STEMdiff™ Myogenic Progenitor Supplement Kit
Serum-free supplements for differentiation of human pluripotent stem cells (hPSCs) to myogenic progenitor cells
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration
-
 Collagenase Type IV (1 mg/mL)
Collagenase Type IV (1 mg/mL)Cell dissociation reagent
-
 D-PBS (Without Ca++ and Mg++)
D-PBS (Without Ca++ and Mg++)Dulbecco’s phosphate-buffered saline without calcium and magnesium
-
 Y-27632 (Dihydrochloride)
Y-27632 (Dihydrochloride)RHO/ROCK pathway inhibitor; Inhibits ROCK1 and ROCK2
-
 mTeSR™1
mTeSR™1cGMP, feeder-free maintenance medium for human ES and iPS cells
-
Labeling Antibodies
Compatible antibodies for purity assessment of isolated cells
Overview
Data Figures

Figure 1. Schematic of Myogenic Induction Using the STEMdiff™ Myogenic Progenitor Supplement Kit
Prior to differentiation (Day -1), human pluripotent stem cells (hPSCs) are plated in Corning® Matrigel®-coated plates. On Day 0, differentiation is initiated and continued by performing daily media changes with STEMdiff™ Myogenic Progenitor Supplement A at Days 0 - 2, STEMdiff™ Myogenic Progenitor Supplement B at Days 2 - 4, STEMdiff™ Myogenic Progenitor Supplement C at Days 4 - 6, and STEMdiff™ Myogenic Progenitor Supplement D for the remainder of the protocol (Days 6 - 30). Upon completion of the 30-day protocol, the culture contains an abundance of PAX7+MyoD+ myogenic progenitors that can be further expanded using MyoCult™-SF Expansion Supplement Kit (Human; Catalog #05980) for use in downstream applications.
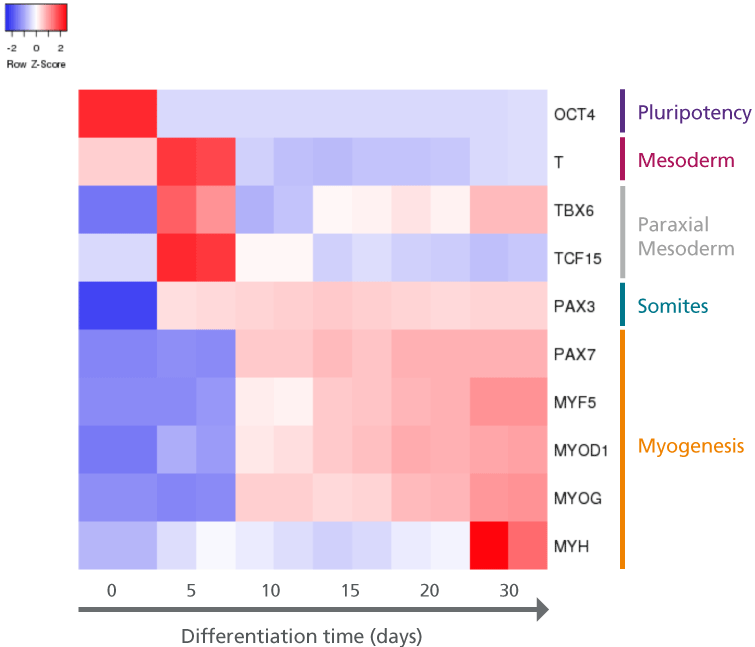
Figure 2. hPSCs Differentiated Using STEMdiff™ Myogenic Progenitor Kit Demonstrate Transcript Patterns of Embryonic Muscle Development
hPSCs (H9 cell line) were differentiated using the STEMdiff™ Myogenic Progenitor Kit and harvested at indicated time points for transcript analysis via qPCR; transcription was normalized against 18s as the housekeeping gene. The resulting heatmap indicates transcriptional changes to genes that mark specific stages of muscle development (mesoderm, paraxial mesoderm, somites, and myogenesis) and demonstrates a cellular trajectory that resembles embryonic muscle development.
