EasySep™ Direct Human B-CLL Cell Isolation Kit
Immunomagnetic negative selection from whole blood kit

Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration
Overview
This cell isolation kit targets non-B cells for removal based on specific surface antigens. Unwanted cells are labeled with antibodies and EasySep™ Direct RapidSpheres™, and separated using an EasySep™ magnet. Desired cells are simply collected into a new tube and can immediately be used in downstream applications such as nucleic acid extraction, flow cytometry or culture.
Data Figures
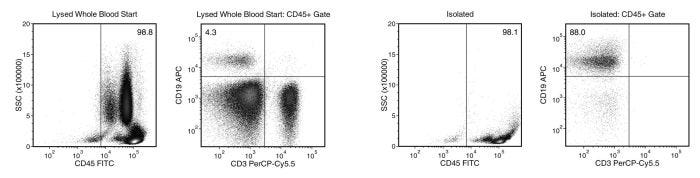
Figure 1. Typical EasySep™ Direct Human B-CLL Cell Isolation Profile
Starting with human whole blood from normal healthy donors, the typical B cell (CD3-CD19+) content of the non-lysed final isolated fraction is 87.0 ± 7.6% (gated on CD45) or 84.1 ± 11.7% (not gated on CD45). In the example above, the B cell (CD3-CD19+) content of the lysed whole blood start and non-lysed final isolated fraction is 4.3% and 88.0% (gated on CD45), respectively, or 4.3% and 86.2% (not gated on CD45), respectively. The starting frequency of B cells in the non-lysed whole blood start sample above is 0.004% (data not shown). The purity of isolated cells is typically higher when processing samples that have an elevated starting frequency of B cells (e.g. CLL samples).
Protocols and Documentation
Find supporting information and directions for use in the Product Information Sheet or explore additional protocols below.
Applications
This product is designed for use in the following research area(s) as part of the highlighted workflow stage(s). Explore these workflows to learn more about the other products we offer to support each research area.
Resources and Publications
Educational Materials (15)
Frequently Asked Questions
Can EasySep™ be used for either positive or negative selection?
How does the separation work?
Which columns do I use?
How can I analyze the purity of my enriched sample?
Can EasySep™ separations be automated?
Can EasySep™ be used to isolate rare cells?
Are the EasySep™ magnetic particles FACS-compatible?
Can the EasySep™ magnetic particles be removed after enrichment?
Can I alter the separation time in the magnet?
For positive selection, can I perform more than 3 separations to increase purity?
How does the binding of the EasySep™ magnetic particle affect the cells? is the function of positively selected cells altered by the bound particles?
If particle binding is a key concern, we offer two options for negative selection. The EasySep™ negative selection kits can isolate untouched cells with comparable purities, while RosetteSep™ can isolate untouched cells directly from whole blood without using particles or magnets.
Related Products
Labeling Antibodies
-
 Anti-Human CD19 Antibody, Clone HIB19
Anti-Human CD19 Antibody, Clone HIB19Mouse monoclonal IgG1 antibody against human, chimpanzee CD19
-
 Anti-Human CD20 Antibody, Clone 2H7
Anti-Human CD20 Antibody, Clone 2H7Mouse monoclonal IgG2b antibody against human, rhesus, cynomolgus CD20
-
 Anti-Human CD45 Antibody, Clone HI30
Anti-Human CD45 Antibody, Clone HI30Mouse monoclonal IgG1 antibody against human, chimpanzee CD45
-
 Anti-Human CD5 Antibody, Clone UCHT2
Anti-Human CD5 Antibody, Clone UCHT2Mouse monoclonal IgG1 antibody against human, rhesus, cynomolgus CD5
Item added to your cart

EasySep™ Direct Human B-CLL Cell Isolation Kit
Quality Statement:
PRODUCTS ARE FOR RESEARCH USE ONLY AND NOT INTENDED FOR HUMAN OR ANIMAL DIAGNOSTIC OR THERAPEUTIC USES UNLESS OTHERWISE STATED. FOR ADDITIONAL INFORMATION ON QUALITY AT STEMCELL, REFER TO WWW.STEMCELL.COM/COMPLIANCE.
















