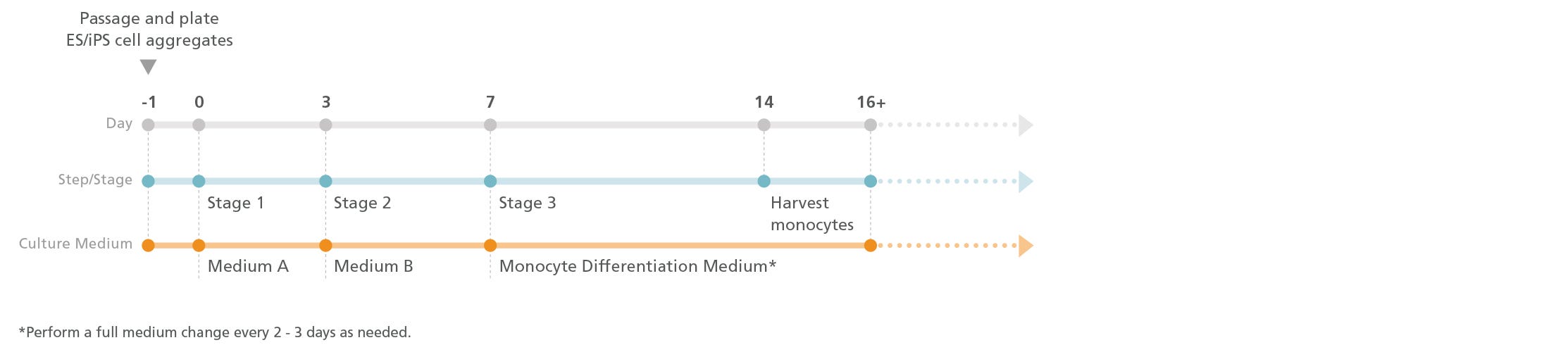
Figure 1. Monocyte Differentiation Protocol
One day prior to differentiation, human pluripotent stem cell (hPSC) colonies are harvested and seeded as small aggregates (100 - 200 μm in diameter) at 10 - 20 aggregates/cm2 in mTeSR™1, TeSR™-E8™, or mTeSR™ Plus media. After one day, the medium is replaced with Medium A (STEMdiff™ Hematopoietic Basal Medium + Supplement A) to induce mesodermal specification (stage 1). On day 3, the medium is changed to Medium B (STEMdiff™ Hematopoietic Basal Medium + Supplement B) to promote hematopoietic specification (stage 2). On day 7, the medium is replaced with Monocyte Differentiation Medium (StemSpan™ SFEM II + STEMdiff™ Monocyte Differentiation Supplement) to promote the production of CD14+ monocytes (stage 3). Monocyte Differentiation Medium is used for all medium changes for the remaining culture period. CD14+ cells can be detected in suspension starting after day 14, and their frequency gradually increases until day 17 - 23. CD14+ cells can be harvested directly from the culture supernatant during medium changes.
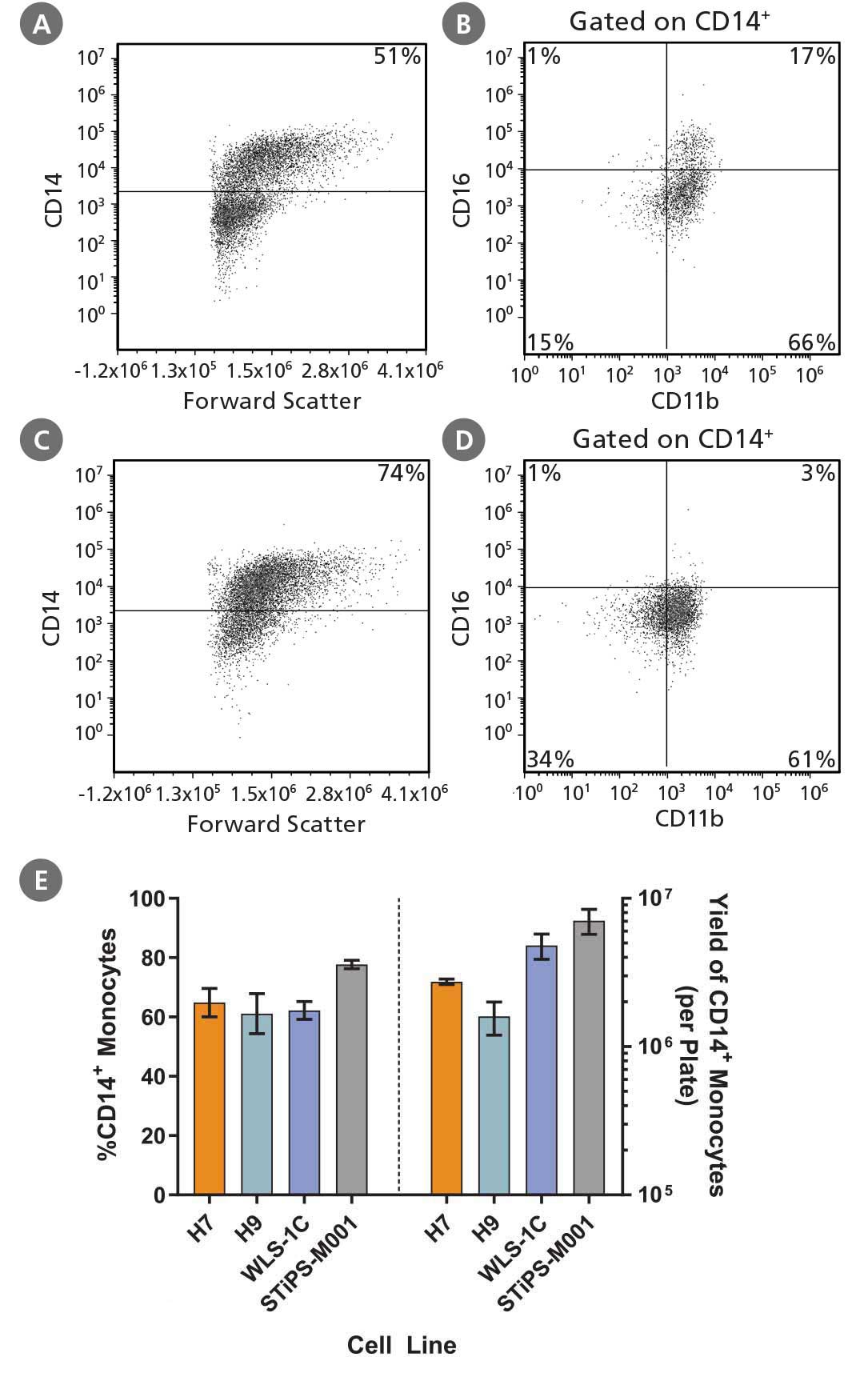
Figure 2. Robust and Efficient Generation of CD14⁺ Monocytes Using STEMdiff™ Monocyte Kit
hPSCs were differentiated to monocytes using the 2D culture system described in Figure 1. Between days 17 and 23, cells were harvested every 2 - 3 days and analyzed by flow cytometry for CD14 expression. Representative flow cytometry