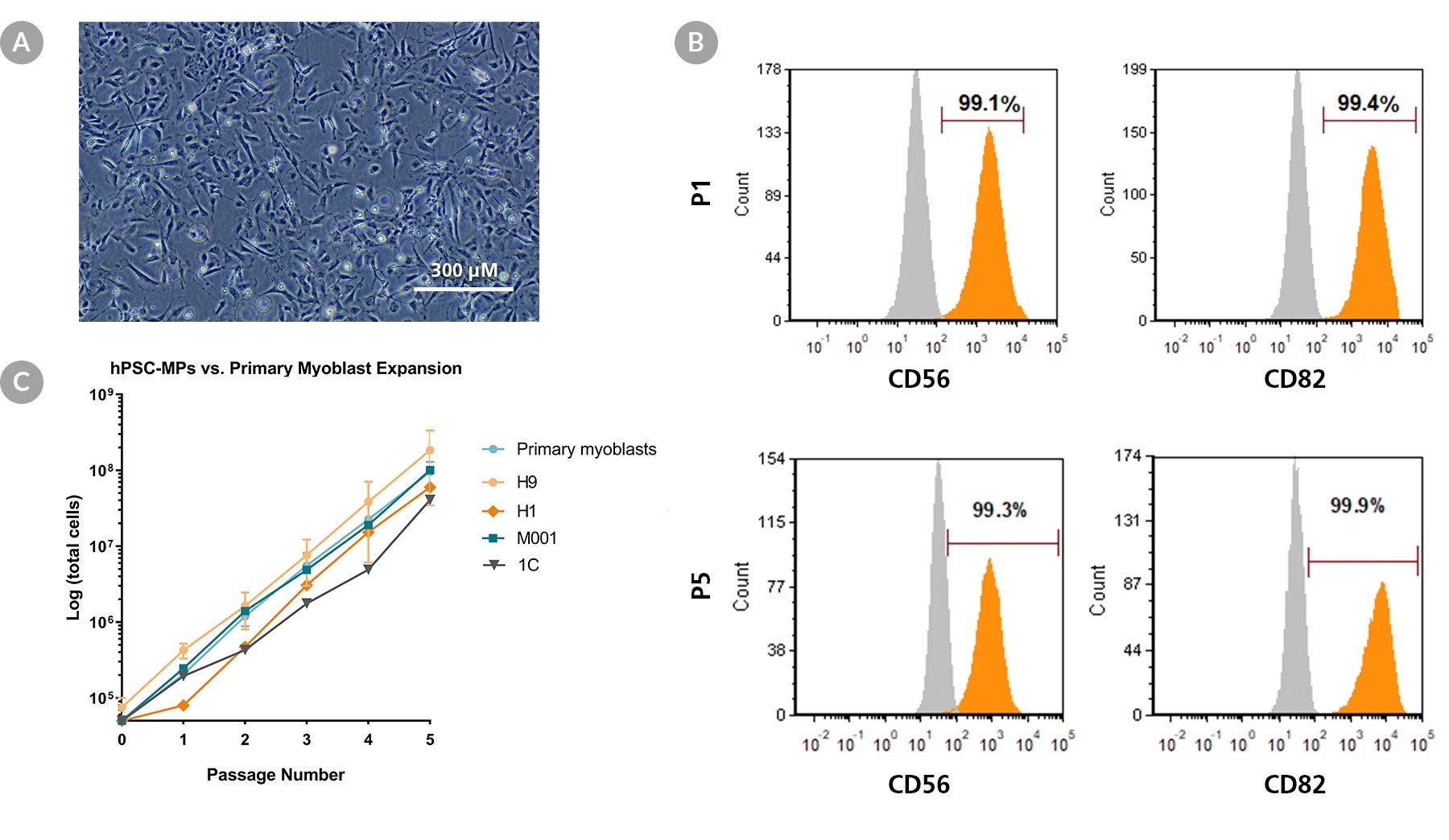
Figure 3. STEMdiff™ Myogenic Progenitor Kit Generates Expandable hPSC-Derived Myogenic Progenitors
(A) Representative image of proliferating sub-cultured hPSC-derived myogenic progenitors generated using the STEMdiff™ Myogenic Progenitor Kit. (B) hPSC-derived myogenic progenitors harvested at passage 1 and 5 expressed human myoblast markers CD56 and CD82. (C) Expansion rates of hPSC-derived myogenic progenitors (hSPC-MP) over 5 passages across multiple hPSC lines are comparable to human primary myoblasts. Error bars represent standard error of mean, n = 3.
