NaïveCult™ Induction Kit
Defined medium kit for transgene-free induction of reset naïve human ES and iPS cells
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration
-
 0.1% Gelatin in Water
0.1% Gelatin in WaterCoating for cultureware
-
 Sodium Butyrate
Sodium ButyrateEpigenetic modifier; Inhibits histone deacetylase
-
 Valproic Acid (Sodium Salt)
Valproic Acid (Sodium Salt)Epigenetic modifier; Inhibits histone deacetylase (HDAC)1
-
 Y-27632 (Dihydrochloride)
Y-27632 (Dihydrochloride)RHO/ROCK pathway inhibitor; Inhibits ROCK1 and ROCK2
-
Labeling Antibodies
Compatible antibodies for purity assessment of isolated cells
Overview
Reset naïve human pluripotent stem cells (hPSCs) generated using this kit demonstrate uniformly domed, phase-bright colonies with refractive edges and maintain gene expression profiles associated with reset naïve hPSCs (Guo et al. Stem Cell Reports, 2016; Guo et al. Development, 2017; Takashima et al. Cell, 2014).
Based on the formulation by Guo et al. (2017) and developed under license from the Cambridge Stem Cell Institute at the University of Cambridge, UK, this kit works robustly across multiple primed human ES and iPS cells maintained in mTeSR™1 (Catalog #85850) or TeSR™-E8™ (Catalog #05990). Note that NaïveCult™ Induction Kit requires a histone deacetylase inhibitor (HDACi; either Sodium Butyrate (Catalog #72242) or Valproic Acid (Catalog #72292)) that is sold separately.
Data Figures
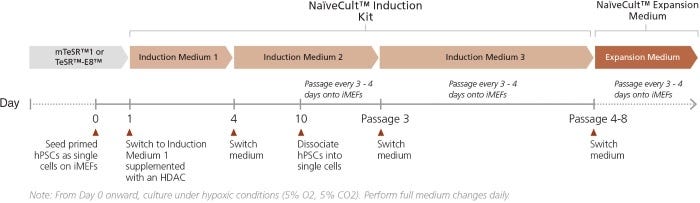
Figure 1. Schematic of Reversion of Primed to Naïve hPSCs
Primed hPSCs, grown either in mTeSR™1 or TeSR™-E8™ on Corning® Matrigel®, are plated as single cells on inactivated murine embryonic fibroblasts (iMEFs) and treated with Rho-kinase inhibition (10 μM Y-27632) for 24 hours in hypoxic conditions. On day 1, medium is changed to Induction Medium 1 supplemented with a histone deacetylase inhibitor (HDACi) and cells are cultured for 3 days with daily medium changes. On day 4, medium is changed to Induction Medium 2 and cells are cultured until day 11 with daily medium changes. On day 11, hPSCs are passaged as single cells onto fresh iMEFs and subsequently passaged every 3 - 4 days until the end of passage 2 in Induction Medium 2. From passage 3, hPSCs are cultured in Induction Medium 3. Background differentiation will decrease between passage 3 and 8. At this time cells can be transferred into NaïveCult™ Expansion Medium for long-term maintenance and expansion.
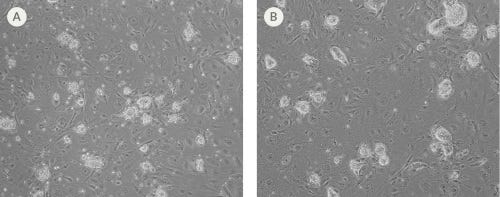
Figure 2. Human ES and iPS Cells Can Be Reverted to a Naïve State
Representative images of human (A) passage-7 H9 ES cells and (B) passage-9 WLS-1C iPS cells that were reverted to a naïve state using the NaïveCult™ Induction Kit and subsequently cultured in NaïveCult™ Expansion Medium. During reversion, colonies change from a flat morphology to a tightly packed, domed morphology with refractive edges characteristic of naïve-state hPSCs.
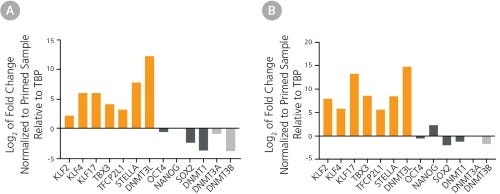
Figure 3. hPSCs Cultured in the NaïveCult™ Media System Express High Levels of Factors Associated with Naïve hPSCs
Gene expression profile of human (A) H9 ES cells and (B) WLS-1C iPS cells reverted using the NaïveCult™ Induction Kit and maintained in NaïveCult™ Expansion Medium.
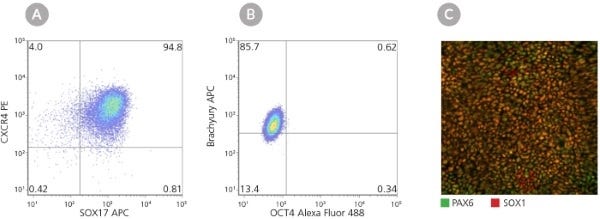
Figure 4. Reset Naïve Human PSCs Cultured in NaïveCult™ Expansion Medium are Capable of Tri-Lineage Differentiation Following Re-Priming
Reset naïve human iPS cells cultured in NaïveCult™ Expansion Medium were re-primed in mTeSR™1 and differentiated to all three somatic lineages. (A) Representative flow cytometry plot of human WLS-1C iPS cells differentiated using the STEMdiff™ Definitive Endoderm Kit (Catalog #05110) demonstrating >90% CXCR4+/SOX17+ definitive endodermal progenitors. (B) Representative flow cytometry plot of human WLS-1C iPS cells differentiated using STEMdiff™ Mesodermal Induction Medium (Catalog #05220) demonstrating >80% Brachyury+/OCT4- mesodermal progenitors. (C) Representative immunofluorescence image of PAX6 (green) and SOX1 (red) double positive neural progenitors derived from human WLS-1C iPS cells differentiated using the STEMdiff™ SMADi Neural Induction Kit (Catalog #08581).
Protocols and Documentation
Find supporting information and directions for use in the Product Information Sheet or explore additional protocols below.
Applications
This product is designed for use in the following research area(s) as part of the highlighted workflow stage(s). Explore these workflows to learn more about the other products we offer to support each research area.
Resources and Publications
Educational Materials (5)
Related Products
-
 NaïveCult™ Expansion Medium
NaïveCult™ Expansion MediumDefined cell culture medium for expansion of reset naïve human ES and iPS cells
-
 TeSR™-E8™
TeSR™-E8™Feeder-free, animal component-free culture medium for maintenance of human ES and iPS cells
-
 ACCUTASE™
ACCUTASE™Cell detachment solution
-
 mTeSR™1
mTeSR™1cGMP, feeder-free maintenance medium for human ES and iPS cells
Item added to your cart

NaïveCult™ Induction Kit
Legal Statement:
Developed under license from Cambridge Enterprise Limited
Quality Statement:
PRODUCTS ARE FOR RESEARCH USE ONLY AND NOT INTENDED FOR HUMAN OR ANIMAL DIAGNOSTIC OR THERAPEUTIC USES UNLESS OTHERWISE STATED. FOR ADDITIONAL INFORMATION ON QUALITY AT STEMCELL, REFER TO WWW.STEMCELL.COM/COMPLIANCE.