plots are shown for (A, B) iPS (WLS-1C)-derived cells and (C, D) ES (H9)-derived cells. (E) The average frequency of viable CD14+ monocytes at the peak harvest was 61 - 78%. The average yield of CD14+ monocytes produced per 6-well plate at the peak harvest was between 1.6 x 10^6 and 7.1 x 10^6 cells. Data are shown as mean ± SEM (n = 3 - 14).
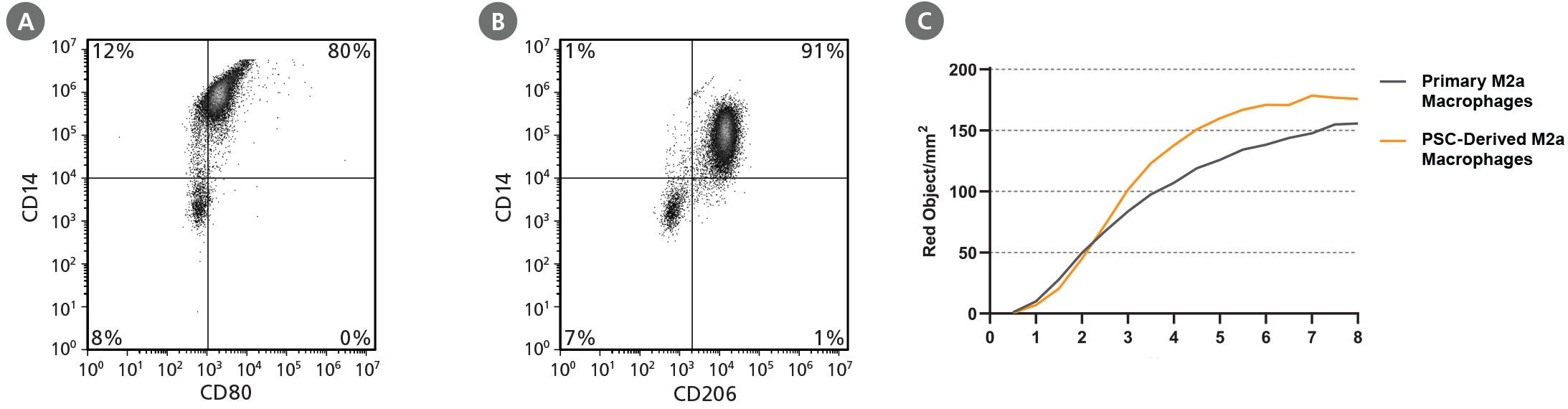
Figure 3. STEMdiff™ Monocyte Kit Generates Monocytes That Are Capable of Differentiation to Macrophages
hPSC-derived monocytes were harvested after 21 days of culture. These were then differentiated to macrophages using ImmunoCult™-SF Macrophage Medium (Catalog #10961) with 100 ng/mL M-CSF for 4 days. Macrophages were then incubated for an additional 2 days with either 10 ng/mL of LPS and 50 ng/mL of IFN-γ, or 10 ng/mL IL-4, to become polarized to M1 or M2a macrophages, respectively. Representative flow cytometry plots of (A) M1 and (B) M2a macrophages produced from the WLS-1C iPS cell line are shown. (C) To measure phagocytosis, PSC-derived M2a macrophages and peripheral blood (PB) monocyte-derived M2a macrophages (primary M2a macrophages), were incubated with pHrodo™ Red Zymosan A BioParticles® Conjugate and incubated at 37°C for 8 hours. Images were acquired using the IncuCyte® ZOOM every 30 minutes and analyzed for internalization of pHrodo™ Red Zymosan A BioParticles® (measured as red object/mm2). hPSC-derived and primary M2a macrophages show similar phagocytic activity.