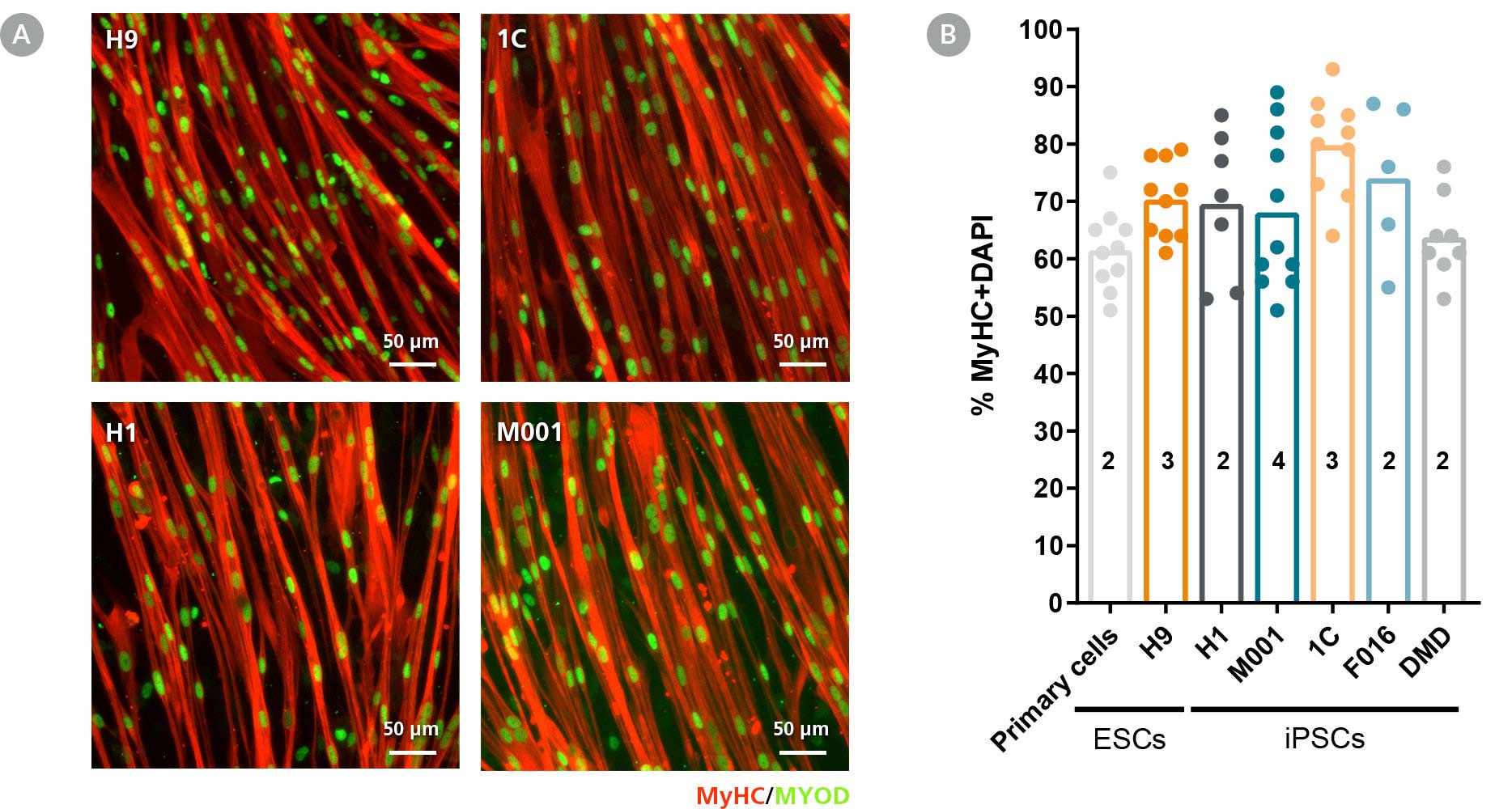
Figure 4. STEMdiff™ Myogenic Progenitor Kit Facilitates Efficient Differentiation of hPSC-Derived Myogenic Progenitors into hPSC-Derived Myotubes
(A) hPSC-derived myogenic progenitors were generated from multiple hPSC lines using the STEMdiff™ Myogenic Progenitor Kit and then induced to differentiate into myotubes using MyoCult™ Differentiation Medium (Human; Catalog #05965). After 8 days, myotubes were fixed and stained for MyHC and MyoD. (B) Multiple hPSC cell lines differentiated and induced using said method exhibited high fusion indices similar to primary human myoblasts (Numbers in bars represent the n number and dots represent technical replicates).
