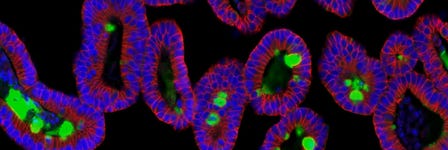
Critical Tips for Establishing and Maintaining Mouse Intestinal Organoid Culture
Technical tip from our dedicated team of Product and Scientific Support specialists
Mouse intestinal organoids can be established for long-term maintenance in IntestiCult™ Organoid Growth Medium (Mouse). Below are the critical tips to ensure successful isolation of mouse intestinal crypts and culture of the intestinal organoids. Keep these considerations in mind as you perform your procedures.
Looking for complete step-by-step protocols for culturing mouse intestinal organoids?
If you don’t have established procedures in your lab yet, start with our step-by-step protocols and videos for isolating mouse intestinal crypts, establishing intestinal organoid culture, and passaging mouse intestinal organoids.
General Tips:
- Throughout the procedure you will need to pre-wet pipettes and pipette tips before manipulating intestinal pieces or crypts. This will prevent the tissue from sticking to the wall of the pipette.
- To minimize cellular disruption and maximize recovery, use IntestiCult™ Organoid Growth Medium (Mouse) medium and Gentle Cell Dissociation Reagent (GCDR) at room temperature, whereas the PBS and DMEM/F12 used for cleaning and washing steps should be ice cold.
- Please refer to this video for an overview of intestinal organoid culture with IntestiCult™.
Isolation of Mouse Intestinal Crypts:
- When harvesting the intestine, remove the mesentery (membrane that attaches the intestine to the abdominal wall) prior to cutting the section of small intestine. If it is not removed first, it will be difficult to spin it out when the intestinal segments are being washed in subsequent steps.
- After cutting the intestine into 2 mm segments, 15 - 20 rounds of washing in PBS are necessary to make sure the intestinal segments are clean. If fewer than 15 rounds of washing are performed, there may be some toxic matter still left in the folds, which will negatively affect the growth of the organoids.
- It is important to let the intestinal pieces settle by gravity, as centrifugation during washing steps may result in pelleting of additional impurities, resulting in poor crypt recovery.
Plating Intestinal Crypts for Organoid Culture:
- Once the crypts have been isolated and are ready to be plated, it is critical to use pre-warmed plates and ice-cold Matrigel® to make sure that crypts stay in suspension. If crypts touch and stick down to the surface of the well, they will differentiate.
- When the drop of Matrigel® containing the intestinal crypts has set, add IntestiCult™ medium to the well along the side of the wall. If medium is added directly onto the drop of Matrigel®, the force of the liquid will disrupt the Matrigel® dome.
- The Matrigel® dome must be completely covered by the IntestiCult™ medium.
- It is important to plate crypts at 3 different densities. Both the medium and the organoids themselves produce factors that are necessary for expansion and survival of the organoids. If the crypts are seeded too densely, they will not receive enough nutrients from the medium for proper expansion. If very few crypts are seeded, there will not be enough factors produced by the organoids for proper expansion.
Passaging Intestinal Organoid Cultures:
- As organoids begin to bud, the mature epithelial cells shed into the lumen. Ensure that organoid cultures are passaged before the lumen gets too dark when examining them under the microscope.
- There are two components that break up the organoids during the passaging procedure: the time of incubation in GCDR and the manual agitation with the pipette. If the organoids are over-incubated in GCDR or too aggressively agitated, there will be more single cells, which is not desirable.
For further assistance and information, please contact techsupport@stemcell.com.
Tools and Technologies
IntestiCult™ Intestinal Organoid Culture Media
Establish, expand, maintain, and further differentiate intestinal organoid cultures from human or mouse intestinal crypts using IntestiCult™ complete and defined growth media.
Try IntestiCult™ Organoid Growth Medium (Mouse) in your own lab for efficient establishment and long-term maintenance of mouse intestinal organoids from intestinal crypts or single intestinal stem cells.
Training Programs
On-Demand Intestinal Organoid Course
Access free, self-paced training on how to culture mouse intestinal organoids.
Virtual or In-Person Intestinal Organoid Training
Gain experience in growing mouse intestinal organoids, with guidance from our team of scientific experts.
Request Pricing
Thank you for your interest in this product. Please provide us with your contact information and your local representative will contact you with a customized quote. Where appropriate, they can also assist you with a(n):
Estimated delivery time for your area
Product sample or exclusive offer
In-lab demonstration