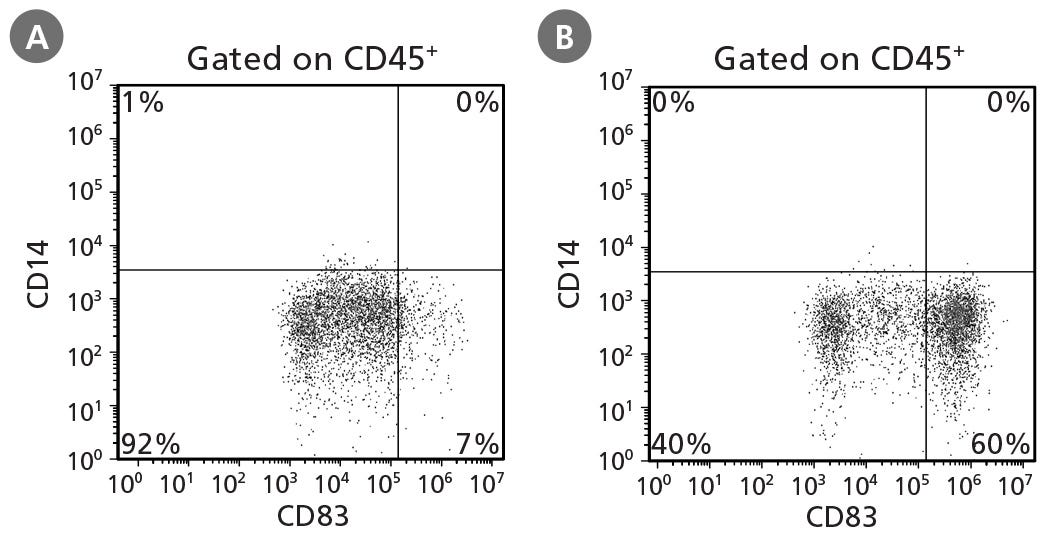
Figure 4. STEMdiff™ Monocyte Kit Generates Monocytes That Can Be Differentiated to Dendritic Cells
hPSCs were differentiated into monocytes, harvested after 21 days, and differentiated to dendritic cells using ImmunoCult™ Dendritic Cell Culture Kit (Catalog #10985). Half of the dendritic cells were harvested on day 7 and examined for CD14 and CD83 expression to identify CD14⁻CD83⁻/lo immature dendritic cells. The remaining dendritic cells were activated for 2 days and assessed for the presence of CD14⁻CD83⁺ mature dendritic cells at day 7. Representative cultures initiated with ES (H9) cells are shown for production of (A) immature dendritic cells and (B) mature dendritic cells.
Protocols and Documentation
Find supporting information and directions for use in the Product Information Sheet or explore additional protocols below.
Applications
This product is designed for use in the following research area(s) as part of the highlighted workflow stage(s). Explore these workflows to learn more about the other products we offer to support each research area.
Resources and Publications
Educational Materials (10)
Related Products
-
 STEMdiff™ Monocyte Differentiation Suppleme...
STEMdiff™ Monocyte Differentiation Suppleme...Supplement for the differention to CD14+ monocytes
-
 ImmunoCult™-SF Macrophage Medium
ImmunoCult™-SF Macrophage MediumSerum-free medium for differentiation of human monocytes to macrophages
-
 ImmunoCult™ Dendritic Cell Culture Kit
ImmunoCult™ Dendritic Cell Culture KitComplete kit for differentiation of human monocytes into dendritic cells
Item added to your cart

STEMdiff™ Monocyte Kit
Quality Statement:
PRODUCTS ARE FOR RESEARCH USE ONLY AND NOT INTENDED FOR HUMAN OR ANIMAL DIAGNOSTIC OR THERAPEUTIC USES UNLESS OTHERWISE STATED. FOR ADDITIONAL INFORMATION ON QUALITY AT STEMCELL, REFER TO WWW.STEMCELL.COM/COMPLIANCE.