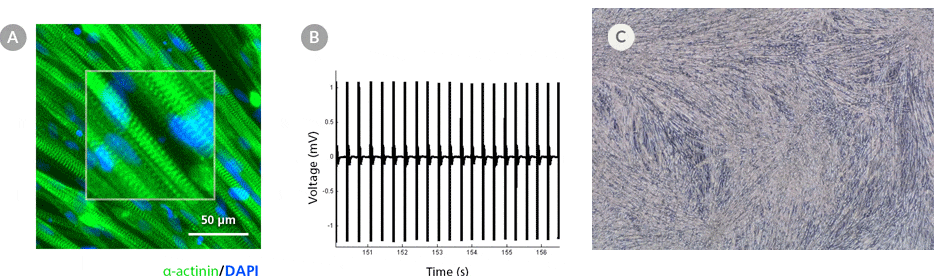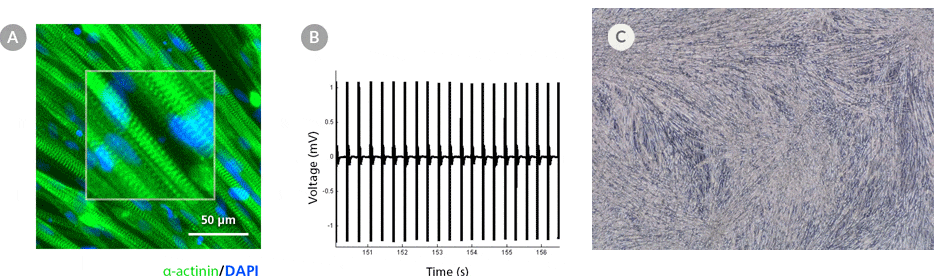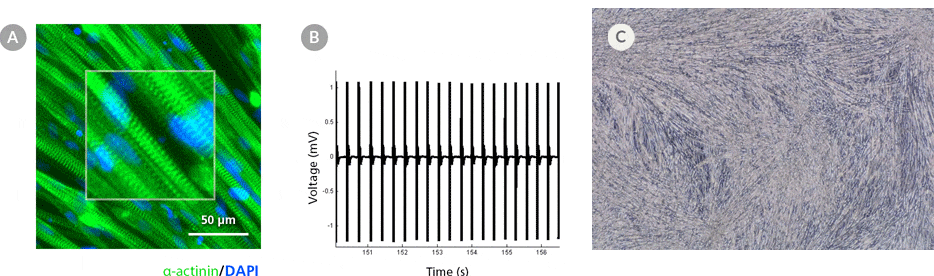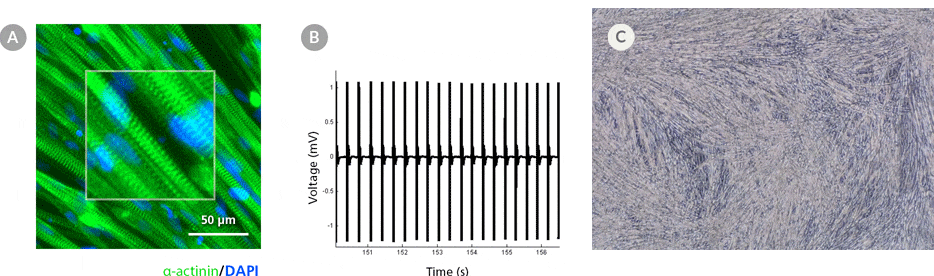
Figure 5. hPSC-Derived Myotubes Generated Using STEMdiff™ Myogenic Progenitor Kit Are Functionally Contractile
hPSC-derived myogenic progenitors were generated using the STEMdiff™ Myogenic Progenitor Kit and then induced to differentiate into myotubes using MyoCult™ Differentiation Medium. (A) hPSC-derived myotubes were stained for alpha-actinin and displayed organized sarcomeric structures as indicated by the zoomed-in area. (B) Spontaneous field potential recordings of hPSC-derived myotubes using microelectrode assay plate indicated that the derived myotubes are contractile. (C) Representative video of contracting hPSC-derived myotubes.
Protocols and Documentation
Find supporting information and directions for use in the Product Information Sheet or explore additional protocols below.
Applications
This product is designed for use in the following research area(s) as part of the highlighted workflow stage(s). Explore these workflows to learn more about the other products we offer to support each research area.
Resources and Publications
Educational Materials (6)
Related Products
-
 MyoCult™ Differentiation Kit (Human)
MyoCult™ Differentiation Kit (Human)Medium for the differentiation of human skeletal muscle progenitor cells (myoblasts) into myotubes
-
 MyoCult™-SF Expansion Supplement Kit (Human...
MyoCult™-SF Expansion Supplement Kit (Human...Serum-free supplement and attachment substrate for the derivation and expansion of human skeletal muscle progenitor cells (myoblasts)
-
 Collagenase A, ACF
Collagenase A, ACFAnimal component-free collagenase for the digestion of native collagen fibrils
-
 CryoStor® CS10
CryoStor® CS10Animal component-free, defined cryopreservation medium with 10% DMSO
Item added to your cart

STEMdiff™ Myogenic Progenitor Supplement Kit
Quality Statement:
PRODUCTS ARE FOR RESEARCH USE ONLY AND NOT INTENDED FOR HUMAN OR ANIMAL DIAGNOSTIC OR THERAPEUTIC USES UNLESS OTHERWISE STATED. FOR ADDITIONAL INFORMATION ON QUALITY AT STEMCELL, REFER TO WWW.STEMCELL.COM/COMPLIANCE.